Abstract
Amides are important intermediates in organic chemistry and the pharmaceutical industry, but their low reactivity requires catalysts and/or severe reaction conditions for esterification. Here, a novel approach was devised to convert amides into esters without the use of transition metals. The method effectively overcomes the inherent low reactivity of amides by employing dimethylsulfate-mediated reaction to activate the C-N bonds. To confirm the proposed reaction mechanism, control experiments and density functional theory (DFT) calculations were conducted. The method demonstrates a wide array of substrates, including amides with typical H/alkyl/aryl substitutions, N,N-disubstituted amides, amides derived from alkyl, aryl, or vinyl carboxylic acids, and even amino acid substrates with stereocentres. Furthermore, we have shown the effectiveness of dimethylsulfate in removing acyl protective groups in amino derivatives. This study presents a method that offers efficiency and cost-effectiveness in broadening the esterification capabilities of amides, thereby facilitating their increased utilization as synthetic compounds in diverse transformations.
Similar content being viewed by others
Introduction
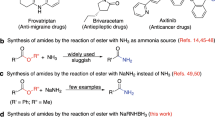
Amides play significant role as functional groups and important structural patterns in organic molecules.1,2,3,4 They serve as the fundamental units of proteins and can be found in various natural and synthetic compounds.5,6 They are generally considered to be weak electrophiles, which is mainly attributed to the resonance stability of the amide bond7. The stability and reactivity of the amide bond are associated with the planar resonance of the bond (15–20 kcal mol-1)8,9,10,11,12,13. However, any distortion of the amide bond, disrupting the planar conjugation, leads to notable changes in the physical and chemical properties of the amide. Additionally, this implies that the conversion of amides to other functional groups typically necessitates demanding experimental conditions and extended reaction durations. Specifically, the direct formation of esters from amides necessitates the application of highly acidic or alkaline conditions, which are unsuitable for the delicate functional groups. To address this limitation, various methods have been devised for the esterification of amides. Transition metal-catalyzed reactions can be considered as one of the most effective methods for achieving this goal14,15,16,17,18,19, as shown in Fig. 1a. Amides have been esterified by Garg and colleagues utilizing a catalyst consisting of Ni (0) /NHC20,21. Additionally, Danoun and colleagues22 have expanded upon this method by applying a cobalt system. Metal-free conditions are commonly used for amide esterification, with several protocols23,24,25,26 involving amide alcoholysis in basic conditions or Boc activation/base elimination (as shown in Fig. 1b). The conversion of amides to esters using alkyloxonium tetrafuoroborate salts has been previously reported in several studies27,28,29,30,31,32, involving the formation of imidate esters followed by subsequent hydrolysis to obtain amines and esters (as shown in Fig. 1c).The limited scope of the substrate still remains despite these advances. Developing a novel protocol without transition metals that can be applied to amides with normal H/alkyl/aryl substitutions remains a challenging and attractive task.
To overcome the longstanding issue related to the limited reactivity of amides and their limited application in C-N bond cleavage processes, we present the initial direct esterification of amides mediated by dimethyl sulfate (Fig. 1d). Notable aspects of our research comprise (a) the demonstration of a novel conceptual pathway for amide esterification utilizing dimethyl sulfate-mediated amide esterification; (b) the establishment of a single versatile dimethyl sulfate-mediated system capable of esterifying various amides; (c) the pioneering use of the easily accessible, stable, and cost-effective dimethyl sulfate, eliminating the need for expensive transition metal catalysts and ligands. Overall, this work demonstrates that dimethyl sulfate can significantly improve the esterification range of amides, which may contribute to the increasing use of amides as synthetics in various transformations. However, dimethyl sulfate is a known hazardous chemical33, highly toxic, seemingly harmless chemical commonly used in industry as a methylating agent. The substance can be readily absorbed through the skin or respiratory tract, potentially leading to poisoning or fatal outcomes during use, handling, or transportation33,34. To reduce potential hazards, the process should be confined to a controlled environment with adequate local exhaust ventilation. Operators should receive specialized training, adhere strictly to established procedures, and wear closed-circuit filter respirators (half-mask style), chemical safety goggles, enclosed gas suits, and rubber gloves to prevent exposure. It’s important to note that, empty containers or post-treatment reaction solutions may contain residual hazardous substances that necessitate proper disposal, which can be achieved by degrading residual hazardous materials with solutions of sodium hydroxide (1 mol/L), sodium carbonate (1 mol/L), or ammonia (1.5 mol/L)33,35.
Results and discussion
Optimization of reaction conditions
Amide 1a was chosen as a model substrate, dimethyl sulfate and methanol were mixed in a solvent, and the product methyl benzoate (2a) was observed to investigate our proposed transformation. We screened several solvents for coordination with amide 1a using dimethyl sulfate as a catalyst and found that only a trace of 2a was observed in acetonitrile (MeCN), tetrahydrofuran (THF), toluene, dioxane, and chlorobenzene (Table 1, entries 1–5). To our delight, after surveying various solvents, methanol was determined to be the most suitable reaction medium (Table 1, entries 6–7). It was evident that the esterification progressed at a sluggish pace when conducted at a temperature of 25 °C (Table 1, entry 6).
Reaction scope of alcohols for amide esterification
After optimizing the reaction conditions, we proceeded to assess the esterification of amides using the dimethyl sulfate mediated method. The scope of alcohols was investigated (Fig. 2). Under similar reaction conditions, esters 2a, 2ab, 2ac, 2ad, 2ae, and 2af were obtained in high yields (85–95%) from primary alcohols including methanol, ethanol, 1-butanol (n-BuOH), benzyl alcohol, isobutanol, and ethylene glycol. Nevertheless, when secondary alcohols (such as i-PrOH, s-BuOH or CyOH) and tertiary alcohols (like t-BuOH) were employed in accordance with our standard reaction procedure, the resulting esters 2ag, 2ah, 2ai, and 2aj exhibited diminished yields ranging from 25% to 50%.
Reaction scope of amides for esterification
In order to further investigate the extent and constraints of this direct esterification method, a diverse range of amides were employed to produce the intended outcomes using the standard reaction conditions. This protocol was found to be compatible with a diverse array of amides, as indicated in Table 2. Significantly, primary and secondary amides derived from substituted phenyl carboxylic acids could be esterified smoothly in comparable yields (55 − 93%). Furthermore, apart from the primary benzamide (1ab and 1ac), other compounds with the electron-withdrawing the nitroxyl, chloride or fluoride substituents (1c, 1cb, 1d, 1db, 1j and 1jb) or the electron-donating methoxy or methyl substituents (1b, 1bb, 1 h and 1hb) exhibited good compatibility. In addition to phenyl derivatives, we explored substrates of naphthyl and heterocyclic nature. Esterification of naphthyl compounds (1i and 1ib) and pyridine substrates (1 g and 1gb) resulted in high yields ranging from 83% to 93%. Additionally, amides derived from alkyl carboxylic acid substrates (1e, 1eb, 1 f and 1fb) were also amenable to the reaction. Motivated by the investigation of the esterification process of amides derived from alkyl carboxylic acids, our exploration commenced on 8-aminoquinoline amides. These amides serve as a remarkably efficient and extensively employed directing group amide, functioning as a bidentate auxiliary in diverse metal-catalyzed C − H functionalization reactions36,37,38,39,40,41,42,43. 8-Aminoquinoline amide derived from 3-phenylpropanoic acid (1k) reacted to give the corresponding ester (2k) in 92% yield. Among them, the chiral compounds, amides derived from amino acids (1 m, 1n and 1nn) were esterified to the corresponding ester products (2 m, 2 mb, 2n, 2nb, 2nn and 2nnb) in 80 − 93% yields. Additionally, it is worth mentioning that there was no racemization of ester products (Supplementary Figs. S1−S12) observed, which emphasizes the gentle reaction conditions that effectively hinder significant epimerization of the α stereocenters. We also found that the methodology tolerates α,β-unsaturated 8-aminoquinoline amide (1 l), N-acyl-succinicimide (1of), N-acyl glutarimide (1og), as well as some substrates (1pp, 1qq, 1rr) containing nucleophilic sites, such as amino or hydroxyl groups. This resulted in the formation of 2 l, 2of, 2og, 2pp, 2qq and 2rr, respectively, in good yields (85 − 93%). However, tertiary amide substrates (1ad, 1ae, 1af, 1ag, 1bc, 1cc, 1dc, 1ec, 1 fc, 1gc, 1hc, 1ic and 1jc) undergo the dimethyl sulphate-mediated esterification under our reaction conditions failed to give ester products in desirable yields
Cleavage of acyl protective group on amines with dimethyl sulphate
We next applied this methodology to the cleavage of acyl protective group on amines with dimethyl sulphate. In order to explore the scope and limitations of our method, we investigated a series of amino derivatives containing several acyl protective groups, for example acetyl group, benzoyl group, pivaloyl group, succinic acyl group, Boc and Cbz (Table 3). We began to investigate primary aromatic amino derivatives substrates, such as 8-aminoquinoline amides (1o, 1ob, 1oc, 1od, 1oe, 1of and 1og), 1-naphthylamine amide (1p, 1pb, 1 pc, 1pd and 1pe). We found that acetyl, benzoyl, pivaloyl, Boc and Cbz protective groups were easily removed under our cleavage conditions, leading to the corresponding amines (3o and 3p) in 85−95% yields; while the N-acyl-succinicimide 1of or N-acyl-glutarimide 1og counterparts required doubled-loading of dimethyl sulphate mediate and extended reaction time (24 hours) to achieve satisfactory reaction out comes in 85 − 88% yields. In addition, treatment of primary aliphatic amino derivatives (1q, 1qb, 1qc, 1qd and 1qe) using the current conditions provided the desired product 3q in 84 − 95% yields. Gratifyingly, the protocol was compatible with secondary amino derivatives (1r−1re, 1 s−1se, 1t−1td, 1 u−1ue and 1 v−1ve) containing acyl protective groups, and all of these reactions proceeded successfully, resulting in the formation of products 3r − 3 v with yields ranging from 60% to 88%. Afterwards, we examined the reaction’s tolerance by screening the chiral amino acids derivatives with either the Boc protective group (1w, 1x, and 1 y) or the Cbz protective group (1wb, 1xb, and 1yb).To our delight, the transformation also proceeded smoothly, in that the corresponding products (3w, 3x and 3 y) were furnished in 82 − 90% yields with >98% ee (Supplementary Figs. S13−S18).
Mechanistic studies
In order to gain insight into the esterification mechanism of amides mediated by dimethylsulfate, representative time course of the esterification of 1a and a series of control experiments were conducted. The kinetic behavior of the esterification reaction with dimethyl sulfate-mediattion was studied using GC. As depicted in Table 4 (Fig. 3), the esterification reaction was observed to occur in three stages: an induction period (Fig. 3, 0 − 60 min), an active period characterized by rapid formation of product 2ac (60 − 240 min), and a final period with significantly reduced reaction rate ( > 240 min). These experiments aimed to capture and isolate any potential reaction intermediates, as depicted in Fig. 4a. Specifically, the reaction between amide 1z and dimethylsulfate was conducted at a temperature of 75 °C for an extended duration in the absence of alcohols (Fig. 4a). It is worth mentioning that a yield of 95% was achieved for INT-1z (Supplementary Data 4 and Supplementary Table S2−S8), with no detection of 2z, which is believed to be a potential intermediate for the esterification process. Fortunately, INT-1z could be readily converted to ester 2z with a high yield of 93% by heating it at 100 °C in n-BuOH for a few hours (Fig. 4a). A similar outcome was observed when amide 1z was reacted with methyl hydrogen sulfate in MeOH at a temperature of 50 °C for a duration of 30 min, or under ambient conditions for a duration of 24 hours. It is worth noting that the compound INT-3z, which is O-protonated amide, was successfully isolated (Fig. 4b). This compound serves as a possible intermediate for the esterification process. Upon heating compound INT-3z at 100 °C in n-BuOH for several hours, ester 2z was obtained with a high yield of 88% (Fig. 4b). The isolation and conversion of INT-1z and INT-3z demonstrate that dimethylsulfate plays a significant role in the formation of these intermediates, which are essential for the esterification reaction.
a Control experiment for capture and transformation of INT-1z. b Control experiment for capture and transformation of INT-3z. c DFT methods were used to calculate the dimethylsulfate-mediated activation of amide C–N bonds for the amide-to-ester conversion.The calculation is performed at the M06-2X SMD/6-311 + + G(d,p)//M06-2X PCM/6-31 G(d) level of theory. The relative Gibbs free energies (ΔGsol) are represented in kcal/mol.
To gain a deeper understanding of this novel amide esterification pathway, density functional theory (DFT) calculations were conducted at M06-2X SMD/6-311 + + G(d,p)//M06-2X PCM/6-31 G(d) level. Drawing upon both experimental and theoretical evidence, the mechanism of the reaction was proposed (Fig. 4c and Supplementary Table S1). 1a and dimethyl sulfate was selected as the zero-point of the potential surface (1a + Me2SO4, 0.0 kcal/mol). Firstly, the methylation of carbonyl oxygen by dimethyl sulfate occurs through transition state TS1 (25.2 kcal/mol) to form imidoesterification intermediate INT-1. Then the oxygen atom of n-butanol attacks the carbon atom of INT-1 through TS-2 (26.8 kcal/mol), yielding INT-2. INT-1 has been observed in the experiment, probably because of the stability of this amide salt and a relatively high energy barrier. Next, INT-2 transfers into INT-3, where the hydrogen bond species is converted into an O-protonated amide species. The formation of INT-3 has been proved by control experiment and it sets the stage for the next step. INT-3 undergoes a transformation to yield INT-4 via TS-3 (27.1 kcal/mol), where a second molecule of n-butanol attacks the carbocation. Afterwards, a proton transfer happens, giving rise to two new hydrogen bonds within INT-5. Ultimately, INT-5 undergoes an amino group departure process TS-4 (10.6 kcal/mol) to form the desired product 2ac.
In summary, a novel and efficient transition-metal-free method has been devised for the direct esterification of amides through the activation of C-N bonds, employing dimethyl sulfate-mediated reaction. This approach successfully addresses the inherent reactivity limitations of amides as functional groups and offers a diverse array of substrates amenable to different transformations. Control experiments and Density Functional Theory (DFT) calculations have contributed to a more comprehension of the proposed mechanism governing the amide esterification pathway. This mechanism entails an initial imidoesterification step, succeeded by n-butanol attacks resulting in the formation of an O-protonated amide and the subsequent departure of an amino group. The protocol presented in this study exhibits remarkable versatility and operates under mild conditions. Furthermore, its metal-free composition supplements the conventional transition-metal-catalyzed esterification of amides. Consequently, this protocol represents a promising and viable new pathway for achieving catalytic cleavage of amide C-N bonds, yielding up to 95%. The findings of this research significantly broaden the scope of esterification of amides, thereby facilitating their increased utilization as synthetic intermediates in the fields of organic chemistry and the pharmaceutical industry.
Methods
General procedure for amide esterification (Supplementary Note 1)
To a solution of amides 1 (1.0 equiv) in alcohols (0.2 M) was added dimethyl sulfate (1.0 equiv), and heated for 8 − 24 h at 65 − 120 °C in a sealed vial under an atmosphere of N2 (monitored by TLC). The resulting mixture concentrated in vacuo to give residues. Then the residues were dissolved in ethyl acetate and washed with brine, dried over Na2SO4, filtered, and concentrated in vacuo. The crude product was purified by silica-gel column chromatography to give ester products 2.
General procedure for cleavage of acyl protective group on amines with dimethyl sulphate
To a solution of amides 1 (1.0 equiv) in n-butanol (0.2 M) was added dimethyl sulfate (1.0 equiv), and heated for 8 h at 120 °C in a sealed vial under an atmosphere of N2 (monitored by TLC). The resulting mixture concentrated in vacuo to give residues. Then the residues were dissolved in ethyl acetate and washed with sodium bicarbonate saturated solution, brine, dried over Na2SO4, filtered, and concentrated in vacuo. The crude product was purified by silica-gel column chromatography to give amine products 3.
Data availability
All data supporting the findings of this study are available within this article and its Supplementary Information file. The copies of 1H NMR and 13C NMR spectra of the compounds obtained in this manuscript are available in Supplementary Data 1. Source Data for Supplementary Tables S1−S8 are available within Supplementary Data 2. Source Data for Supplementary Figs. S1-S18 for copies of compound chromatograms obtained in this manuscript are provided in Supplementary Data 3. Cif (crystallographic data) for compound INT-1z as Supplementary Data 4. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition number 2347148 (for INT-1z). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. The data are also available from the corresponding author upon reasonable request.
References
Bode, J. Emerging methods for the synthesis of amide and peptide bonds. Curr. Opin. Drug Discovery Dev. 9, 765–775 (2006).
Clayden, J. Book Review: The Amide Linkage Structural Significance in Chemistry, Biochemistry, and Materials Science. Edited by Arthur Greenberg, Curt M. Breneman and Joel F. Liebman. Angew. Chem. Int. Ed. 42, 1788–1789 (2003).
Cupido, T. et al. The synthesis of naturally occurring peptides and their analogs. Curr. Opin. Drug Discovery Dev. 10, 768–783 (2007).
Greenberg, A., Breneman, C. M. & Liebman, J. F. The amide linkage: Structural significance in chemistry, biochemistry, and materials science. 76–79 (John Wiley & Sons Inc.; Hoboken, NJ, USA, 2000).
Pauling, L., Corey, R. B. & Branson, H. R. The structure of proteins: two hydrogenbonded helical configurations of the polypeptide chain. Proc. Natl Acad. Sci. USA 37, 205–211 (1951).
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among US FDA approved pharmaceuticals: miniperspective. J. Med. Chem. 57, 10257–10274 (2014).
Greenberg, A., Breneman, C. M. & Liebman, J. F. (eds) The Amide Linkage: Structural Significance in Chemistry, Biochemistry, and Materials Science. 33–58 (John Wiley & Sons Inc.; Hoboken, NJ, USA, 2003)
Basch, H. & Hoz, S. Resonance in formamide: resolution of contending models. Chem. Phys. Lett. 294, 117–125 (1998).
Ilieva, S., Hadjieva, B. & Galabov, B. Theory supplemented by experiment. Electronic effects on the rotational stability of the amide group in p-substituted acetanilides. J. Org. Chem. 67, 6210–6215 (2002).
Kemnitz, C. R. & Loewen, M. J. Amide resonance correlates with a breadth of C− N rotation barriers. J. Am. Chem. Soc. 129, 2521–2528 (2007).
Lauvergnat, D. & Hiberty, P. C. Role of conjugation in the stabilities and rotational barriers of formamide and thioformamide. An ab initio valence-bond study. J. Am. Chem. Soc 119, 9478–9482 (1997).
Quiñonero, D. et al. The resonance model in amides: a ̀ combined crystallographic and ab initio investigation. New J. Chem. 25, 259–261 (2001).
Eisenberg, D. The discovery of the α-helix and β-sheet, the principal structural features of proteins. Proc. Natl Acad. Sci. USA 100, 11207–11210 (2003).
Ouyang, K. et al. Transition-metal-catalyzed cleavage of C-N single bonds. Chem. Rev. 115, 12045–12090 (2015).
Wang, Q. et al. Transition-metal catalysed C–N bond activation. Chem. Soc. Rev. 45, 1257–1272 (2016).
Deguchi, T. et al. Direct catalytic alcoholysis of unactivated 8-aminoquinoline amides. ACS Catal. 7, 3157–3161 (2017).
Kita, Y. et al. Zinc-catalyzed amide cleavage and esterification of β-hydroxyethylamides. Angew. Chem. Int. Ed. 51, 5723–5726 (2012).
Kita, Y. et al. Combined catalytic system of scandium triflate and boronic ester for amide bond cleavage. Adv. Synth. Catal. 355, 3391–3395 (2013).
Hirai, T. et al. Esterification of tertiary amides: remarkable additive effects of potassium alkoxides for generating hetero manganese–potassium dinuclear active species. Chem. Eur. J. 26, 10735 (2020).
Hie, L. et al. Conversion of amides to esters by the nickel-catalysed activation of amide C–N bonds. Nature 524, 79–83 (2015).
Hie, L. et al. Nickel-catalyzed esterification of aliphatic amides. Angew. Chem. Int. Ed. 55, 15129–15132 (2016).
Bourne-Branchu, Y., Gosmini, C. & Danoun, G. Cobalt-catalyzed esterification of amides. Chem. Eur. J. 23, 10043–10047 (2017).
Li, G., Lei, P. & Szostak, M. Transition-metal-free esterification of amides via selective N-C cleavage under mild conditions. Org. Lett. 20, 5622–5625 (2018).
Ye, D. et al. Cesium carbonate catalyzed esterification of N-benzyl-N-boc-amides under ambient conditions. Org. Lett. 21, 6888–6892 (2019).
Huang, C. et al. Hydrogen-bond-assisted transition-metal-free catalytic transformation of amides to esters. Sci. China Chem. 64, 66–71 (2021).
Wu, H. et al. Fluoride-catalyzed esterification of amides. Chem. Eur. J. 24, 3444–3447 (2018).
Sheehan, J. C. & Nafssi, V. M. M. α-Lactams. VIII. O-Alkylation of α-Lactams. J. Org. Chem. 35, 4246–4248 (1970).
Kiessling, A. & McClure, C. K. S. The Conversion of Amides to Esters with Meerwein’S Reagent. Application to the Synthesis of a Carfentanil Precursor. Synth. Commun. 27, 923–937 (1997).
Hanessian, S. Selective hydrolysis of amide bonds in acetamido deoxy sugars. o-Ethyl acetamidium fluoroborates. Tetrahedron Lett. 8, 1549–1552 (1967).
Kishi, Y. et al. Synthetic studies on tetrodotoxin and related compounds. IV. Stereospecific total syntheses of DL-tetrodotoxin. J. Am. Chem. Soc. 94, 9219–9221 (1972).
Menezes, R. & Smith, M. B. A mild and facile route to ω-amino esters. Synth. Commun. 18, 1625–1636 (1988).
Keck, G. E., McLaws, M. D. & Wager, T. T. A direct and mild conversion of tertiary aryl amides to methyl esters using trimethyloxonium tetrafluoroborate: a very useful complement to directed metalation reactions. Tetrahedron 56, 9875–9883 (2000).
Dimethyl suphate-Hazardous Substance Fact Sheet. 1–6 https://nj.gov/health/eoh/rtkweb/documents/fs/0768.pdf (2007).
Zhang, J. et al. Dimethyl sulfate poisoning in China: a fatal case and a 45-year retrospective study. Forensic. Sci. Med. Pathol. Published online November 4 1–8 (2023).
Lunn, G. & Sansone, E. B. Validation of techniques for the destruction of dimethyl sulfate. Am Ind Hyg Assoc J. 46, 111–114 (1985).
Lee, S. J. et al. Cu-mediated aminoquinoline-directed radiofluorination of aromatic C-H bonds with K8 F. Angew. Chem. Int. Ed. 58, 3119–3122 (2019).
Mondal, S. & Hajra, A. Ruthenium(II)-catalyzed remote C-H addition of 8-aminoquinoline amide to activated aldehyde. Org. Biomol. Chem. 16, 2846–2850 (2018).
Ghosh, T., Maity, P. & Ranu, B. C. Cu(OAc)2-promoted ortho C(sp(2))-H amidation of 8-aminoquinoline benzamide with acyl azide: selective formation of aroyl or acetyl amide based on catalyst loading. J. Org. Chem. 83, 11758–11767 (2018).
Zhang, Z. Z. et al. Synthesis of Bicyclo n.1.0 alkanes by a Cobalt-Catalyzed Multiple C(sp(3))-H Activation Strategy. Angew. Chem. Int. Ed. 56, 13145–13149 (2017).
Xia, H. et al. Palladium-catalyzed direct sulfonylation of C-H bonds with the insertion of sulfur dioxide. Chem. Commun 53, 12548–12551 (2017).
Nguyen, T. T. & Daugulis, O. Palladiumcatalyzed, aminoquinoline-directed arylation of phosphonamidate and phosphinic amide sp(3) C-H bonds. Chem. Commun 53, 4609–4611 (2017).
Nguyen, T. T., Grigorjeva, L. & Daugulis, O. Cobalt catalyzed, aminoquinoline-directed functionalization of phosphinic amide sp(2) C-H Bonds. ACS Catal 6, 551–554 (2016).
Sattar, M. et al. Functionalization of ferrocene-carboxylic acid by using 8-Aminoquinoline as a Removable Directing Group. Adv. Synth. Catal 358, 240–253 (2016).
Acknowledgements
This work was supported financially by the West Light Foundation of The Chinese Academy of Sciences (Grant No. 2018-XBYJRC-001) and Special Foundation of Chinese Academy of Sciences for strategic pilot technology (grant no. XDA12050411).
Author information
Authors and Affiliations
Contributions
H.Q., Z.H., and J.S. conceived the concept and prepared the manuscript. H.Q., Z.H., and H.S. conducted the experiments and analyzed the data. E.B., A.O., A.S., and F.Z. provided revisions. All the authors participated in the discussion and preparation of the manuscript. J.S., W.Z., and H.A. directed the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interest.
Peer review
Peer review information
Communications Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Qin, H., Han, Z., Bonku, E.M. et al. Direct esterification of amides by the dimethylsulfate-mediated activation of amide C–N bonds. Commun Chem 7, 93 (2024). https://doi.org/10.1038/s42004-024-01180-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-024-01180-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.