Abstract
NGLY1 deficiency is a genetic disease caused by biallelic mutations of the Ngly1 gene. Although epileptic seizure is one of the most severe symptoms in patients with NGLY1 deficiency, preclinical studies have not been conducted due to the lack of animal models for epileptic seizures in NGLY1 deficiency. Here, we observed the behaviors of male and female Ngly1−/− mice by video monitoring and found that these mice exhibit spontaneous seizure-like behaviors. Gene expression analyses and enzyme immunoassay revealed significant decreases in oxytocin, a well-known neuropeptide, in the hypothalamus of Ngly1−/− mice. Seizure-like behaviors in Ngly1−/− mice were transiently suppressed by a single intranasal administration of oxytocin. These findings suggest the therapeutic potential of oxytocin for epileptic seizure in patients with NGLY1 deficiency and contribute to the clarification of the disease mechanism.
Similar content being viewed by others
Introduction
Peptide:N-glycanase (NGLY1) is an enzyme that removes the N-linked glycans from misfolded N-glycoproteins1,2. NGLY1 is mainly localized in the cytoplasmic side of the endoplasmic reticulum (ER) and involved in the ER-associated degradation of misfolded N-glycoproteins, which plays a crucial role in the quality control of newly synthesized N-glycoproteins3,4. NGLY1 also activates the transcription factor NRF1 by deglycosylating its glycan chains and substituting their amino acid from asparagine to aspartic acid residues5,6.
NGLY1 deficiency is a genetic disease caused by biallelic mutations in the Ngly1 gene7. Patients with NGLY1 deficiency exhibit several symptoms, including developmental delay, intellectual disability, movement disorders, alacrima, and epileptic seizures8. The first patient with NGLY1 deficiency was reported in 20129. The number of patients has increased to over 10010; however, a satisfactory treatment method has not yet been established for NGLY1 deficiency, probably due to the lack of knowledge on the disease mechanism11,12,13.
We previously established Ngly1−/− mice and Ngly1−/− rats by deleting the exons 11 and 12 of the Ngly1 gene. These animals exhibited developmental delay and movement disorders14,15. Movement disorders in Ngly1−/− rats were significantly improved by a single intracerebroventricular administration of AAV-NGLY116,17. This replacement therapy could be a fundamental treatment, but it requires invasive surgery. Other treatment options are anticipated to reduce patient burden.
For epileptic seizures in patients with NGLY1 deficiency, some approved drugs, including levetiracetam, valproate, lamotrigine, and clobazam, have been tested; however, their efficacies were limited11. Researching the disease mechanism is necessary to discover more effective anti-epileptic drugs. The Ngly1−/− mouse model is a candidate preclinical model of epileptic seizure in NGLY1 deficiency that is used to uncover the disease mechanism, although it is unknown whether Ngly1−/− mice exhibit epileptic seizures.
Herein, we observed the behaviors of Ngly1−/− mice by video monitoring and found that these mice exhibited seizure-like behaviors spontaneously. Gene expression analyses and enzyme immunoassay revealed significant decreases in oxytocin in the hypothalamus of Ngly1−/− mice. The effects of intranasal oxytocin administration on the seizure-like behaviors in Ngly1−/− mice were evaluated to confirm the involvement of oxytocin with seizure-like behaviors in Ngly1−/− mice.
Results
Spontaneous seizure-like behaviors in Ngly1 −/− mice
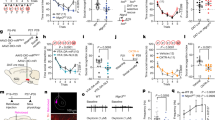
To clarify whether Ngly1−/− mice exhibit epileptic seizures, we observed the behaviors of male and female Ngly1−/− mice by video monitoring for 24 h. As shown in the representative figures and video of female Ngly1−/− mice, these mice exhibited several seizure-like behaviors spontaneously (Fig. 1a, Supplementary Movie 1). These behaviors seemed to be tonic-clonic or absence seizures, characterized by body stiffness and rhythmical jerking of the limbs or a sudden lapse in awareness. The average frequencies of seizure-like behaviors in 4 and 10-week-old male Ngly1−/− mice were 21 and 28 episodes per 12-hour light phase, 12 and 14 episodes per 12-hour dark phase, respectively. Those of female Ngly1−/− mice were 18 and 26 episodes per 12 h-light phase, 11 and 15 episodes per 12-hour dark phase, respectively. Ngly1+/+ and Ngly1+/− mice did not exhibit any seizure-like behavior in both light and dark phases at 4 and 10-week-old (Fig. 1b). Although the time zone of occurrence varied individually, almost all mice did not exhibit seizure-like behaviors at the beginning of the dark phase (zeitgeber time 12–13) when they began to move actively (Fig. 1c).
a Representative photographs of spontaneous seizure-like behaviors in female Ngly1−/− mice. b Frequencies of seizure-like behaviors in male and female Ngly1+/+, Ngly1+/−, and Ngly1−/− mice (4 and 10-week-old). Data represents mean and individuals (N = 3–7). c Hourly variation of seizure-like behaviors in male and female Ngly1−/− mice (4 and 10-week-old). Data represent mean + standard deviation and individual values (N = 6 or 7).
Transcriptome analyses in the brain and spinal cord of male Ngly1 −/− mice
To elucidate the disease mechanism of seizure-like behaviors in Ngly1−/− mice, we analyzed the differences in gene expression profiles in the cerebellum, hippocampus, thalamus, striatum, cerebral cortex, and spinal cord of 4 and 10-week-old male Ngly1−/− and Ngly1+/+ mice. Principal component analysis (PCA) of the transcriptome data depicted the differences between these tissues, whereas differences between ages or Ngly1−/− and Ngly1+/+ mice were unclear in the PCA plot (Fig. 2a). However, volcano plots of the transcriptome data revealed significant decreases in oxytocin transcripts in the thalami of 4-week-old Ngly1−/− mice (Fig. 2b). Significant decreases in oxytocin transcripts were also detected in the striata of 4-week-old Ngly1−/− mice and the thalami of 10-week-old Ngly1−/− mice (Fig. 2c). Although 159 Gene Ontology terms were obtained from the enrichment analysis of differentially expressed genes in Ngly1−/− mice, NGLY1-related terms, such as proteasome and autophagy, were not included among the 159 terms. Instead, 21 neurotransmission-related terms were included among the 159 terms, suggesting an abnormal neurotransmission in Ngly1−/− mice (Supplementary Table 1).
Cerebellum (CBM), hippocampus (HIP), thalamus (THAL), striatum (STR), cerebral cortex (CC), and spinal cord (SC) were collected from male Ngly1+/+ (WT) and Ngly1−/− (KO) mice (4 and 10-week-old). Total RNAs were extracted from each tissue. RNA sequencing was performed on the DNBSEQ platform in Azenta. a Principal component analysis plot of all results. X and Y axes show principal component 1 (PC1) and principal component 2 (PC2) for the data set. The explained variance is indicated in each plot axis. b Volcano plot of the results in the thalami of 4-week-old male Ngly1−/− mice. X axis represents the log2 of the fold change; Y-axis represents the negative decade logarithm of the significance value. Oxytocin (Oxt) is marked with a red circle. c Expression levels of oxytocin transcripts in 4- and 10-week-old mice. Data represent mean + standard deviation and individual values (N = 3). n.s., not significant; *, p < 0.05; **, p < 0.01 vs. Ngly1+/+ by Bayes algorithm.
Specific down-regulation of oxytocin transcripts in the hypothalamus of Ngly1 −/− mice
The expression levels of oxytocin transcripts were also measured using quantitative PCR (Supplementary Tables 2 and 3) in the hypothalamus of 10-week-old male and female Ngly1−/− mice because the hypothalamus is the main production site of oxytocin. The decreased rates of oxytocin transcripts in Ngly1−/− mice were 56% (male) and 58% (female) compared to those of Ngly1+/+ mice. Oxytocin levels were not significantly changed in male and female Ngly1+/− mice compared to those of Ngly1+/+ mice (Fig. 3a). Transcripts of other major hypothalamic neuropeptides, i.e., vasopressin and corticotropin-releasing hormone, were not significantly changed in male and female Ngly1−/− mice compared to those of Ngly1+/+ mice, suggesting that among the hypothalamic neuropeptides, the down-regulation was specific to oxytocin (Fig. 3b, c).
Hypothalamus was collected from 10-week-old male and female Ngly1+/+, Ngly1+/− and Ngly1−/− mice. Transcripts of a oxytocin, b vasopressin, and c CRH were measured using quantitative PCR and normalized to transcripts of the neuronal marker MAP2. Data represent mean + standard deviation and individual values (N = 7). n.s., not significant; **p < 0.01 vs. Ngly1+/+ by Dunnett’s test. CRH, corticotropin-releasing hormone.
Oxytocin peptide levels in the hypothalamus, pituitary gland, and plasma of Ngly1 −/− mice
To confirm oxytocin down-regulation at peptide levels, we quantified oxytocin peptides in the hypothalamus of 10-week-old male and female Ngly1−/− mice. The decrease rates of oxytocin peptides in Ngly1−/− mice were 78% (male) and 66% (female) compared to those of Ngly1+/+ mice (Fig. 4a). Down-regulation of oxytocin peptides was also detected in the pituitary gland of Ngly1−/− mice, which is the secretory tissue of oxytocin. The decrease rates were 79% (male) and 76% (female) compared to those of Ngly1+/+ mice (Fig. 4b). At that timing, down-regulation of oxytocin peptides was not detected in the plasma of Ngly1−/− mice, probably due to the diurnal variation and rapid clearance of plasma oxytocin (Supplementary Fig. 1). Therefore, plasma oxytocin levels were repeatedly measured every 27 h four times in 7-week-old male and female Ngly1−/− mice. Although statistical significance was not detected at most intervals, plasma oxytocin levels showed a tendency to be decreased in Ngly1−/− mice compared to Ngly1+/+ mice (Fig. 4c). Down-regulation of oxytocin peptides was further confirmed in the hypothalamus and pituitary gland of 29-week-old male Ngly1−/− mice to increase the credibility of oxytocin down-regulation (Supplementary Fig. 2). In all experiments, oxytocin levels were not significantly changed in Ngly1+/− mice compared to Ngly1+/+ mice.
a Hypothalamus, and b pituitary gland were collected from 10-week-old male or female Ngly1+/+, Ngly1+/−, and Ngly1−/− mice (N = 5). c Plasma was collected from 7-week-old male or female Ngly1+/+, Ngly1+/−, and Ngly1−/− mice (N = 3). Oxytocin levels were measured using enzyme immunoassay. Data represent mean + standard deviation and individual values. n.s., not significant; *, p < 0.05; **, p < 0.01; ***, p < 0.001 vs. Ngly1+/+ by Dunnett’s test.
Oxytocin levels in Ngly1 −/− mice after intranasal administration of oxytocin
Delivery of intranasal oxytocin to the central nervous system was confirmed using 17 to 18-week-old female Ngly1−/− mice. Dosing was initiated at 10 or 100 mg/kg (6000 or 60,000 IU/kg) due to a lack of effect on seizure-like behaviors in Ngly1−/− mice at 1 and 3 mg/kg in our preliminary study. Five minutes after the single intranasal administration, oxytocin levels were increased in the plasma, hippocampus, striatum, cerebral cortex, and spinal cord of Ngly1−/− mice, suggesting that this regimen could be applied when evaluating the effects of oxytocin on seizure-like behaviors in Ngly1−/− mice (Fig. 5a–f). An increasing trend was also observed in the thalamus, whereas it was not observed in the hypothalamus, probably because the endogenous oxytocin levels were relatively high compared to that in other tissues (Fig. 5g, h).
Oxytocin (10, 100 mg/kg) or saline was intranasally administered to female Ngly1−/− mice (17 or 18-week-old) under anesthesia with 2.5% isoflurane (a). b Plasma, c hippocampus, d striatum, e cerebral cortex, f spinal cord, g thalamus, and h hypothalamus were collected 5 min after the single administration. Oxytocin levels were measured using enzyme immunoassay. Data represent mean + standard deviation and individual values (N = 3).
Transient suppression of seizure-like behaviors by intranasal oxytocin in Ngly1 −/− mice
Effects of intranasal oxytocin administration on seizure-like behaviors in Ngly1−/− mice were evaluated in crossover studies using 12-week-old male and female Ngly1−/− mice (Fig. 6a). Seizure-like behaviors in male and female Ngly1−/− mice were transiently suppressed by intranasal oxytocin administration. The differences of mean episodes per 4 h-light phase (and two-sided 95% confidence interval) compared to control group (0 mg/kg) were −3.3 (−10.2, 3.7) at 10 mg/kg, −2.5 (−9.1, 4.1) at 30 mg/kg, −8.5 (−14.8, −2.2) at 100 mg/kg in male Ngly1−/− mice and −9.0 (−17.1, −0.9) at 10 mg/kg, −11.0 (−21.4, −0.6) at 30 mg/kg, −14.5 (−22.8, −6.2) at 100 mg/kg in female Ngly1−/− mice, respectively (Fig. 6b).
a Experimental schedule of crossover studies. b Oxytocin (0, 10, 30, 100 mg/kg in saline) was intranasally administered to 12-week-old male or female Ngly1−/− mice under anesthesia with 2.5% isoflurane. Seizure-like behaviors were monitored for 4 h. This assessment was carried out with a crossover design with washout periods of 20 h. Data represent mean + standard deviation and individual values (N = 4). The estimated differences in seizure-like behaviors between oxytocin-treated groups (10, 30, 100 mg/kg) and control group (0 mg/kg) and their adjusted two-sided 95% confidence intervals (CI) were calculated.
Discussion
Although epileptic seizure is one of the most severe symptoms in patients with NGLY1 deficiency, preclinical studies have not been carried out due to the lack of animal models for epileptic seizures in NGLY1 deficiency. Here we firstly clarified that Ngly1−/− mice exhibited several seizure-like behaviors spontaneously (Fig. 1, Supplementary Movie 1). This finding enabled us to analyze the disease mechanism of epileptic seizure in NGLY1 deficiency and explore treatment options.
Significant decreases in oxytocin transcripts were detected in the thalami of male Ngly1−/− mice (Fig. 2). Oxytocin is a hypothalamic neuropeptide known for its role in parturition18,19, lactation20, and social interaction21. Oxytocin is also reported to play an important role in the maintenance of brain homeostasis by regulating stress-related hormones, such as corticotropin-releasing hormone, adrenocorticotropic hormone and corticosterone, in the hypothalamic–pituitary–adrenal (HPA) axis22,23,24,25. Dysregulation of these hormones in HPA axis is a risk factor for epileptic seizure26. Therefore, it was speculated that the downregulation of oxytocin in Ngly1−/− mice could cause the dysregulation of HPA axis and led to seizure-like behaviors (Fig. 1). The facts that seizure-like behaviors were not detected in Ngly1+/− mice (Fig. 1) and that oxytocin was not significantly decreased in Ngly1+/− mice (Figs. 3 and 4) also reinforced our hypothesis.
To evaluate the effect of oxytocin on seizure-like behaviors in Ngly1−/− mice, oxytocin must be delivered to the central nervous system (CNS) of Ngly1−/− mice. Delivery of oxytocin to the CNS had already been demonstrated by intranasal administration in mice27, rats28, rhesus macaques29, and patients with autism spectrum disorder30. Therefore, we selected intranasal administration of oxytocin and confirmed its delivery to the CNS in Ngly1−/− mice (Fig. 5).
Seizure-like behaviors in Ngly1−/− mice were transiently suppressed by single intranasal administration of oxytocin (Fig. 6). The safety of single or chronic intranasal oxytocin administration has been investigated in several clinical studies. For example, no side-effect was detected in school-age children (8–12 years old) 90 min and 24 h after a single administration31; chronic administration (twice daily for four weeks) was well-tolerated in generally healthy older men (55–95 years old)32. Based on these clinical studies, oxytocin has the potential to be administered to patients with NGLY1 deficiency by carefully optimizing the dosing regimen.
This study has some limitations. While the down-regulation of oxytocin in the hypothalamus of Ngly1−/− mice was clarified by bulk RNA-seq analyses, the detailed mechanism of oxytocin down-regulation by Ngly1 deletion could not be clarified by our subsequent single-nucleus RNA-seq analyses in the hypothalamus of Ngly1−/− mice. Other approaches are required to clarify this mechanism.
In conclusion, we observed spontaneous seizure-like behaviors in Ngly1−/− mice and the down-regulation of oxytocin in the hypothalamus of Ngly1−/− mice. Our results also demonstrated the suppressive effects of intranasal oxytocin administration on seizure-like behaviors in Ngly1−/− mice. These findings suggest the therapeutic potential of oxytocin for epileptic seizures in patients with NGLY1 deficiency and would contribute to the clarification of the disease mechanism.
Methods
Approval of animal experiments
All animal care procedures and experiments conformed to the Association for Assessment and Accreditation of Laboratory Animal Care guidelines and were approved by the Institutional Animal Care and Use Committee of Takeda Pharmaceutical Co., Ltd. (Kanagawa, Japan).
Establishment and breeding of Ngly1 −/− mice
Homozygous Ngly1 knockout (Ngly1−/−) mice, heterozygous Ngly1 knockout (Ngly1+/−) mice, and wild-type (Ngly1+/+) littermates were established previously by mating C57BL/6 background Ngly1+/− mice with Japanese fancy mouse 1 background Ngly1+/− mice to rescue the embryonic lethality of Ngly1−/− mice15. These mice were bred and provided by Axcelead Drug Discovery Partners (Kanagawa, Japan).
Video analysis of seizure-like behaviors in Ngly1 −/− mice
Mice were habituated to single housing for one day in clear acrylic cages (W15 cm, D15 cm, H25 cm) with vents and water insertion slots. The next day, videos were recorded for 24 h from the top of the cage using an infrared camera. Seizure-like behaviors were manually counted using HDWriterAE5.4 software (Panasonic, Osaka, Japan) in a blinded manner by Orizuru Therapeutics (Kanagawa, Japan). All types of seizure-like behaviors were counted as one regardless of the degree and duration. These video analyses were repeated using the same mice at 4 and 10 weeks old.
Transcriptome analyses in the brain tissues and spinal cord of Ngly1 −/− mice
The cerebellum, hippocampus, thalamus, striatum, cerebral cortex, and spinal cord were collected from 4- and 10-week-old male Ngly1+/+ and Ngly1−/− mice (N = 3). Total RNA was extracted from each tissue using an RNeasy Mini QIAcube kit (QIAGEN, Hilden, Germany). RNA sequencing was performed on the DNBSEQ platform in Azenta. Transcriptome data was analyzed by Axcelead Drug Discovery Partners. Differentially expressed genes were identified using the edgeR (3.1.1) and limma (3.46.0) R packages. In brief, gene expression counts were initially transformed to counts per million (CPM). Subsequently, genes with low expression, where the minimum log-CPM was less than or equal to zero, were removed. Data were then subjected to the trimmed mean of M-values normalization.
Quantification of oxytocin and other transcripts using real-time PCR
Total RNAs were extracted from the hypothalamus of 10-week-old male and female Ngly1+/+, Ngly1+/− and Ngly1−/− mice using the RNeasy Mini QIAcube kit (N = 7, combined two experiments). The RNAs were reverse-transcribed using the High-Capacity RNA to cDNA kit (ThermoFisher Scientific, Tokyo, Japan). Transcripts of oxytocin, vasopressin, CRH, and MAP2 were quantified using a 7900HT Fast Real-Time PCR system (ThermoFisher Scientific). Primers, probes, and standard oligos for quantitative PCR were designed using Primer ExpressTM software v3.0.1 (ThermoFisher Scientific), and their sequences are listed in Supplementary Tables 2 and 3.
Measurement of oxytocin peptides using enzyme immunoassay
Plasma was collected every 27 h four times from the left ventricles of 7-week-old male and female Ngly1+/+, Ngly1+/−, and Ngly1−/− mice (N = 3) under anesthesia with 2.5% isoflurane. The hypothalamus and pituitary gland were extracted after euthanasia from 10-week-old male and female Ngly1+/+, Ngly1+/−, and Ngly1−/− mice (N = 5). Oxytocin levels were measured by a chemiluminescent enzyme immunoassay in ASKA Pharma Medical Co., Ltd. (Kanagawa, Japan).
Intranasal administration of oxytocin
Oxytocin peptide (CAS number 50-56-6) was purchased from AA Blocks (San Diego, CA, USA) and dissolved in saline at a maximum concentration of 200 mg/mL. Female Ngly1−/− mice were anesthetized with 2.5% isoflurane and intranasally administered with oxytocin using a manual pipette at a volume of 0.5 ml/kg. Oxytocin solutions were prepared in Protein LoBind Tubes (Eppendorf, Hamburg, Germany) just before the intranasal administration.
Crossover studies to evaluate the effects of oxytocin on seizure-like behaviors
Oxytocin (0, 10, 30, 100 mg/kg in saline) was intranasally administered to 12-week-old male and female Ngly1−/− mice. Seizure-like behaviors were counted just after the single administration for 4 h, as previously described. This assessment was carried out with a crossover design with washout periods of 20 h.
Statistical analyses
Unless otherwise specified, the results were analyzed using EXSUS2014 software version 8.0, SAS 9.3 TS Levle1M2 (SAS Institute, Inc., NC). Limma package, with its voom method33, was used to identify differentially expressed genes. Thresholds for identification were a p value less than 0.05 and an absolute log-FC (fold change) greater than 1. Bayes algorithm was used to test the statistical significance of differences between Ngly1+/+ and Ngly1−/− mice. The estimated differences in seizure-like behaviors between oxytocin-treated groups (10, 30, 100 mg/kg) and the control group (0 mg/kg) and their adjusted two-sided 95% confidence intervals were calculated to evaluate effects of oxytocin on seizure-like behaviors in male and female Ngly1−/− mice. The sample size was chosen based on our preliminary studies with similar methods.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data supporting the findings described in this manuscript are available in the Supplementary Data and from the corresponding author upon reasonable request. Ngly1−/− mice, Ngly1+/− mice, and Ngly1+/+ littermates will be available upon reasonable request under MTA.
Code availability
Transcriptome data were analyzed using a publicly available algorithm. https://www.bioconductor.org/packages/devel/workflows/vignettes/RNAseq123/inst/doc/limmaWorkflow.html.
References
Suzuki, T., Park, H., Hollingsworth, N. M., Sternglanz, R. & Lennarz, W. J. PNG1, a yeast gene encoding a highly conserved peptide:N-glycanase. J. Cell Biol. 149, 1039–1052 (2000).
Suzuki, T., Huang, C. & Fujihira, H. The cytoplasmic peptide: N-glycanase (NGLY1) - structure, expression and cellular functions. Gene 577, 1–7 (2016).
Suzuki, T., Park, H., Till, E. A. & Lennarz, W. J. The PUB domain: a putative protein-protein interaction domain implicated in the ubiquitin-proteasome pathway. Biochem. Biophys. Res. Commun. 287, 1083–1087 (2001).
Suzuki, T., Kwofie, M. A. & Lennarz, W. J. Ngly1, a mouse gene encoding a deglycosylating enzyme implicated in proteasomal degradation: expression, genomic organization, and chromosomal mapping. Biochem. Biophys. Res. Commun. 304, 326–332 (2003).
Lehrbach, N. J., Breen, P. C. & Ruvkun, G. Protein sequence editing of SKN-1A/Nrf1 by peptide: N-glycanase controls proteasome gene expression. Cell 177, 737–750.e715 (2019).
Tomlin, F. M. et al. Inhibition of NGLY1 inactivates the transcription factor Nrf1 and potentiates proteasome inhibitor cytotoxicity. ACS Cent. Sci. 3, 1143–1155 (2017).
Ge, H. et al. Two novel compound heterozygous mutations in NGLY1as a cause of congenital disorder of deglycosylation: a case presentation. BMC Med. Genet. 21, 135 (2020).
Enns, G. M. et al. Mutations in NGLY1 cause an inherited disorder of the endoplasmic reticulum-associated degradation pathway. Genet. Med. 16, 751–758 (2014).
Need, A. C. et al. Clinical application of exome sequencing in undiagnosed genetic conditions. J. Med. Genet. 49, 353–361 (2012).
Pandey, A. & Jafar-Nejad, H. Tracing the NGLY1 footprints: insights from drosophila. J. Biochem. 171, 153–160 (2022).
Levy, R. J., Frater, C. H., Gallentine, W. B., Phillips, J. M. & Ruzhnikov, M. R. Delineating the epilepsy phenotype of NGLY1 deficiency. J. Inherit. Metab. Dis. 45, 571–583 (2022).
Stanclift, C. R. et al. NGLY1 deficiency: estimated incidence, clinical features, and genotypic spectrum from the NGLY1 registry. Orphanet J. Rare Dis. 17, 440 (2022).
Nolan, D. K., Pastore, M. T. & McBride, K. L. Expanding the NGLY1 deficiency phenotype: case report of an atypical patient. Eur. J. Med. Genet. 65, 104558 (2022).
Asahina, M. et al. Ngly1 -/- rats develop neurodegenerative phenotypes and pathological abnormalities in their peripheral and central nervous systems. Hum. Mol. Genet. 29, 1635–1647 (2020).
Asahina, M. et al. JF1/B6F1 Ngly1(-/-) mouse as an isogenic animal model of NGLY1 deficiency. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 97, 89–102 (2021).
Asahina, M. et al. Correction to: reversibility of motor dysfunction in the rat model of NGLY1 deficiency. Mol. Brain 14, 127 (2021).
Zhu, L. et al. AAV9-NGLY1 gene replacement therapy improves phenotypic and biomarker endpoints in a rat model of NGLY1 deficiency. Mol. Ther. Methods Clin. Dev. 27, 259–271 (2022).
Ott, I. & Scott, J. C. The action of infundibulin upon the mammary secretion. Proc. Soc. Exp. Biol. Med. 8, 48–49 (1910).
Sharpey-Schafer, E. A. & Mackenzie, K. The action of animal extracts on milk secretion. Proc. Roy. Soc. London. Ser. B 84, 16–22 (1911).
Dale, H. H. On some physiological actions of ergot. J. Physiol. 34, 163–206 (1906).
Uvnäs-Moberg, K. Oxytocin may mediate the benefits of positive social interaction and emotions. Psychoneuroendocrinology 23, 819–835 (1998).
Windle, R. J., Shanks, N., Lightman, S. L. & Ingram, C. D. Central oxytocin administration reduces stress-induced corticosterone release and anxiety behavior in rats. Endocrinology 138, 2829–2834 (1997).
Amico, J., Mantella, R., Vollmer, R. & Li, X. Anxiety and stress responses in female oxytocin deficient mice. J. Neuroendocrinol. 16, 319–324 (2004).
Takahashi, T. Sensory stimulation of oxytocin release is associated with stress management and maternal care. Front. Psychol. 11, 588068 (2020).
Zinni, M., Colella, M., Batista Novais, A. R., Baud, O. & Mairesse, J. Modulating the oxytocin system during the perinatal period: a new strategy for neuroprotection of the immature brain? Front. Neurol. 9, 229 (2018).
van Campen, J. S., Jansen, F. E., de Graan, P. N., Braun, K. P. & Joels, M. Early life stress in epilepsy: a seizure precipitant and risk factor for epileptogenesis. Epilepsy Behav. 38, 160–171 (2014).
Smith, A. S., Korgan, A. C. & Young, W. S. Oxytocin delivered nasally or intraperitoneally reaches the brain and plasma of normal and oxytocin knockout mice. Pharmacol. Res. 146, 104324 (2019).
Yeomans, D. C. et al. Nasal oxytocin for the treatment of psychiatric disorders and pain: achieving meaningful brain concentrations. Transl. Psychiatry 11, 388 (2021).
Lee, M. R. et al. Labeled oxytocin administered via the intranasal route reaches the brain in rhesus macaques. Nat. Commun. 11, 2783 (2020).
Guastella, A. J. et al. Intranasal oxytocin improves emotion recognition for youth with autism spectrum disorders. Biol. Psychiatry 67, 692–694 (2010).
Verhees, M. et al. No side-effects of single intranasal oxytocin administration in middle childhood. Psychopharmacology 235, 2471–2477 (2018).
Rung, J. M. et al. Safety and tolerability of chronic intranasal oxytocin in older men: results from a randomized controlled trial. Psychopharmacology 238, 2405–2418 (2021).
Law, C. W., Chen, Y., Shi, W. & Smyth, G. K. voom: precision weights unlock linear model analysis tools for RNA-seq read counts. Genome Biol. 15, R29 (2014).
Acknowledgements
We are deeply grateful to Drs. Shinya Yamanaka and Yasushi Kajii of the T-CiRA program for supervising this project and the T-CiRA members for the fruitful discussions. We also thank Tomoyuki Kakizume of Takeda Pharmaceutical Company for their helpful suggestions in the statistical analyses and design of crossover trials, Masahiro Ide and Moe Nomura of RABICS LTD. for supporting the animal experiments, Noriyasu Sano and Koji Murakami of Axcelead Drug Discovery Partners for the preliminary analysis of seizure-like behaviors, Akira Mitsui and Katsuyuki Nakanishi of Axcelead Drug Discovery Partners for the transcriptome analyses, and Ayami Onodera of Orizuru Therapeutics for quantifying the seizure-like behaviors. Finally, we express our deep gratitude to the patients, parents, caregivers, and researchers of Grace Science Foundation.
Author information
Authors and Affiliations
Contributions
Y.M., H.Y. and T.S. conceptualized the research. Y.M. designed the experiments, collected and analyzed the data, wrote the main manuscript text, and prepared all figures and tables. M.A. and R.F. assisted with data collection. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
Y.M., H.Y. and M.A. are employees of Takeda Pharmaceutical Company, Ltd. and own stocks or stock options. The other authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Rebecca Levy and Slobodan Janković for their contribution to the peer review of this work. Primary Handling Editor: Benjamin Bessieres.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Makita, Y., Asahina, M., Fujinawa, R. et al. Intranasal oxytocin suppresses seizure-like behaviors in a mouse model of NGLY1 deficiency. Commun Biol 7, 460 (2024). https://doi.org/10.1038/s42003-024-06131-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-024-06131-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.