Abstract
Vitamin D (VitD) is a naturally occurring, fat-soluble vitamin which regulates calcium and phosphate homeostasis in the human body and is also known to have a neuroprotective role. VitD deficiency has often been associated with impaired cognition and a higher risk of dementia. In this study, we aimed to explore the relationship between levels of VitD and cognitive functioning in adult individuals. 982 cognitively healthy adults (≥ 45 years) were recruited as part of the CBR-Tata Longitudinal Study for Aging (TLSA). Addenbrooke’s cognitive examination-III (ACE-III) and Hindi mental status examination (HMSE) were used to measure cognitive functioning. 25-hydroxyvitamin D [25(OH)D] levels were measured from the collected serum sample and classified into three groups— deficient (< 20 ng/ml), insufficient (20–29 ng/ml) and normal (≥ 30 ng/ml). Statistical analysis was done using IBM SPSS software, version 28.0.1.1(15). The mean age of the participants was 61.24 ± 9 years. Among 982 participants, 572 (58%) were deficient, 224 (23%) insufficient and only 186 (19%) had normal levels of VitD. Kruskal–Wallis H test revealed a significant difference in age (p = 0.015) and education (p = 0.021) across VitD levels and the Chi-square test revealed a significant association between gender (p = 0.001) and dyslipidemia status (p = 0.045) with VitD levels. After adjusting for age, education, gender and dyslipidemia status, GLM revealed that individuals with deficient (p = 0.038) levels of VitD had lower scores in ACE-III verbal fluency as compared to normal. Additionally, we also found that 91.2% individuals who had VitD deficiency were also having dyslipidemia. It is concerning that VitD deficiency impacts lipid metabolism. Lower levels of VitD also negatively impacts verbal fluency in adult individuals. Verbal fluency involves higher order cognitive functions and this result provides us with a scope to further investigate the different domains of cognition in relation to VitD deficiency and other associated disorders.
Similar content being viewed by others
Vitamin D (VitD) is a naturally occurring fat-soluble vitamin that is endogenously synthesized in our body when the skin is exposed to sunlight1. Besides its role in calcium and phosphate metabolism, VitD has neurotropic roles and promotes nerve growth factors which have implications for brain health1,2.
The growing body of research suggests that low serum VitD levels are associated with impaired cognition with a high risk of dementia and Alzheimer’s Disease (AD)2,3. A meta-analysis by Goodwill and Szoeke, found that in both cross-sectional and longitudinal studies, lower levels of VitD were associated with cognitive decline4. Vedak et al. found that individuals with Mild Cognitive Impairment (MCI) and dementia exhibited low serum VitD levels and high homocysteine levels, indicating low VitD to be a marker of cognitive decline5. Low levels of VitD are also associated with a decline in executive functioning and episodic memory which are the two major domains affected in dementia and AD6,7.
Cognitive impairment is a major issue in the aging population and although a significant number of studies suggest the importance of VitD in cognition, the results are mixed 8,9,10. Yang et al. suggested that VitD supplementation improves the cognitive functioning of individuals suffering from MCI9. Other clinical studies found no improvement in cognition with VitD supplementation in individuals with MCI or mild to moderate AD11,12. Results from the randomized control trial study on Vitamin D, Omega-3 Fatty Acids and Cognitive Decline (VITAL-Cog), suggested VitD3 supplementation to have no significant impact on cognitive functioning in older adults13.
The endogenous production of VitD is greatly dependent on the geographical region in which an individual resides. It is assumed that individuals residing in regions away from direct exposure to sunlight will in general have a deficiency of VitD. This idea led to several studies on VitD in the Western populations but are limited in number in tropical countries like India14. In this scenario, it is important to explore the patterns of VitD levels and their effect on the cognitive functioning of individuals residing in a region that is well exposed to sunlight throughout the year.
The optimal level of VitD requirement for the human body has always been a debatable topic and the recommendations between several expert advisory bodies vary. The adequate level of VitD as per the Institute of Medicine is 20 ng/ml 15. In contrast, the US Endocrine Society and the International Osteoporosis Foundation define VitD levels as—“Deficient” (< 20 ng/ml), “Insufficient” (20–29 ng/ml) and “Sufficient” (> 30 ng/ml)16,17.
These insights led us to explore the patterns of VitD and its role in cognitive functioning of aging individuals from southern India. Our hypothesis posits that individuals with low levels of VitD will more likely exhibit impaired cognitive functioning.
Materials and methods
Study population
The subjects for this study consisted of 982 (494 males and 488 females), adult (≥ 45 years) individuals who are part of the Tata Longitudinal Study for Aging (TLSA)—a longitudinal observational study investigating cognitive trajectories of adult individuals. The cohort consist of participants from urban Bangalore, the capital city of Karnataka State in the southern part of India, wherein we recruit adults (middle aged and above) without dementia and perform detailed clinical, cognitive, imaging and genetic assessments to understand their risk and protective factors predisposing them to dementia and other neurodegenerative disorders. This is along the lines with Longitudinal Ageing Study in India (LASI) which is the first longitudinal study on aging in India, Canadian Longitudinal Study on Aging (CLSA) and Chinese Health and Retirement Longitudinal Study (CHARLS)18,19,20.
The prevalence of dementia due to modifiable risk factors (hearing loss, traumatic brain injury, hypertension, alcohol use and obesity) cumulatively come up to 15% in midlife as reported by the Lancet Commission21. Also, there are associated cognitive performance measures getting affected with perimenopausal age and less studied in our population22,23. Due to these factors, it is beneficial to look at cognitive trajectories in adults (middle-aged and above) without dementia (Clinical Dementia Rating Score < 1) and follow them up regularly to identify pre-dementia syndromes (Subjective Cognitive Decline, Motoric Cognitive Risk Syndrome, Mild Cognitive Impairment) while comprehensively evaluating for risk factors and comorbidities (diabetes, features of metabolic syndrome like hypertension, dyslipidemia etc.) and protective factors (multilingualism, musical training, physical activity) 21,24,25,26.
All our participants (n = 982) had normal cognitive functioning as indicated by Clinical Dementia Rating (CDR) score of zero. The CDR is a well-known global standard measure of dementia with high reliability and validity27. The TLSA Study protocol follows all ethical guidelines ensuring participants’ safety, informed consent, confidentiality of data and voluntary participation, and has been approved by the Institutional Ethics Committee of Centre for Brain Research, IISc. Informed consent was obtained from all the participants prior to the study.
Serum 25(OH)D measurement
Blood samples were collected for all the participants of the TLSA and analysed using chemiluminescence immunoassays on VITROS ECiQ Immunodiagnostic System using Intellicheck® Technology. VitD status was determined by levels of metabolite 25(OH)D which is the most reliable clinical indicator28 and was categorised into three groups as per the guidelines of the Endocrinology Society of America—Deficient (< 20 ng/ml), Insufficient (20–29 ng/ml) and Normal (≥ 30 ng/ml)16.
Cognitive Assessments
For this study cognitive functioning of the participants were assessed using two tools:
Addenbrooke’s cognitive examination-III (ACE-III)
It is a cognitive assessment tool with high sensitivity to assess both global and domain specific cognitive functioning. It has high efficacy in diagnosis of MCI. This tool has tests for memory, attention, orientation, language, visuo-perceptual and visuo-spatial abilities. The maximum score of 100 is distributed across the different domains 29. The ACE-III has been translated to multiple Indian languages like Hindi, Tamil, Telegu, Kannada, Malayalam, Gujarati, Urdu and Indian English due to the socio-cultural and lexical relevance and has been validated29,30,31.
Hindi mental status examination (HMSE)
It is the Indian adaptation of the Mini Mental Status Examination (MMSE) consisting of time bound subtests and is a popular measure of global cognition. The HMSE was developed as part of the Indo-US Cross-National Dementia Epidemiology Study that compared cognitive functioning between two rural populations of the US and India. A culturally and lexically relevant and sensitive version each item of the MMSE was modified according to the need of the Indian population and validated32.
Statistical Analysis
Statistical Package for Social Sciences (SPSS) version 28.0.1.1(15) was used for all statistical analysis. Kolmogorov–Smirnov test of normality was used to check the distribution of the variables in the dataset. To find the mean differences between the demographic features and VitD levels, Kruskal–Wallis H test was used. The association between the categorical variables were established using chi-square test of significance. Generalized Linear Regression Model (GLM) was performed to find the relationship between cognitive scores across VitD levels. Significant variables (age, education, gender, and dyslipidemia status) from Kruskal Wallis and chi-square tests were considered as covariates and added to the regression model. Statistical significance was considered at p < 0.05.
Results
Demographic characteristics
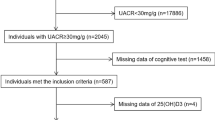
Out of the 1092 participants who had Vitamin D levels at the baseline, 110 participants had a CDR score above zero which indicated impaired cognitive functioning and were thus excluded from analysis. 982 participants who had a CDR score of zero, indicative of normal cognitive functioning, were included in this study. The mean age of the participants was 61.24 ± 9.00 years. Our cohort consisted of 494 males and 488 females. The mean VitD level of the cohort was 21.54 ± 12.60 ng/ml. There was a gross deficiency of VitD in our cohort with a total of 572 (58%) participants with VitD levels below 20 ng/ml, 224 (23%) had insufficient levels (20-29 ng/ml) and only 186 (19%) of individuals had normal levels (≥ 30 ng/ml) of VitD (Fig. 1).
Kolmogorov–Smirnov was performed to test for the distribution of variables, which were found to be not normally distributed.
The demographic details of the study population are summarized in Tables 1 and 2. Kruskal–Wallis H test revealed a significant difference in age (p = 0.015) and education (p = 0.021) across VitD levels. Chi-square test revealed a significant association with gender (p = 0.001) and dyslipidemia status (p = 0.045) and the three levels of VitD. There was no significant difference between the included participants with respect to socioeconomic status and dietary patterns.
Additionally, 91.2% of individuals who had VitD deficiency in our cohort, were also found to have dyslipidemia. Since a high prevalence of dyslipidemia was found, we performed further analysis to check for an association between different lipid parameters and VitD levels. The cut-off for dyslipidemia was considered according to the National Cholesterol Education Program—Adult Treatment Pannel III (NCEP─ATP III) criteria [triglycerides ≥ 150 mg/dl or high-density lipoprotein (HDL) < 50 mg/dl or low-density lipoprotein (LDL) > 100 mg/dl]33,34. Chi-square test revealed that there existed a significant association of triglyceride (p = 0.021), HDL (p = 0.002) as well as LDL (p = 0.047) levels with VitD deficiency. Individuals with VitD deficiency had abnormal levels of the three lipid parameters as compared to normal (Table. 3).
Neurocognitive findings
The findings of the neurocognitive measures have been summarized in Table 4. Kruskal–Wallis H test revealed a significant difference in ACE-III Attention (p = 0.016) and Fluency (p = 0.004) scores across the different levels of VitD.
GLM (unadjusted) revealed that individuals with insufficient VitD levels had significantly poorer scores in attention tasks as compared to that of normal levels of VitD (p = 0.047). Individuals with insufficient (p = 0.015) and deficient (p = 0.007) VitD levels also scored lesser in fluency domain of ACE-III. Even after adjusting for age, education, gender and dyslipidemia status, GLM revealed significantly lower verbal fluency scores in individuals with deficient (p = 0.038) levels of VitD as compared to that of normal (Table 5).
Discussion
The results of our study indicate that most of the participants had suboptimal levels of VitD (58% deficient and 23% insufficient). The mean VitD level of the cohort was also below optimal levels (21.54 ± 12.60 ng/ml). These results imply that the urban adult population in southern India had an overall inadequacy of VitD. Our results are in line with the SANSCOG (Srinivaspura Aging, Neurosenescence and COGnition) study, where Sundarakumar et al. reported the burden of VitD inadequacy in a rural population from southern India35. Similar findings have been also observed in a previous study from northern India, where high levels of VitD deficiency (91.2%) was reported in healthy adult population above 50 years of age36. The prevalence of VitD deficiency ranges between 50 and 94% as reported from several community-based studies on VitD in India37. Hospital based studies in India reported a prevalence of VitD deficiency ranging from 37 to 99%37. Previous population-based studies provide evidence that the problem of VitD inadequacy in India has existed for nearly two decades38,39. The concerning amount of low VitD levels in our cohort also opposes the traditional view that individuals from tropical countries have adequate levels of VitD due to yearlong exposure of these regions to direct sunlight. A recent study from Ecuador have also reported similar findings with high prevalence of VitD deficiency despite being a tropical country40. The possible explanation of this could be short duration of exposure to sun due to the generally hot climate of India, limited outdoor activities in the aging population, extensive clothing, air-conditioning in urban households and popularity of sunscreens41,42. India has a huge number (39%) of vegetarian population which limits the intake of dietary VitD from animal food sources41,43.
Previous studies have suggested a link between growing age and decreasing levels of VitD36,44. Adults who lack direct exposure to sunlight are also at risk of developing VitD deficiency which in turn might negatively impact other physiological mechanisms45. We observed similar findings in our cohort, where age and levels of VitD had a significant association between them. Apart from sun exposure, diet forms an integral part of maintaining VitD levels in the human body. Seafood, egg yolk, meat, mushrooms and dairy products are rich sources of dietary VitD1. Diet patterns did not vary between the individuals in our cohort; thus, no significant effect of diet was seen on VitD status. Females are prone to have VitD deficiency in general46. In our cohort we observe a significant sex difference in VitD levels, although more men are found to be deficient than females. Similar findings have been reported by a study from Latin America where the prevalence of VitD deficiency was more in males than females47.
The existing body of literature suggests an association of VitD deficiency with an increased risk for several diseases like hypertension, diabetes and dyslipidemia. Adequate levels of VitD often act as a protective factor against development of the above-mentioned diseases which are independent risk factors for cognitive impairment24,48,49. The individuals in our cohort did not differ significantly with respect to hypertension and diabetes, although dyslipidemia was found in a large number of individuals with low levels of VitD. Similar findings were reported by a study in Saudi Arabia where an association was found between VitD deficiency and dyslipidemia50. Individuals with VitD deficiency in our cohort had abnormal levels of triglycerides, HDL as well as LDL when compared to individuals with normal VitD levels. Our findings resonate with previous studies which reported significant association of VitD deficiency with high LDL and triglyceride levels51,52. A meta-analysis by Radkhah et al. found VitD supplementation to help improve lipid profiles, thus strengthening the importance of VitD in maintenance of serum lipid levels53. VitD is known to aid lipid metabolism by increasing calcium levels in the blood48. Liu et al. suggested that VitD deficiency had a negative impact on cholesterol metabolism which might further give rise to several cardiovascular conditions which are independent risk factors of cognitive decline54. Therefore, low levels of VitD are often associated with poor lipid metabolism. Previous studies have suggested dyslipidemia in mid-life to be a risk factor in the development of cognitive impairment and dementia in later life55,56. Therefore, it becomes imperative to check for dyslipidemia when an individual is VitD deficient.
Low VitD levels are associated with impaired cognitive functioning in several domains, but whether these associations tantamount to causation is still debatable. This makes it worth exploring the role of VitD on cognitive functioning, especially in the adult population as literature suggests VitD levels to decrease with age44. According to the results of the present study, low VitD levels did not affect global cognition as evidenced from non-significant HMSE and ACE-III total scores, but individuals with insufficient levels of VitD showed decreased level of attention. Several previous studies have reported that VitD did not affect global cognition, neither improved cognitive functioning on supplementation13,40,42. Deficits in attention is a part of aging and it has been found that attention training interventions improved cognitive functioning in individuals with MCI or mild to moderate dementia57. VitD deficiency is a well-known biomarker of Attention-Deficit/ Hyperactivity Disorder (ADHD) in children58. In older adults, there is evidence of an association between low VitD levels and the decreased ability to focus attention59. Our results are also in line with a study in India by Vedak et al., where a significant association was found between low serum VitD levels and the domain of attention5. Therefore, more assessments and further research in this domain is desirable to discern the existence of a cause-effect relationship.
The results of our study also revealed an association between suboptimal levels of VitD and verbal fluency. Verbal fluency is closely associated with semantic processing, language, working memory and executive functioning60,61. Previous studies have suggested a correlation between low VitD levels and decline in executive functioning and working memory 62,63. So, it may be postulated that the association between low levels of VitD and verbal fluency has an indirect relationship with aspects of executive functioning and memory. Though the literature on direct association between verbal fluency and VitD is scarce, a study in Canada reported similar findings where supratherapeutic levels of VitD supplementation significantly improved scores on verbal fluency tasks7. Another study by Nerhus et al. suggested VitD deficiency to be associated with poorer verbal fluency and processing speed64.
The primary objective of this study was to understand the patterns of VitD in adult population (middle aged and above) and its impact on cognition. The significant burden of VitD deficiency in India posits the need for strategical community level measures like food fortification and vitamin supplementation. VitD fortification of milk has been proved to be safe and effective in children as reported by prospective trials65,66. Besides its impact on cognition (verbal fluency and attention), VitD inadequacy has a significant negative impact on lipid metabolism giving rise to conditions like dyslipidemia which might act as a precursor to several other cardiovascular conditions. The findings further confirm the role of VitD in cognition and health and future research is imperative to ascertain a causational relationship.
Our study is not without limitations. The cross-sectional study design might not be suitable for generalizability of the results. We did not have any means of quantifying endogenous production of VitD in our participants. Also, the study sample is restricted to a particular region; a pan India study sample would provide us with a better understanding of the issue of VitD and cognition. The strength of our study in is its large sample size which provides us with some understanding of VitD patterns and the cognitive implications of the same. These findings pave way for public health measures with a focus on primary prevention.
Conclusion
This study suggests there exists a significant prevalence of VitD deficiency in urban Indian population and lower levels of serum VitD levels are associated with high prevalence of dyslipidemia. VitD deficiency also negatively impacts verbal fluency and attention. Both verbal fluency and attention involve higher cognitive functions and this result provides us with a scope to further investigate the said domains in relation to VitD. The need for food fortification as well as adequate exposure to sunlight may be necessary in city dwelling adult population to mitigate the cognitive deficits as well as other health issues associated with Vitamin D deficiency.
Data availability
The datasets generated and/or analysed during the current study are not publicly available as the study is a longitudinal cohort study and is currently ongoing, the data is still being collected and curated and being monitored by the Institutional Ethics Committee (IEC) and Technical Advisory Committee (TAC). Therefore, it is not made public at this point of time. Data request can be directed to the corresponding author Dr. Thomas Gregor Issac who is the PI of TLSA study and data will be shared if approved by the IEC and TAC.
References
Vitamin D - Health Professional Fact Sheet [Internet]. [cited 2023 Dec 27];Available from: https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/
Sultan, S. et al. Low vitamin D and its association with cognitive impairment and dementia. J. Aging Res. 2020, 1–10 (2020).
Wang, W., Li, Y. & Meng, X. Vitamin D and neurodegenerative diseases. Heliyon 9(1), e12877 (2023).
Goodwill, A. M. & Szoeke, C. A systematic review and meta-analysis of the effect of low vitamin D on cognition. J. Am. Geriatr. Soc. 65(10), 2161–2168. https://doi.org/10.1111/jgs.15012 (2017).
Vedak, T. K. et al. Vitamin D as a marker of cognitive decline in elderly Indian population. Ann. Indian Acad. Neurol. 18(3), 314–319 (2015).
Miller, J. W. et al. Vitamin D status and rates of cognitive decline in a multiethnic cohort of older adults. JAMA Neurol. 72(11), 1295–1303 (2015).
Pettersen, J. A. Vitamin D and executive functioning: Are higher levels better?. J. Clin. Exp. Neuropsychol. 38(4), 467–477. https://doi.org/10.1080/13803395.2015.1125452 (2016).
Aghajafari, F., Pond, D., Catzikiris, N. & Cameron, I. Quality assessment of systematic reviews of vitamin D, cognition and dementia. BJPsych Open 4(4), 238–249 (2018).
Yang, T. et al. Vitamin D supplementation improves cognitive function through reducing oxidative stress regulated by telomere length in older adults with mild cognitive impairment: A 12-month randomized controlled trial. J. Alzheimer’s Dis. 78(4), 1509–1518 (2020).
Duchaine, C. S. et al. Vitamin D status, cognitive decline and incident dementia: The Canadian Study of Health and Aging. Canad. J. Public Health 111(3), 312–321. https://doi.org/10.17269/s41997-019-00290-5 (2020).
Stein, M. S., Scherer, S. C., Ladd, K. S. & Harrison, L. C. A randomized controlled trial of high-dose vitamin D2 followed by intranasal insulin in alzheimer’s disease. J. Alzheimer’s Dis. 26(3), 477–484 (2011).
Rossom, R. C. et al. Calcium and vitamin D supplementation and cognitive impairment in the women’s health initiative. J. Am. Geriatr. Soc. 60(12), 2197–2205. https://doi.org/10.1111/jgs.12032 (2012).
Kang, J. H. et al. Effect of vitamin D on cognitive decline: Results from two ancillary studies of the VITAL randomized trial. Sci. Rep. 11(1), 1–14 (2021).
Wahl, D. A. et al. A global representation of vitamin D status in healthy populations. Archiv. Osteoporos. 7(1), 155–172. https://doi.org/10.1007/s11657-012-0093-0 (2012).
Ross, A. C. et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the institute of medicine: What clinicians need to know. J. Clin. Endocrinol. 96(1), 53–58. https://doi.org/10.1210/jc.2010-2704 (2011).
Holick, M. F. et al. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 96(7), 1911–1930. https://doi.org/10.1210/jc.2011-0385 (2011).
Dawson-Hughes, B. et al. IOF position statement: Vitamin D recommendations for older adults. Osteoporosis Int. 21(7), 1151–1154. https://doi.org/10.1007/s00198-010-1285-3 (2010).
LASI—Longitudinal Aging Study of India [Internet]. [cited 2024 Apr 29];Available from: https://lasi-india.org/
Raina, P. et al. Erratum: Cohort profile: The Canadian Longitudinal Study on Aging (CLSA). Int. J. Epidemiol. 48(6), 2066. https://doi.org/10.1093/ije/dyz173 (2019).
Zhao, Y., Hu, Y., Smith, J. P., Strauss, J. & Yang, G. Cohort Profile: The China Health and Retirement Longitudinal Study (CHARLS). Int. J. Epidemiol. 43(1), 61–68. https://doi.org/10.1093/ije/dys203 (2014).
Livingston, G. et al. Dementia prevention, intervention, and care: 2020 Report of the Lancet Commission. Lancet 396(10248), 413–446 (2020).
Metcalf, C. A., Duffy, K. A., Page, C. E. & Novick, A. M. Cognitive problems in perimenopause: A review of recent evidence. Curr. Psychiatry Rep. 25(10), 501–511. https://doi.org/10.1007/s11920-023-01447-3 (2023).
Sathiyaseelan, A., Chaudhury, S. & Patangia, B. Cognitive and biological challenges of menopausal women in India. Life Res. 5(3), 23 (2022).
Pál, É., Ungvári, Z., Benyó, Z. & Várbíró, S. Role of vitamin D deficiency in the pathogenesis of cardiovascular and cerebrovascular diseases. Nutrients 15(2), 334 (2023).
Menon, A. J. et al. Association between multilingualism and cognitive performance among older adults in rural southern India. J. Neurosci. Rural Pract. 15(1), 81 (2024).
Ghosh A, Singh S, S. M, Jagtap T, Issac TG, Ghosh A, et al. Music and the aging brain—Exploring the role of long-term Carnatic music training on cognition and gray matter volumes. J. Neurosci. Rural Pract. 1–7 (2024).
Morris, J. C. Clinical dementia rating: A reliable and valid diagnostic and staging measure for dementia of the Alzheimer type. Int. Psychogeriatr. 9(S1), 173–176 (1997).
Lai, J. K. C., Lucas, R. M., Clements, M. S., Harrison, S. L. & Banks, E. Assessing vitamin D status: Pitfalls for the unwary. Mol. Nutr. Food Res. 54(8), 1062–1071. https://doi.org/10.1002/mnfr.200900468 (2010).
JCDR—Cognition disorders, Cross cultural adaptation, Cross cultural comparison, Diagnosis, Language, Screening. Available from: https://jcdr.net/article_fulltext.asp?issn=0973-709x&year=2018&volume=12&issue=6&page=VC11&issn=0973-709x&id=11696
Bajpai, S. et al. Hindi version of Addenbrooke’s cognitive examination III: Distinguishing cognitive impairment among older indians at the lower cut-offs. Clin. Interv. Aging 15, 329–339 (2020).
Mekala, S. et al. Dementia diagnosis in seven languages: The Addenbrooke’s cognitive examination-III in India. Archiv. Clin. Neuropsychol. 35(5), 528–538. https://doi.org/10.1093/arclin/acaa013 (2020).
Ganguli, M. et al. A hindi version of the MMSE: The development of a cognitive screening instrument for a largely illiterate rural elderly population in India. Int. J. Geriatr. Psychiatry 10(5), 367–377. https://doi.org/10.1002/gps.930100505 (1995).
Sundarakumar, J. S., Stezin, A., Menesgere, A. L. & Ravindranath, V. Rural-urban and gender differences in metabolic syndrome in the aging population from southern India: Two parallel, prospective cohort studies. EClinicalMedicine 47, 101395. https://doi.org/10.1016/j.eclinm.2022.101395 (2022).
NCEP ATP-III Cholesterol Guidelines -- Cholesterol 2.0 - SCYMED [Internet]. [cited 2024 Apr 19];Available from: https://www.scymed.com/en/smnxdj/edzr/edzr9610.htm
Sundarakumar, J. S., Shahul Hameed, S. K. & Ravindranath, V. Burden of vitamin D, vitamin B12 and folic acid deficiencies in an aging, rural Indian community. Front Public Health 9, 707036 (2021).
Vitamin D status in healthy Indians aged 50 years and above - PubMed [Internet]. [cited 2023 Dec 28];Available from: https://pubmed.ncbi.nlm.nih.gov/22616336/
Vitamin D deficiency in India : Journal of Family Medicine and Primary Care [Internet]. [cited 2024 Jan 1];Available from: https://journals.lww.com/jfmpc/fulltext/2018/07020/vitamin_d_deficiency_in_india.9.aspx
High prevalence of low dietary calcium and low vitamin D status in healthy south Indians—PubMed [Internet]. [cited 2023 Dec 28];Available from: https://pubmed.ncbi.nlm.nih.gov/15563441/
Vitamin D status in Andhra Pradesh: A population based study—PubMed [Internet]. [cited 2023 Dec 28];Available from: https://pubmed.ncbi.nlm.nih.gov/18497434/
Vinueza Veloz, A. F. et al. Cognitive function and vitamin B12 and D among community-dwelling elders: A cross-sectional study. Clin. Nutr. ESPEN 50, 270–276 (2022).
Rao, P. V. P., Siddhartha, P. V. & Yeshwante, P. Vitamin D deficiency, the urban epidemic: A cross-sectional study from Bhopal, India. Int. J. Commun. Med. Public Health 7(11), 4541 (2020).
Menesgere, A., Giridhar, V., Bota, R. & Ravindranath, V. Role of Vitamin D on cognitive performance among healthy volunteers of SANSCOG cohort. Clin. Nutr. Open Sci. 44, 1–8 (2022).
In India, 81% limit meat in diet and 39% say they are vegetarian | Pew Research Center [Internet]. [cited 2023 Dec 28];Available from: https://www.pewresearch.org/short-reads/2021/07/08/eight-in-ten-indians-limit-meat-in-their-diets-and-four-in-ten-consider-themselves-vegetarian/
Gallagher, J. C. Vitamin D and aging. Endocrinol. Metab. Clin. North Am. 42(2), 319–332 (2013).
Patient education: Vitamin D deficiency (Beyond the Basics)—UpToDate [Internet]. [cited 2024 Apr 17];Available from: https://www.uptodate.com/contents/vitamin-d-deficiency-beyond-the-basics
Wierzbicka, A. & Oczkowicz, M. Sex differences in vitamin D metabolism, serum levels and action. Br. J. Nutr. 128(11), 2115–2130 (2022).
Vallejo, M. S. et al. Gender differences in the prevalence of vitamin D deficiency in a southern Latin American country: A pilot study. Climacteric 23(4), 410–416. https://doi.org/10.1080/13697137.2020.1752171 (2020).
Yang, K. et al. Vitamin D status and correlation with glucose and lipid metabolism in Gansu Province, China. Diabetes Metab. Syndrome Obes. 13, 1555–1563 (2020).
Latic, N. & Erben, R. G. Vitamin D and cardiovascular disease, with emphasis on hypertension, atherosclerosis, and heart failure. Int. J. Mol. Sci. 21(18), 6483 (2020).
Alquaiz, A. J. M., Kazi, A., Youssef, R. M., Alshehri, N. & Alduraywish, S. A. Association between standardized vitamin 25(OH)D and dyslipidemia: A community-based study in Riyadh. Saudi Arabia. Environ Health Prev Med 25(1), 1–9. https://doi.org/10.1186/s12199-019-0841-5 (2020).
Han, Y. Y., Hsu, S. H. J. & Su, T. C. Association between vitamin D deficiency and high serum levels of small dense LDL in middle-aged adults. Biomedicines 9(5), 464 (2021).
Kim, M. R. & Jeong, S. J. Relationship between vitamin D level and lipid profile in non-obese children. Metabolites 9(7), 125 (2019).
Radkhah, N., Zarezadeh, M., Jamilian, P. & Ostadrahimi, A. The effect of vitamin D supplementation on lipid profiles: An umbrella review of meta-analyses. Adv. Nutr. 14(6), 1479–1498 (2023).
Liu, W. et al. Vitamin D deficiency is associated with disrupted cholesterol homeostasis in patients with mild cognitive impairment. J. Nutr. 151(12), 3865–3873 (2021).
Wee, J. et al. The relationship between midlife dyslipidemia and lifetime incidence of dementia: A systematic review and meta-analysis of cohort studies. Alzheimer’s & Dement.: Diagnosis Assess. Dis. Monitor. 15(1), e12395. https://doi.org/10.1002/dad2.12395 (2023).
Yu, Y. et al. Correlation between serum lipid profiles and cognitive impairment in old age: A cross-sectional study. Gen Psychiatr 36(2), e101009 (2023).
Sung, C. M. et al. The effect of cognitive training on domains of attention in older adults with mild cognitive impairment and mild dementia: A meta-analysis of randomised controlled trials. J. Glob Health https://doi.org/10.7189/jogh.13.04078 (2023).
Hemamy, M. et al. The effect of vitamin D and magnesium supplementation on the mental health status of attention-deficit hyperactive children: A randomized controlled trial. BMC Pediatr 21(1), 1–8. https://doi.org/10.1186/s12887-021-02631-1 (2021).
Buell, J. S. et al. Vitamin D is associated with cognitive function in elders receiving home health services. J. Gerontol.: Series A 64A(8), 888–895. https://doi.org/10.1093/gerona/glp032 (2009).
Kavé, G. & Sapir-Yogev, S. Associations between memory and verbal fluency tasks. J. Commun. Disord. 83, 105968 (2020).
Amunts, J. et al. Comprehensive verbal fluency features predict executive function performance. Sci. Rep. 11(1), 1–14 (2021).
Annweiler, C. et al. Meta-analysis of memory and executive dysfunctions in relation to vitamin D. J. Alzheimer’s Dis. 37(1), 147–171. https://doi.org/10.3233/JAD-130452 (2013).
Shea, M. K. et al. Brain vitamin D forms, cognitive decline, and neuropathology in community-dwelling older adults. Alzheimer’s Dement. 19(6), 2389–2396. https://doi.org/10.1002/alz.12836 (2023).
Nerhus, M. et al. Vitamin D deficiency associated with cognitive functioning in psychotic disorders. J. Clin. Psychiatry 78(7), 22252 (2017).
Garg, M. K. et al. Efficacy of vitamin D loading doses on serum 25-hydroxy vitamin D levels in school going adolescents: An open label non-randomized prospective trial. J. Pediatric Endocrinol. Metab. 26(5–6), 515–523. https://doi.org/10.1515/jpem-2012-0390/html (2013).
Khadgawat, R. et al. Impact of vitamin D fortified milk supplementation on vitamin D status of healthy school children aged 10–14 years. Osteoporosis Int. 24(8), 2335–2343. https://doi.org/10.1007/s00198-013-2306-9 (2013).
Acknowledgements
We would like to acknowledge all the members of the CBR-TLSA for helping us with the collection of data. We would like to thank the participants for their participation and kind cooperation. We also extend our gratitude to Tata Trusts for their financial support and the Director, Centre for Brain Research for his constant support and encouragement.
Author information
Authors and Affiliations
Contributions
T.G.I., L.D. and A.G. were involved in designing the study. T.G.I., L.D., A.S.S. and A.G., conceptualised the manuscript, defined intellectual content and contributed to the literature search and acquiring data. L.D. helped with the biochemical analysis to acquire Vitamin D data. M.S. and A.G. performed primary statistical analysis. L.D., A.S.S. and M.S. were involved in secondary statistical analysis. A.G. and M.S. wrote the manuscript text and prepared the tables and figures. All the authors edited and reviewed the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ghosh, A., S, M., Sunny, A.S. et al. Prevalence and patterns of vitamin D deficiency and its role in cognitive functioning in a cohort from South India. Sci Rep 14, 11215 (2024). https://doi.org/10.1038/s41598-024-62010-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-62010-5
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.