Abstract
Anti-thymocyte globulin (ATG) is currently the most widely prescribed induction regimen for preventing acute rejection after solid organ transplantation. However, the optimal dose of ATG induction regimen in Asian kidney recipients is unclear. Using the Korean Organ Transplantation Registry, we performed a retrospective cohort study of 4579 adult patients who received renal transplantation in South Korea and divided them into three groups according to the induction regimen: basiliximab group (n = 3655), low-dose ATG group (≤ 4.5 mg/kg; n = 467), and high-dose ATG group (> 4.5 mg/kg; n = 457). We applied the Toolkit for Weighting and Analysis of Nonequivalent Groups (TWANG) package to generate high-quality propensity score weights for intergroup comparisons. During four-year follow-ups, the high-dose ATG group had the highest biopsy-proven acute rejection rate (basiliximab 20.8% vs. low-dose ATG 22.4% vs. high-dose ATG 25.6%; P < 0.001). However, the rates of overall graft failure (4.0% vs. 5.0% vs. 2.6%; P < 0.001) and mortality (1.7% vs. 2.8% vs. 1.0%; P < 0.001) were the lowest in the high-dose ATG group. Our results show that high-dose ATG induction (> 4.5 mg/kg) was superior to basiliximab and low-dose ATG induction in terms of graft and patient survival in Asian patients undergoing kidney transplant.
Similar content being viewed by others
Introduction
Anti-thymocyte globulin (ATG) is currently the most widely prescribed induction regimen for solid organ transplantation globally. Along with T-cell depletion, ATG is known to be involved in the modulation of adhesion and cell surface molecules that regulate leukocyte-endothelial interaction and T-cell function1,2,3,4. ATG was shown to be superior to interleukin 2 antagonist for the prevention of acute rejection after kidney transplantation (KT)5,6, although post-transplant complications such as infection and malignancy are more common7,8,9.
According to previous reports, the proper doses of ATG as an induction regimen in kidney transplantation ranged from 1.5 to 7.5 mg/kg1,10,11,12,13. Over a few decades, there has been a trend toward lowering the ATG dose considering the balance between efficacy and safety14. A retrospective study reported that the incidence of early acute rejection was higher if the total ATG dose was less than 6 mg/kg15. However, favorable outcomes with lower doses have also been reported. Gurk-Turner et al. showed that total ATG doses ≤ 7.5 mg/kg are safe and effective even in high-risk kidney transplant recipients compared with higher doses12. Klem et al. suggested that induction with a total ATG dose of 4.5 or 6.0 mg/kg (1.5 mg/kg/dose) was enough to prevent acute rejection even in recipients with high risk10.
Recently, the efficacy and immunophenotyping of ATG doses lower than 4.5 mg/kg have also been evaluated. A randomized pilot study suggested that low-dose ATG (2.25 mg/kg) was efficacious in preventing acute rejection and depleting T cells with lower infectious complications3. According to Kho et al., T cells in the low-dose (3.0 mg/kg) and ultra-low-dose (1.5 mg/kg) ATG groups returned to control values earlier than the standard dose (6.0 mg/kg) group, although there was no significant differences in clinical outcomes11. However, the existing studies on different dosing strategies for ATG are limited due to the small number of patients, predominance of Caucasians, and the exclusion of sensitized patients. Multicenter studies are needed to determine the optimal dose of anti-thymocyte globulin for improved outcomes after KT in different ethnicities.
The aim of this study was to compare the clinical outcomes of KT in an Asian population according to the induction regimen (basiliximab, low-dose, and high-dose ATG) using the nationwide Korean Organ Transplantation Registry (KOTRY) database.
Results
Baseline characteristics
The clinical characteristics of the recipients and donors are shown in Table 1 with adjustment by propensity score weighting (TWANG package, Fig. S1). Recipients in the basiliximab group were significantly younger than those in the low-dose ATG and the high-dose ATG groups (48.9 ± 11.6 vs. 51.4 ± 10.7 vs. 51.1 ± 10.8 years, P < 0.001). The high-dose ATG group had the highest proportion of female recipients (37.3% vs. 44.5% vs. 56.5%, P < 0.001) and the lowest body mass index (23.2 ± 3.6 vs. 23.5 ± 3.4 vs. 22.6 ± 3.1 kg/m2, P < 0.001). The basiliximab group had the highest proportion of those who underwent preemptive KT (16.4% vs. 9.9% vs. 10.7%, P < 0.001) and ABO-incompatible KT (17.8% vs. 15.2% vs. 12.5%, P = 0.009) and the lowest proportion of those with a history of previous transplant (5.4% vs. 14.4% vs. 16.2%, P < 0.001). The basiliximab group also had the lowest numbers of ABDR mismatch (3.91 ± 1.17 vs. 4.18 ± 1.16 vs. 4.17 ± 1.13, P < 0.001) and DR mismatch (1.29 ± 0.50 vs. 1.40 ± 0.52 vs. 1.39 ± 0.51, P < 0.001). Compared with the ATG groups, the basiliximab group had lower proportions of deceased donors (32.6% vs. 57.6% vs. 54.1%, P < 0.001) and donors with hypertension (14.1% vs. 20.1% vs. 20.1%, P < 0.001) and a higher proportion of current smokers (67.1% vs. 57.8% vs. 59.1%, P < 0.001). Lastly, the basiliximab group had the lowest level of serum creatinine before donation (0.95 ± 0.70 vs. 1.41 ± 1.32 vs. 1.46 ± 1.39 mg/dL, P < 0.001).
Clinical outcomes
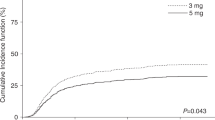
A total of 1947 kidney transplant recipients had kidney allograft biopsies due to an elevated serum creatinine (n = 773), aggravated proteinuria (n = 40), scheduled protocol (n = 1081), and other reasons (n = 53). Cumulative incidences of biopsy-proven acute rejection (BPAR) were significantly higher in the high-dose ATG group than in the other groups during a four-year follow-up period, although the increase nearly plateaued two years after transplantation (Table 2) (Fig. 1). The rates of acute antibody-mediated rejection (ABMR) and chronic ABMR were the highest in the high-dose ATG group. In contrast, acute T-cell mediated rejection (TCMR) within 1 year post-transplantation was the highest in the low-dose ATG (Table 3).
The rate of delayed graft function was the highest in the high-dose ATG group (3.40% vs. 2.95% vs. 4.28%, P = 0.009) (Table 2). In contrast, the cumulative rate of overall graft failure was the lowest in the high-dose ATG group (Fig. 2a). Also, the rate of death-censored graft failure (DCGF) was the lowest in the high-dose ATG group, albeit without statistical significance (P = 0.08; Fig. 2b). The cumulative rate of mortality was the lowest in the high-dose ATG group and the highest in the low-dose ATG group (Fig. 3). Since significant differences in the mortality among groups is identified, the graft survival was re-analyzed using Fine-Gray Subdistribution Hazard Ratio (HR) analysis (Fig. 4). The results of the Fine-Gray Subdistribution HR analysis for DCGF and mortality show a significant increase in mortality risk for the low-dose ATG group compared to both the Basiliximab and high-dose ATG groups, with the highest risk observed when comparing low-dose ATG to high-dose ATG. Additionally, the risk of DCGF was significantly lower in the high-dose ATG group compared to Basiliximab, while there was no significant difference between low-dose ATG and Basiliximab. These findings suggest that patients receiving low-dose ATG may have a higher risk of mortality, and that high-dose ATG may provide better protection against DCGF compared to Basiliximab.
Cytomegalovirus infection was the most common in the high-dose ATG group (Fig. 5a), and hematologic abnormalities including neutropenia and thrombocytopenia were higher in the ATG groups than in the basiliximab group (Fig. 5b). Although there was no significant difference in the incidence of post-transplant malignancy until two years after transplantation, the incidence in the low-dose ATG group was higher thereafter (Fig. 5c).
Furthermore, we evaluated clinical outcomes based on induction regimens used in patients considered at high immunological risk. High immunological risk patients were defined as those who underwent desensitization before kidney transplantation due to the presence of pre-transplant donor-specific antibodies, ABO-incompatible KT, or human leukocyte antigen (HLA)-incompatible KT. Out of the 4579 enrolled recipients, 1072 were classified as high-risk immunologically. Among them, 779 recipients were administered basiliximab, while 133 and 160 recipients received low- and high-dose ATG, respectively. Over a four-year follow-up period, the cumulative incidences of BPAR were significantly higher in the low- and high-dose ATG groups compared to the basiliximab group, but no significant difference was observed between the low- and high-dose ATG groups (Table 4). Conversely, the cumulative rates of overall graft failure and mortality were significantly lower in the high-dose ATG group compared to the low-dose ATG group, while no significant difference was found in the cumulative rate of DCGF between the groups.
Discussion
Using the KOTRY database, we found that the mean, median, 25th percentile, and 75th percentile of the ATG dose in Korean KT recipients were 4.85 mg/kg, 4.50 mg/kg, 4.26 mg/kg, and 5.45 mg/kg, respectively. We also found that high-dose ATG induction (> 4.5 mg/kg) showed superior outcomes in terms of graft and patient survival compared with basiliximab and low-dose ATG (< 4.5 mg/kg), although BPAR was the most common in the high-dose ATG group. To our knowledge, this is the first study to examine the currently used dose of ATG as an induction regimen and to compare the clinical outcomes according to the doses of ATG using a national kidney transplant database in an Asian population.
Previous randomized controlled trials compared clinical outcomes according to the doses of ATG as an induction regimen, which ranged from 5 to 10 mg/kg19,20,21,22,23,24,25. On the other hand, recent studies examined the efficacies of lower doses of ATG on the depletion of immune cells as well as clinical outcomes. Kho et al. compared the counts of peripheral immune cells after kidney transplantation among 1.5, 3.0, and 6.0 mg/kg ATG induction groups and showed that only the 6.0 mg/kg ATG induction group had a significantly lower T cell count compared with recipients without an induction regimen11. According to one randomized pilot study, 2.25 mg/kg ATG induction with early steroid tapering was effective not only in preventing acute rejection but also in depleting T cells3.
A recent study by Mehta et al. showed that patients in the regular dose ATG (≥ 5 mg/kg) group had more favorable outcomes in terms of early (within six months posttransplant) rejection compared with the low-dose (< 5 mg/kg) group26. On the other hand, another study by Linhares et al. reported that the 3 mg/kg and 5 mg/kg ATG induction groups did not show significant differences in the incidence of efficacy failure (first BPAR, graft failure, or mortality) and in safety outcomes at twelve months posttransplant27; however, the study by Linhares et al. was limited due to its single-center, retrospective design, small number of patients, short follow-up duration, and the fact that only those who underwent repeat kidney transplantation were included.
Even though the incidence of BPAR was the highest in the high-dose ATG group in our study, recipients in this group showed superior outcomes in terms of graft and patient survival. Considering that the high-dose ATG group had the highest incidence of BPAR during the first 6 months, it is likely that the high-dose ATG group had more patients with higher immunologic risk although we tried to create high-quality propensity score weights with the TWANG package. Although it is known that ATG is effective to prevent and treat TCMR, there are several reports that ATG is also effective against ABMR by suppression of natural killer (NK) and follicular helper T cells. Several studies using microarray transcriptomic analysis showed that NK and follicular helper T cells are actively involved in the pathophysiology of ABMR and graft failure after kidney transplantation28,29,30,31,32. Recently, we reported the results of a prospective, randomized, pilot study showing that NK cells in recipients with ATG 6.0 mg/kg induction remained suppressed whereas NK cells in those with ATG 6.0 mg/kg induction had recovered to the pre-transplant levels33. Therefore, it is considered that those with high-dose ATG induction had superior outcomes in terms of graft failure and mortality in spite of higher incidences of ABMR.
Interestingly, not only the rate of developing malignancy but also the rates of graft failure and mortality were the highest in the low-dose ATG group. Higher incidences of cytomegalovirus infection, neutropenia, or thrombocytopenia in the high-dose ATG group did not have a significant impact on graft failure and mortality. The unique observation of mortality preceding death-censored graft failure in the low-dose ATG group highlights the need for further investigation to understand the underlying factors contributing to this phenomenon.
This study has several limitations. We could not assess the presence of donor-specific antibodies before transplantation, which is necessary for estimating the immunologic risk of each patient. In addition, data on calcineurin inhibitor trough levels were not collected during the follow-up period, and Banff scores for each BPAR were not recorded in the registry. However, we could investigate the incidences of TCMR and ABMR separately. Due to the limited follow-up duration in this nationwide registry, we could not evaluate the long-term clinical outcomes according to the induction regimen. Lastly, the protocols for desensitization and treatment of TCMR and ABMR could not be determined at each center, which could affect graft survival and patient survival after an episode of BPAR.
Nevertheless, it is noteworthy that this study investigated the contemporary practice of KT induction regimens in an Asian population and compared the clinical outcomes according to the induction regimen and ATG dosage using data from a nationwide kidney transplant cohort. Notably, low-dose ATG induction had the worst outcomes even than basiliximab induction in terms of graft survival and patient survival.
In conclusion, our current study in an Asian nationwide cohort showed that high-dose ATG induction (> 4.5 mg/kg) had superior outcomes to basiliximab and low-dose ATG induction in terms of graft and patient survival.
Materials and methods
Study population
KOTRY is a nationwide transplantation cohort for five solid organs (kidney, liver, heart, lung, and pancreas) consisting of 59 participating centers in South Korea. Longitudinal data collection was initiated in 2014. Details about the design and methods were previously reported16. Briefly, KOTRY collects data on demographics, comorbidities, laboratory measurements, concentration and dosage of immunosuppressants including induction and maintenance regimen, allograft rejection, allograft failure, infectious events, cardiovascular complications, malignancies, recipient mortality, donor comorbidity, and outcomes of live donors. The KOTRY dataset includes a total of 5652 kidney transplant recipients from 30 centers between 2015 and 2019, from which 931 were excluded due to follow-up loss except for death and 142 were excluded because both ATG and basiliximab were administered as induction regimen (Fig. 6). Therefore, a total of 4579 recipients were included in this study. Among them, ATG was administered to 924 recipients whereas basiliximab was used for 3655 recipients. The mean, median, 25th percentile, and 75th percentile of the ATG dose were 4.85 mg/kg, 4.50 mg/kg, 4.26 mg/kg, and 5.45 mg/kg, respectively (Fig. 7). Of the 924 recipients with ATG induction, 467 were classified as low-dose ATG group and 457 as high-dose ATG group based on the median value of ATG dose (4.50 mg/kg). Written informed consent was achieved from all recipients and live kidney donors before KT and donor nephrectomy, respectively. This study was approved by the institutional review board of Asan Medical Center (approval number: 2021–0032) and was performed according to the Declaration of Helsinki and the Declaration of Istanbul. No organs/tissues were procured from prisoners.
Clinical outcomes
The primary outcomes were the rates of BPAR, DCGF, overall graft failure, and mortality. BPAR was diagnosed according to the Banff 2015 criteria17. Secondary outcomes were the level of serum creatinine and the incidence of cytomegalovirus infection, bacterial infection, neutropenia or thrombocytopenia, or malignancy. CMV infection was defined as end-organ infection by CMV or persistent detection of CMV viremia (> 3.0 log copies/mL) in polymerase chain reaction tests. Neutropenia was defined as white blood cell count less than 1,500 /uL whereas thrombocytopenia was considered if the platelet count was less than 150,000 /uL. The primary and secondary outcomes were assessed at six months after transplantation and at one-year intervals thereafter.
Statistical analysis
Continuous variables are presented as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was used to compare continuous variables among the three groups. Primary and secondary outcomes at each period were compared between groups using ANOVA. Categorical variables are summarized as absolute and relative frequencies, and differences among categorical values were analyzed using the Chi-squared test. In order to create high-quality propensity score weights, the Toolkit for Weighting and Analysis of Nonequivalent Groups (TWANG) package was applied to estimate the probability that a recipient developed primary and secondary outcomes for calculating weights and generalized boosted regression18. A total of fifteen recipient characteristics (age, sex, diabetes, previous transplant, ABO-incompatible KT, ABDR mismatch, DR mismatch, and calcineurin inhibitor) and donor characteristics (age, sex, body mass index, relation to the recipient, current smoker, serum creatinine, and cold ischemic time) were included in the weighting algorithm. After the matched samples were identified, generalized linear mixed models were estimated by including all recipient and donor characteristics to adjust for any possible differences among the groups. P values < 0.05 were considered statistically significant. Statistical analyses were performed using R software version 3.1.2 (R Foundation for Statistical Computing, Vienna, Austria).
Data availability
Raw data were generated at Korea Centers for Disease Control and Prevention Agency. Derived data supporting the findings of this study are available from the corresponding author on request.
Abbreviations
- ABMR:
-
Antibody-mediated rejection
- ATG:
-
Anti-thymocyte globulin
- BPAR:
-
Biopsy-proven acute rejection
- DCGF:
-
Death-censored graft failure
- KOTRY:
-
Korean organ transplantation registry
- KT:
-
Kidney transplantation
- NK:
-
Natural killer
- PRA:
-
Panel reactive antibody
- TCMR:
-
T-cell mediated rejection
- TWANG:
-
Toolkit for weighting and analysis of nonequivalent groups
References
Halloran, P. F. Immunosuppressive drugs for kidney transplantation. N. Engl. J. Med. 351, 2715–2729. https://doi.org/10.1056/NEJMra033540 (2004).
Thiyagarajan, U. M., Ponnuswamy, A. & Bagul, A. Thymoglobulin and its use in renal transplantation: A review. Am. J. Nephrol. 37, 586–601. https://doi.org/10.1159/000351643 (2013).
Grafals, M. et al. Immunophenotyping and efficacy of low dose ATG in non-sensitized kidney recipients undergoing early steroid withdrawal: a randomized pilot study. PLoS ONE 9, e104408. https://doi.org/10.1371/journal.pone.0104408 (2014).
Kirk, A. D. Induction immunosuppression. Transplantation 82, 593–602. https://doi.org/10.1097/01.tp.0000234905.56926.7f (2006).
Brennan, D. C., Daller, J. A., Lake, K. D., Cibrik, D. & Del Castillo, D. Rabbit antithymocyte globulin versus basiliximab in renal transplantation. N. Engl. J. Med. 355, 1967–1977. https://doi.org/10.1056/NEJMoa060068 (2006).
Libório, A. B. et al. Induction antibody therapy in renal transplantation using early steroid withdrawal: long-term results comparing anti-IL2 receptor and anti-thymocyte globulin. Int. Immunopharmacol. 11, 1832–1836. https://doi.org/10.1016/j.intimp.2011.07.012 (2011).
Meier-Kriesche, H. U., Arndorfer, J. A. & Kaplan, B. Association of antibody induction with short- and long-term cause-specific mortality in renal transplant recipients. J. Am. Soc. Nephrol. 13, 769–772. https://doi.org/10.1681/ASN.V133769 (2002).
Opelz, G., Naujokat, C., Daniel, V., Terness, P. & Dohler, B. Disassociation between risk of graft loss and risk of non-Hodgkin lymphoma with induction agents in renal transplant recipients. Transplantation 81, 1227–1233. https://doi.org/10.1097/01.tp.0000219817.18049.36 (2006).
Thibaudin, D., Alamartine, E., Mariat, C., Absi, L. & Berthoux, F. Long-term kinetic of T-lymphocyte subsets in kidney-transplant recipients: influence of anti-T-cell antibodies and association with posttransplant malignancies. Transplantation 80, 1514–1517. https://doi.org/10.1097/01.tp.0000181193.98026.3f (2005).
Klem, P. et al. Reduced dose rabbit anti-thymocyte globulin induction for prevention of acute rejection in high-risk kidney transplant recipients. Transplantation 88, 891–896. https://doi.org/10.1097/TP.0b013e3181b6f38c (2009).
Kho, M. M. et al. The effect of low and ultra-low dosages Thymoglobulin on peripheral T, B and NK cells in kidney transplant recipients. Transpl. Immunol. 26, 186–190. https://doi.org/10.1016/j.trim.2012.02.003 (2012).
Gurk-Turner, C. et al. Thymoglobulin dose optimization for induction therapy in high risk kidney transplant recipients. Transplantation 85, 1425–1430. https://doi.org/10.1097/TP.0b013e31816dd596 (2008).
Wong, W. et al. Comparison of two dosages of thymoglobulin used as a short-course for induction in kidney transplantation. Transpl Int. 19, 629–635. https://doi.org/10.1111/j.1432-2277.2006.00270.x (2006).
Mohty, M. et al. New directions for rabbit antithymocyte globulin (Thymoglobulin((R))) in solid organ transplants, stem cell transplants and autoimmunity. Drugs 74, 1605–1634. https://doi.org/10.1007/s40265-014-0277-6 (2014).
Tsapepas, D., Mohan, S., Crew, R. J., Cohen, D., & Ratner, L. E. Small thymoglobulin dose adjustments have profound impact on early rejections in renal transplantation. Am. J. Transpl. (2011).
Yang, J. et al. Design and methods of the Korean organ transplantation registry. Transplant Direct. 3, e191. https://doi.org/10.1097/TXD.0000000000000678 (2017).
Loupy, A. et al. The Banff 2015 kidney meeting report: current challenges in rejection classification and prospects for adopting Molecular pathology. Am. J. Transplant. 17, 28–41. https://doi.org/10.1111/ajt.14107 (2017).
Griffin, B. A. et al. Toolkit for weighting and analysis of nonequivalent groups (TWANG) website (RAND Corporation, 2014).
Noël, C. et al. Daclizumab versus antithymocyte globulin in high-immunological-risk renal transplant recipients. J. Am. Soc. Nephrol. 20, 1385–1392. https://doi.org/10.1681/ASN.2008101037 (2009).
Ciancio, G. et al. A randomized trial of three renal transplant induction antibodies: Early comparison of tacrolimus, mycophenolate mofetil, and steroid dosing, and newer immune-monitoring. Transplantation 80, 457–465. https://doi.org/10.1097/01.tp.0000165847.05787.08 (2005).
Lebranchu, Y. et al. Immunoprophylaxis with basiliximab compared with antithymocyte globulin in renal transplant patients receiving MMF-containing triple therapy. Am. J. Transplant. 2, 48–56. https://doi.org/10.1034/j.1600-6143.2002.020109.x (2002).
Abou-Ayache, R. et al. CMV infections after two doses of daclizumab versus thymoglobulin in renal transplant patients receiving mycophenolate mofetil, steroids and delayed cyclosporine A. Nephrol. Dial. Transplant. 23, 2024–2032. https://doi.org/10.1093/ndt/gfm873 (2008).
Mourad, G. et al. Sequential protocols using basiliximab versus antithymocyte globulins in renal-transplant patients receiving mycophenolate mofetil and steroids. Transplantation 78, 584–590. https://doi.org/10.1097/01.tp.0000129812.68794.cc (2004).
Woodle, E. S. et al. A prospective, randomized, double-blind, placebo-controlled multicenter trial comparing early (7 day) corticosteroid cessation versus long-term, low-dose corticosteroid therapy. Ann. Surg. 248, 564–577. https://doi.org/10.1097/SLA.0b013e318187d1da (2008).
Woodle, E. S., Peddi, V. R., Tomlanovich, S., Mulgaonkar, S. & Kuo, P. C. A prospective, randomized, multicenter study evaluating early corticosteroid withdrawal with Thymoglobulin in living-donor kidney transplantation. Clin. Transplant. 24, 73–83. https://doi.org/10.1111/j.1399-0012.2009.01127.x (2010).
Mehta, R. B. et al. Rabbit antithymocyte globulin dose and early subclinical and clinical rejections in kidney transplantation. Clin. Transplant. 36, e14582. https://doi.org/10.1111/ctr.14582 (2022).
Linhares, K. et al. The influence of the antithymocyte globulin dose on clinical outcomes of patients undergoing kidney retransplantation. PLoS ONE 16, e0251384. https://doi.org/10.1371/journal.pone.0251384 (2021).
Louis, K. et al. Coordinated circulating T follicular helper and activated B cell responses underlie the onset of antibody-mediated rejection in kidney transplantation. J. Am. Soc. Nephrol. 31(10), 2457–2474 (2020).
Yang J, et al. Follicular helper T cell derived exosomes promote B cell proliferation and differentiation in antibody-mediated rejection after renal transplantation. Biomed Res Int. (2019). PMID: 31223621
Yazdani, S. et al. Natural killer cell infiltration is discriminative for antibody-mediated rejection and predicts outcome after kidney transplantation. Kidney Int. 95, 188–198 (2019).
Venner, J., Hidalgo, L., Famulski, K., Chang, J. & Halloran, P. The molecular landscape of antibody-mediated kidney transplant rejection: Evidence for NK involvement through CD16a Fc receptors. Am. J. Transplant. 15, 1336–1348 (2015).
Hidalgo, L. et al. NK cell transcripts and NK cells in kidney biopsies from patients with donor-specific antibodies: Evidence for NK cell involvement in antibody-mediated rejection. Am. J. Transplant. 10, 1812–1822 (2010).
Ko, Y. et al. A prospective, randomized, non-blinded, non-inferiority pilot study to assess the effect of low-dose anti-thymocyte globulin with low-dose tacrolimus and early steroid withdrawal on clinical outcomes in non-sensitized living-donor kidney recipients. PLoS ONE 18(3), e0280924. https://doi.org/10.1371/journal.pone.0280924 (2023).
Acknowledgements
This research was supported by a fund (2014-ER6301-00, 2014-ER6301-01, 2014-ER6301-02, 2017-ER6301-00, 2017-ER6301-01, 2017-ER6301-02) by the Research of Korea Centers for Disease Control and Prevention Agency.
Author information
Authors and Affiliations
Consortia
Contributions
Participated in research design: S.S., J.H.J., H.K., Y.H.K. Participated in data collection: J.P.L., J.S.J., H.J., J.S.Y, M.S.K, KOTRY study group. Participated in the writing of the paper: S.S., Y.E.S. Participated in the performance of the research: S.S., Y.E.S., Y.K., S.J.L., H.E.K. Participated in data analysis: S.S, H.K., J.L.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shim, Y.E., Ko, Y., Lee, J.P. et al. Evaluating anti-thymocyte globulin induction doses for better allograft and patient survival in Asian kidney transplant recipients. Sci Rep 13, 12560 (2023). https://doi.org/10.1038/s41598-023-39353-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-39353-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.