Abstract
Biocontrol providing parasitoids can orientate according to volatile organic compounds (VOCs) of their host’s plants, the emission of which is potentially dependent on the availability of soil nitrogen (N). This paper aimed at finding the optimal N fertilization rate for oilseed rape (Brassica napus L.) to favor parasitism of pollen beetles (Brassicogethes aeneus Fab. syn. Meligethes aeneus Fab.) in a controlled environment. Pollen beetles preferred to oviposit into buds of plants growing under higher N fertilization, whereas their parasitoids favored moderate N fertilization. As a part of induced defense, the proportion of volatile products of glucosinolate pathway in the total oilseed rape VOC emission blend was increased. Our results suggest that the natural biological control of pollen beetle herbivory is best supported by moderate N fertilization rates.
Similar content being viewed by others
Introduction
Plants emit volatile organic compounds (VOC) in response to, and for the prevention of, herbivory. Such olfactory signals serve as cues for plant-associated insects to orient their behavior: plant odors emitted in response to herbivory can repel pests1,2 and attract natural enemies of herbivorous insects2,3 such as hymenopteran parasitoids4,5,6. Meanwhile, specialized herbivores often use the same cues to locate food plants7. The emission rates of VOC depend notably on growth conditions8, however, practical knowledge of VOC for parasitoid attraction via growth condition manipulation is still scarce9.
An abiotic factor easily manipulated by farmers is fertilization. Fertilization practices are often poorly justified and/or excessive10,11. The availability of nutrients can affect the emissions of various VOC in complex ways. Typically, the lack of a nutrient affects the production of volatiles indirectly12 via altering the overall plant physiological activity and the share of carbon allocation between primary and secondary metabolism13. Nitrogen (N) availability can increase14 or decrease15 emissions of certain volatiles, depending on plant species, growth stage and interaction with other environmental factors. In actively growing plants, high N availability speeds up primary metabolism and plant growth, while in plants with arrested growth improved nutrition can enhance storage and secondary metabolism16,17. Nitrogen is needed for the biosynthesis of glucosinolates (GLS), the anti-herbivore toxins found in the Brassica family, and their volatile derivatives isothiocyanates (emitted after tissue damage) mediate plant–insect interactions18,19,20. Since specialist herbivores can orient towards GLS21, high N in crops can even benefit pests22,23,24,25.
Oilseed rape (Brassica napus L.) has a relatively poor N use efficiency26,27 and to improve the yield28,29 is conventionally fertilized with high amounts of N that exceed 200 kg N ha−130,31. Brassica napus is the most dominating oilseed crop in Europe32 and an important ‘break crop’ of many arable rotations: oilseed rape and turnip rape (Brassica rapa L.) yields in 2019 were almost 1.5 times larger than sunflower harvest. Brassica napus is mainly cultivated for its seed, which are crushed to extract oil. The oil is used for cooking, lubricant, soaps and also for biofuels33.
Oilseed rape is attacked by a broad variety of pests. These include insects, nematodes, slugs and wood pigeons (Columbia palumbus)34. Pollen beetle (Brassicogethes aeneus Fab. syn. Meligethes aeneus Fab) is the most numerous pest of oilseed rape in Northern countries like Estonia35. Oilseed rape is most susceptible to pollen beetles during the bud stage (BBCH 51–59)36,37 when adults destroy flower buds to feed on the pollen38,39. Subsequently, beetles oviposit into 2–3 mm long buds40, and finally the emerged larvae also feed on pollen34. Although adult pollen beetles are polyphagous and feed on pollen of flowering plants of different families before and after overwintering, they prefer to lay their eggs on Brassica spp40,41. Brassica napus is especially vulnerable due to greater bud availability at a time when other food plants of B. aeneus have already passed the bud stage40.
Insecticides are a widespread weapon against pollen beetles. According to the Integrated Pest Management (IPM) strategy (promoted by the European Parliament and the Council through directive 2009/128/EC), insecticide use has several specific demands before application: pest threshold monitoring, application time (e.g. plant growth stage, weather conditions, wind velocity etc.) and choice of appropriate quantities42. Despite this, pesticides are often applied routinely without regard of pest incidence and abundance38. This situation has caused the development of pollen beetles’ resistance to pyrethroids in Europe42,43,44. The use of insecticides not only pollutes the environment38, but also kills beneficial non-target arthropods such as pollinators and natural enemies of pests34,38,42,45,46.
Sustainable biocontrol of pollen beetles relies on naturally occurring parasitoids. Parasitoids lay eggs into B. aeneus eggs and/or larvae and the emerging parasitoid larvae feed and develop in the host, eventually killing it47. The effectiveness of parasitoids has been demonstrated in natural conditions by many field studies35,48,49,50,51,52,53 as well as in controlled environment25.
In this study, we investigated the effects of N fertilization on the infestation of B. napus by B. aeneus and the parasitism rate of B. aeneus in a controlled environment to gain a detailed insight into how fertilization affects insect host preference and prevalence of parasitism and how insect performance depends on volatile emission profiles of differently fertilized plants. The main aim of our study was to find the optimal N fertilization rate for B. napus to favor parasitism rate among pollen beetles while maintaining high yield.
Two types of experiments were conducted: (i) host selection (HS) by pollen beetles and their parasitoids to link the fertilization status of plants to insect behavior; and (ii) collection of volatile organic compounds (VOC) from plants to find the characteristic components of plant odors that are responsible for attracting or repelling insects. The VOC experiment was repeated in two consequent years, 2013 and 2014.
Materials and methods
Plant and insect material
For the experiments we used cultivated Brassica napus plants and a pest insect Brassicogethes aeneus, as well as three hymenopteran parasitoid genus infecting the pest (Phradis spp., Tersilochus spp. and Diospilus spp.). None of these genus belong to endangered species registers and their use in experiments is not regulated by IUCN Policy Statement on Research Involving Species at Risk of Extinction. Wild plants were not used. In the research we followed all the relevant guidelines and legislatures of the Estonian Code of Conduct for Research Integrity.
Winter oilseed rape plants, pollen beetles and their parasitoids were obtained in Tartu County, Estonia (Supplementary Table S1). Plants were collected from agricultural field (Table S1) after overwintering at an early growth stage (BBCH 30–35) to avoid any pest infestation; thereafter replanted into 5 L pots. Plant individuals for Host selection experiment were kept in growth chamber (type KRK20, Flohr Instruments, Netherlands) with the light intensity 180 μmol m−2 s−1 at plant level (15 h day length), temperature between 2 and 10 °C and with relative humidity 65%. Plants for the VOC experiment were stored with light intensity at plant surface 180 μmol m−2 s−1, 15 h day length and relative humidity 60% in a growth room in 2013 (temperature from 18 to 25 °C) and a growth chamber (type PLG 1000, Flohr Instruments, Netherlands) in 2014 (temperature adjusted to outdoor conditions between 2 and 25 °C). In 2013, the soil was a 1:1 mix of sand and loamy soil taken from the plant growth location. In 2014, the plants were replanted into commercial peat soil (Estonia Peat Products LTD, pH 5.6–6.4) (Table 1) to improve plant nutrition and avoid pathogens naturally occurring in the field soil. In both years, plants (green/yellow bud stage, BBCH 53–59) were fertilized once on 8th of May with different levels of ammonium nitrate (NH4NO3) adjusted to soil surface area of each pot: 3 different N treatments in 2013 and 4 different treatments in 2014 (Table 1).
Host selection experiment
Beetle and parasitoid host selection experiment (HS) was conducted in a walk-in (3 × 3 m) growth chamber (type KRK20, Flohr Instruments, Netherlands). Light intensity was 180 μmol m−2 s−1 at plant level (16 h day length), relative humidity fluctuated with watering in a range between 65 and 90% and the temperature was changed according to outdoor conditions from 10 to 25 °C (the experiment lasted 3 months). As N fertilization affects the odor of B. napus individuals50, plants were placed into groups (allocation in Supplementary Fig. S1a) with the intent to prevent the contamination of VOC signals by neighboring plants with different N treatments.
Five days after N fertilization the plants BBCH (55–62) was recorded and beetles were released uniformly (24 jars opened simultaneously) (Fig. S1a) into the growth chamber to select an oilseed rape plant for feeding and oviposition. In total 240 beetles (kept without food for 24 h) were released (three times the number of plants in the growth chamber) simulating the economic threshold densities (pesticide costs lower than the cost of potential yield loss) for winter oilseed rape in Scandinavia and Baltic Countries (1–2 beetles per plant in the bud stage and four just before flowering). All beetles and open flowers found on each plant were counted after 3, 6, 24, 48 and 72 h from the release of beetles. When pollen beetle parasitoids appeared outdoors on B. napus agricultural fields (21 days after the release of pollen beetles into the growth chamber), 26 adults collected from the field same day were introduced to the chamber (Table S1, Fig. S1a). Before releasing, we checked the sex of parasitoids to be sure that there are female individuals as well but we did not count them and released them as quickly as possible to minimize their stress.
Four weeks after the beetle introduction, water and sticky paper traps were installed to catch beetle larvae as they drop to the ground for pupation. A water trap consisted of a folium plate (30 cm length, 26 cm wide, 2.5 cm high) filled with water. The pot with B. napus was placed in the center of the water trap. After the installation of traps, the plants from different fertilization groups were rearranged inside the growth chamber breaking up the groups (Fig. S1b) to prevent the larvae from migrating between the plants. Water traps were refilled when needed. Metal hoops (100 × 30 × 0.5 cm, Tarha) and polypropylene twine (Horticom) were used to secure plant branches within water trap range. Sticky paper traps (AB “Insect Control—Greenhouse”, Silvandersson, Sweden) with glue on both sides, were placed on top of the soil on each pot. Water traps were emptied weekly, the upper sides of the sticky traps once every two weeks, and the bottom sides once at the end of the experiment. The insects were collected for six weeks.
To determine the parasitism rate, all collected pollen beetle larvae were dissected. All parasitoid larvae and eggs found were counted and identified47 to species level if possible. The parasitism rate of the beetle larvae was determined for each plant.
After the collection of beetle larvae, the remaining open flowers on each plant were counted. All developed siliques were collected, counted and dried outdoors in paper bags for a week (daytime temperature 25 °C). Dried seeds were manually taken from siliques, weighed and counted automatically with a seed counter (Automatic Seed Counter Machine SLY-C, China).
Volatile organic compound (VOC) measurements
VOC collection
The upper parts of B. napus plants were enclosed individually in airtight glass chambers (3 L)54. A small fan in every chamber provided uniform gas concentrations. Four chambers were operated simultaneously having one unique treatment member per chamber; the chamber for each treatment group was chosen randomly avoiding same fertilization representative in the same chamber during next round of measurement. During the experiment, air temperature 28 °C and light intensity at plant surface 200 μmol m−2 s−1 was provided by two halogen lamps (300 W, Bemko). After the experiment, area of leaves and flower number in the chamber during the experiment were estimated.
During each VOC collection, 4 L of air was pumped 20 min through an adsorbent with an air sampling pump (224-PCXR8, Airchek Sampler, SKC Inc., USA). The cartridges were filled with three different carbon-based adsorbents to quantitatively adsorb all volatiles in C3–C17 range55. The chamber inlet air was purified with a charcoal filter (Scrubber Assy 4291, Thermo Electron Corporation).
VOCs were collected at June in 2013 and at the end of May in 2014. Plants used in the experiment had growth stages from flower buds only to already flowering (BBCH 57–65). Blank air samples were collected from each empty chamber before inserting the plants. The collections were carried out in groups of four plants (one from each treatment group, randomly placed), two groups per day, one starting at 12:00 and the other at 16:00. The plants placed into the chamber system were allowed to acclimate for half an hour before VOC collected. Thereafter, four B. aeneus individuals (starved for 24 h beforehand) were placed on each plant, the plants were left to stabilize for an hour and a new collection of VOCs was conducted. As a result, VOCs were collected from each plant before and after the release of pollen beetles. In order to prevent beetles escaping (and damage due to the fan), they were enclosed in a small transparent nylon organza bag (8.5 × 12 cm) surrounding the main raceme of each plant. Each chamber had one and the same bag during all measurements (including the blanks).
VOC Identification and calculation of emission rates
The cartridges were desorbed and volatiles analysed by a combined Shimadzu TD20 automated cartridge desorber and Shimadzu 2010 Plus GC–MS system (Shimadzu Corporation, Kyoto, Japan)56. VOC were identified based on authentic standards and comparison of the mass spectra with NIST library (National Institute of Standards and Technology, 2014). Part of the chromatograms (from 42% of all plant individuals in both years) were investigated manually to develop a custom-made mass spectral library (plant VOC only) for automatic processing with the open source software OpenChrom (Community Edition, ver. 1.2.0, Alder, lablicate.com/platform/openchrom). The following procedures and settings were used in Open Chrom: background noise reduction, AMDIS peak detection (S/N ratio = 2, medium threshold and high shape requirement), trapezoid peak integration and compound identification with 80% similarity match factor. All chromatograms were subsequently checked for false positive/negative compound identification. Empty chamber VOC concentrations were subtracted from the values with the plant and volatile emission rates were calculated57. Adsorbed air was analysed for Geranyl diphosphate (GDP), Geranylgeranyl diphosphate (GGDP), Lipoxygenase (LOX), Glucosinolate (GLS) and Shikimate (SHI) pathways VOC, where emissions were expressed per unit leaf area56,58.
Statistics
The influence of N fertilization on B. napus yield (siliques and seeds dry masses precision of 1 × 10−4 g), the abundance of B. aeneus and its parasitoids and flowers and at the harvest time were tested with One-way ANOVA (ANCOVA for parasitized larval number) type III, Fisher Least Significant Difference for Post Hoc comparison and Pearson correlation using STATISTICA 12 (Statistica 64, version 13.2 Dell Inc.). Plant observations in 0, 3, 6, 24, 48 and 72 h after the beetle release were examined with same software utilizing Repeated ANOVA t. III. All significant correlation results passed the Bonferroni correction.
VOC emissions were Log-transformed to normalize the distributions for parametrical tests. Using STATISTICA 12 the data was tested with Repeated ANOVA t. III, One-way ANOVA t. III and Spearman correlation for N-fertilization and herbivory investigation of total emission, compounds biosynthetic pathways and each compound separately. The relationships between plant growth stage, the number of flowers in the chamber during VOC collection and fertilization rate were tested with Spearman correlations. To test VOC influence on parasitoids and their offspring, an average emission from 2014 VOC experiment was applied on HS plant data (Log-transformed) and tested with Pearson correlation.
VOC and parasitoid species assemblages were visualized with non-metric multidimensional scaling (NMS). Found pattern significance was tested with Multi-Response Permutation Procedure (MRPP) using PC-ORD (ver. 6.19; McCune and Mefford 2011). Each ordination was tested at least 5 times59, stress factor < 19 and the number of randomizations was 250 for NMS. The distance measures of Sørensen (Bray–Curtis) for VOC data. Since in VOC analyses the data of the same plant individual was frequently utilized in the same test twice (non-infested by pollen beetle and infested), general relativization by compound (eg division by compound maximum value) was applied to avoid potential pseudo replication.
Treatment effects were considered statistically significant at p < 0.05. All data was checked for normality of distributions by Kolmogorov–Smirnov test before applying parametric tests.
Results
N effects on plant traits
Yield components depended significantly on fertilization rate (Table 2). There were no differences in the number of flowers between fertilization groups measured during three days after pollen beetle infestation (p > 0.9), or at the harvest time (Table 2). The number of remaining flowers during harvest was negatively correlated with the number (r = − 0.518, n = 79, p < 0.001) and mass (r = − 0.389, n = 79, p < 0.001) of seeds along with the number of siliques per plant (r = − 0.536, n = 79, p < 0.001). Seed number and mass per plant correlated as well (r = 0.860, n = 79, p < 0.001).
Seed number per silique was weakly correlated to N fertilization level (r = − 0.251; p = 0.026), the highest value (i.e. most seeds per one silique) was found in untreated plants. Silique number per plant correlated with seed number (r = 0.791, n = 79, p < 0.001) and seed mass per plant (r = 0.670, n = 79, p < 0.001).
Pest abundance effects on yield
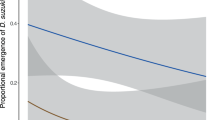
Although seed mass per plant was not linearly correlated with additional nitrogen (Fig. 1), it was in negative linear dependence on B. aeneus larval number per plant (r = − 0.242, n = 79, p = 0.032).
The abundance of pollen beetle larvae was correlated to the number of parasitoids (r = 0.540, n = 79, p < 0.001), however the latter has no significant correlation with other yield traits.
N and year effects on VOC emission
In total, 32 different VOCs were identified in both years (Tables S2–S3). Nitrogen fertilization (A = 0.075, p < 0.001) and the year of experiment (A = 0.075, p < 0.001) had an impact on plant VOC pathways conforming to MRPP test. Half of all N groups were not distinct from each other in MRPP pairwise comparisons (Fig. S2a under the graph).
According to MRPP test volatile compounds were influenced by N fertilization (A = 0.073, p < 0.001) and by year (A = 0.045, p < 0.001) (Fig. S2b). Few N groups were not distinct from each other in MRPP pairwise comparisons (Fig. S2b under the graph). VOC were correlated with the number of flowers and BBCH at the period of beetle release (both r2 > 0.1).
In 2013, there was no significant N fertilization impact on LOX, GDP, GGDP and GLS pathway compounds as well as total VOC. Only SHI pathway products were affected by additional N (F2,20 = 4.028; p = 0.034), with lower emissions for unfertilized plants (Table S2, Fig. 2a).
Emission rates (mean + SE) of different VOC formation pathways (a, d, e) and individual compounds (b, c, f, g, h, i) from Brassica napus plants in 2013 and 2014 in relation to nitrogen fertilization before (white bars) and during pollen beetle infestation (black bars). SHI—shikimate pathway volatiles; GDP—geranyl diphosphate pathway volatiles; C7–C10—long-chained saturated aldehydes.
In 2014, fertilization had no impact on LOX, GGDP, GLS and SHI pathway products as well as total VOC. The number of flowers VOC was collected from was negatively correlated with fertilization rate (rs = − 0.623; n = 11, p = 0.041), but not with growth stage of B. napus (rs = − 0.550; n = 11, p = 0.079) in 2014.
Beetle effects on VOC emission
The smell of B. napus did not significantly change (MRPP test: A = − 0.009, p = 1, Fig. S2a and A = 0.008, p = 1, Fig. S2b centroids adjoin in NMS ordination) with beetle presence (both years). However, GLS pathway products share from total emission increased by 3% in 2013 and by 42% in 2014 with beetle infestation. VOC emissions from different pathways were associated with flowers and BBCH during infestation with beetles (Fig. S2a).
In 2013 among individual LOX representatives 2-pentanone (F2,20 = 3.619; p = 0.046) has been affected by fertilization before infestation with beetles (less emission from N0 individuals, Table S2) but not after. In the same year GDP pathway compound 3-carene (F2,20 = 3.857; p = 0.038) emission increased after infestation for N0 and N120 but decreased for N20 plants (Table S2, Fig. 2b). Among SHI pathway members benzaldehyde emission moderately increased along the fertilization gradient (rs = 0.443, n = 23, p = 0.034) before beetle appearance and fertilized plants had higher emission rate of benzene (F2,20 = 4.503; p = 0.024) in 2013 (Table S2, Fig. 2c). Infested plants emissions of acetaldehyde (rs = 0.44, n = 23, p = 0.036) and acetone (rs = 0.541, n = 23, p = 0.008) were enhanced by additional nitrogen in 2013 (Table S2). In the same year the number of flowers (inside chamber during VOC collection) and the growth stage of B. napus did not correlate with fertilization rate.
In 2014 GDP pathway products emission decreased after B. aeneus appearance (F1,7 = 8.455, p = 0.023; Table S3, Fig. 2d). In the same year long-chained saturated aldehydes had strong negative correlation with nitrogen fertilization during beetle activity (rs = − 0.713, n = 11, p = 0.014) but not before (Table S3, Fig. 2e). Among individual GDP volatiles in 2014, emissions of α-pinene were significantly reduced as a reaction to the beetles (F1,7 = 9.062; p = 0.020; Fig. 2f), whereas limonene emission was reduced for control and intensive nitrogen groups, while plants with moderate fertilization—N80—showed an increase (F3,7 = 5.798; p = 0.026; Table S3, Fig. 2g). The LOX pathway product 2-pentanone (F3,7 = 6.246, p = 0.022) as well as GLS pathway product isocyanatocyclohexane (F3,7 = 4.425, p = 0.048) was affected by N fertilization in uninfested plants (2014) as follows: there was no emission in N80 plants, while the highest values were in N100 plants (Table S3). Beetle activity reduced the emissions of the short-chained oxygenated compound acetaldehyde (F1,7 = 5.836; p = 0.046; Table S3, Fig. 2h). Among long-chained saturated aldehydes, decanal emission was negatively correlated with fertilization rate both before the infestation with B. aeneus (rs = − 0.613, n = 11, p = 0.045) and after (rs = − 0.845, n = 11, p = 0.001) (Table S3, Fig. 2i), with higher emission after infestation. Oilseed rape top part (the VOC was collected from) flowers were negatively correlated with fertilization rate (rs = − 0.623; n = 11, p = 0.041), but not with growth stage of B. napus (rs = − 0.550; n = 11, p = 0.079) in 2014. Several individual compounds were positively or negatively correlated with the number of beetles and/or parasitoids (Table 3), indicating potential attractants and repellents for these insects.
N effects on pest and parasitoid preferences
In total, 957 B. aeneus larvae were collected, 90.7% (868) were uninfested by parasitoids, 7.1% (68) had one parasitoid and 2.2% (21) had more than one. Five parasitoid species were found, three of which were identified to the species level (Table 4). The most abundant parasitoid species found in B. aeneus larvae was Diospilus capito Nees, followed by Tersilochus heterocerus Thomson. The highest number of B. aeneus larvae was found on N160 plants and the lowest on N100 while greatest parasitism rate was discovered on N80 plants and the lowest on N160 (Table 4). The choice of food plant of pollen beetles did not depend on N treatment (p > 0.2) during the first three days and at the end of experiment (Table 2) except for Post Hoc comparison where N160 treatment differed from unfertilized and N100 plants.
Parasitized larvae number per plant depended on N fertilization (Table 2) but not on parasitoid species according to the MRPP test (A = 0.025 p = 0.07). However, N0 and N80 groups were distinct (A = 0.05, p = 0.019) in pairwise comparison.
Discussion
N effects on plant traits
Nitrogen fertilization affected the yield of oilseed rape (Table 2). Total mass and number of seeds per plant did not differ much between fertilized plants, but all these groups had higher yields than N0 (Fig. 1). Similar results were found in other studies50,60,61,62.
Fertilization with N had a clear positive effect on the number of siliques (Table 2, Fig. 1) as in open field conditions28,50 and there was a weak negative correlation on seed number per silique. The trend of decreased number of seeds per siliques with higher N-fertilization may reflect low nitrogen use efficiency63.
Pest abundance effects on yield
The relationship between yield and pest abundance was not linear in our study (Fig. 1). Although N160 plants had the highest pollen beetle infestation, this did not in general decrease the yield of N160 plants. Pest damage might have caused earlier silique formation64,65 and since adult pollen beetles do not feed on seeds, this may imply the pest resistance mechanism called avoidance66. As a result, these plants had the highest silique quantity (Fig. 1).
On the other hand, oilseed rape plants can respond to the floral bud losses caused by pollen stealing by B. aeneus with another well-developed compensation strategy. Compensation is usually achieved by the production of new floral buds67 on already existing or newly developed branches68. The formation of new branches and inflorescences compensates for floral bud losses and finally, may also increase the ratio of seed to total plant mass, increasing crop productivity69. Therefore, despite distinct amount of pollen beetle larvae in zero and highest fertilization treatments, seed mass per plant was similar for mentioned groups70(Table 2).
N and year effects on VOC emission
Nitrogen fertilization impacted VOCs emission differently between experimental years. In 2013, SHI pathway compound emissions (especially benzene and benzaldehyde) from fertilized plants were higher compared to unfertilized, regardless of herbivory due to better nitrogen supply71. In the latter research the concentration of SHI compound benzoic acid increased with N fertilization due to primary and secondary metabolites competition reduction. In infested plants, fertilization rate was positively correlated with acetone and acetaldehyde (short-chained oxygenated volatiles) production. This may be ecologically significant, since both of these compounds are considered to be hazardous VOCs and can be emitted by plants in substantial quantities in certain environmental stress conditions72. On the other hand, high background emissions of these compounds can reflect higher activity of certain metabolic pathways such as isoprenoid or lipid metabolism73, which is plausible in more actively growing plants at higher level of fertilization. Moderately fertilized plants (N20 in 2013 and N80, N100 in 2014) frequently showed different results in our study, most probably because these plants were optimally fertilized, as opposed to others that were under or over fertilized with different nutrients74,75.
In 2014, a strong negative correlation with N-fertilization was revealed, such that no decanal emission was detected in N160. A similar trend was found same year for the whole class of long-chained saturated aldehydes (C7–C10), with no detected emission in N160 and negative correlation with N-fertilization during beetle activity. Since the number of flowers during VOC collection was negatively correlated with N fertilization, we suggest that despite decanal and other long-chained saturated aldehydes, decanal, heptanal, nonanal and octanal, are commonly emitted from plant leaves56,76,77, they can be a part of flower scent. For example, these aldehydes were components of flower scent in hawthorn (Crataegus monogyna) and raspberry (Rubus idaeus)78.
There were various quantitative and qualitative differences in the plant VOCs measured in 2013 vs 2014. Since the nutritional status of the plants was not evaluated and we used commercial soil in 2014 (field soil in 2013) we can speculate that VOC discrepancy occurred mainly due to soil quality variation between experimental years8. Since the B. napus varieties that we used emitted almost the same array of compounds, but with different relative proportions, similarly to other study79, we assume that the varieties along with different N-supply play a part in variances between the years.
Beetle effects on VOC emission
Pollen beetles affected the scent of oilseed rape. The proportion of GLS in the total emission increased in both experimental years after beetle appearance, more GDP pathway compounds were detected (emission is higher than zero) before beetle release (both years) and 2-pentanone (LOX) emission increased in 2014 (except for N100) after the infestation with beetles. GLS and LOX pathway compounds take part in induced defense against herbivores as toxins and repellents; moreover, GLS is specific to Brassicaceae18,19,80,81,82. Herbivores ability to sequester toxins repels their enemies22 explaining the negative correlations of GLS pathway (2-propenenitrile, isocyanatocyclohexane, maethanethiol) and LOX pathway (hexane) compounds with the number of pollen beetle parasitoid offspring found in our study. In addition, pollen beetles elicited the emission of GDP pathway compound 3-carene (except N20) in 2013, as in other research50 and reduced the emissions of α-pinene (except N100) and limonene (except N80) as well as total GDP emission in 2014. GDP pathway compounds are known to attract herbivores50,82; their reduction can therefore serve as induced defense, or olfactory camouflage. Pollen beetle parasitation in our study was positively correlated with (Z)-3-hexen-1-ol, α-pinene, limonene, benzothiazole, octanal and 3-carene emissions. Said compounds instigate herbivores oviposition50,83,84,85 and therefore may attract parasitoids.
Acetaldehyde served as pollen beetle repellent (Table 3) for uninfested B. napus and the infestation in 2014 provoked a reduction of acetaldehyde emission (Fig. 2h) for N0 and N80. In wooden species, acetaldehyde has been found to increase in response to herbivory86,87. However, there is a substantial variation among plant families in the mechanisms of acetaldehyde synthesis88, and different stress responses are therefore expectable. The presence of herbivores somewhat increased decanal emission in 2014 (Fig. 2i). Since decanal is negatively correlated with pollen beetle larvae in our experiment for uninfested plants, we can speculate it signals pollen beetle females to avoid oviposition and the same applies to toluene (Table 3). Pollen beetle larvae were attracted by 6-methyl–5–hepten–2–one (Table 3) which was found to also attract cattle flies89.
There is contrasting evidence about the total VOC response to herbivory in Brassica spp. In our experiments, the total emissions did not change after herbivory treatment, but an increase has been reported for cabbage (Brassica oleracea var. gemmifera)90 and a decrease for Brassica nigra infested with Pieris brassicae larvae65. Discrepancy in our experiment might be explained by different measurement times after herbivory, eg VOC were collected within 9 h in90 and 1.5 h in65. In our experiment VOC were collected 1 h after beetle release, consequently we can speculate that within 1 h B. napus total emission either does not change or could have recovered so that no difference was detected. But this hypothesis needs further investigation.
N effects on pest and parasitoids preference
Nitrogen fertilization affected the oviposition choices of pollen beetle (N160 vs unfertilized and N100 plants) and their parasitoids (Table 2). In our study at 1, 3, 6, 24, 48 and 72 h after infestation, the number of pollen beetles per plant did not differ between treatment groups. We can speculate that the beetles had initially low host plant selectivity due to food deprivation (beetles starved 24 h before the experiment). That could explain why host plant quality did not play a role during the first 72 h, but its importance increased with time based on larval numbers at different N treatments (Fig. 1). In addition, it may also mean that pollen beetles were not selective when feeding but plant quality played a role in the selection of oviposition sites.
Beetles were most abundant at the highest N level although not significantly distinct. The increase of pollen beetle larval abundance along the N gradient most probably reflects host plant quality improvement for the pests, which positively influences their fecundity22. The preference of N160 plants for oviposition by females could be related to chemical cues that insects catch with antennae on bud surface during host selection or to differences in bud size: B. aeneus females strongly prefer to oviposit into 3–5.5 mm B. napus buds91. Consequently, we can speculate that better nutrition intensified B. napus growth in the N160 group, and optimal size buds appeared earlier, attracting more females (Fig. 1). Another possible explanation is a longer period of suitable buds due to compensation strategy of floral bud losses 67 caused by herbivory. However, bud size was not measured in this study and no clear conclusions concerning its importance can be made.
Parasitism rate was the greatest at intermediate fertilization levels. The study of tritrophic interaction on another B. napus pest P. xylostella25 has shown similar results, where low nitrogen levels in leaf tissues enhance parasitoid growth rate and total number of offspring and reduce the length of pupal stage25. Parasitoid preference for N80 may also be related to N-dependent production of volatile organic compounds, which helps insects navigate in host selection5. In such case, parasitoids will not navigate towards a higher number of beetle larvae, but towards a more attractive odor bouquet. Our study revealed that the smell of (z)-3-hexen-1-ol, \(\alpha\)-pinene, benzothiazole, octanal and 3-carene potentially attract and isocyanatocyclohexane and hexane repel pollen beetle parasitoids. On the contrary, N160 plants had significantly lower parasitized larval number compared to N80. If the host plant quality grows with nitrogen fertilization, as has been proven for Coleoptera and Hymenoptera24, the larvae feeding on highly fertilized plants may have developed faster and consequently with a lesser time of exposure to parasitoids. Moreover, additional N could intensify plant toxin92,93 production, which in the case of Brassica are mainly glucosinolates (GLS)18. Brassicaceae glucosinolates deter generalist herbivores but not specialists19 such as B. aeneus 91. It has been found, however, that at high GLS concentrations even specialists cannot completely degrade toxic GLS hydrolytic products. This in turn can favor the infestation by parasitoids due to the weakened immune system of the host80. On the other hand, under the influence of toxins, weakened beetle larvae may be avoided by parasitoids in host selection25. In light of these contradicting results it is clear that further investigation is needed into the reasons why parasitoids avoid plants with a higher N fertilization.
All of the identified parasitoid species were also detected in a three-year field study in Estonia41. The latter confirms the setup of the current study was close to natural conditions in respect to parasitoid diversity, which didn’t significantly differ between N treatments. In Switzerland, despite a high parasitism rate at times (0–54%), parasitism caused only 1–2% of pollen beetle mortality in an oilseed rape field48. Parasitized larvae are palatable and vulnerable to diseases and weather events, most of the mortality was possibly caused by predators and other unspecified factors. In light of this, the current experiment provides important information about parasitism rates when carnivory and weather influences are excluded. Apparently due to low number of parental parasitoids we found only 9% parasitism rate where an average for Estonia is 17–34%41. Based in low parasitism rate in our experiment we can speculate than the prey from N80 and N100 plants was rather chosen by their quality than by general availability.
Conclusions
Fertilized with nitrogen Brassica napus plants had higher yield compared to untreated, with no difference between fertilized (N80–N160) individuals. Under more severe pollen beetle attack compared to untreated plants, N160 oilseed rape reached similar yield values, potentially avoiding and/or compensating for the herbivory. Moderately fertilized groups N80 and N100 had the highest parasitoid loads on pollen beetle larvae, with potential contributions from the following factors:
-
1.
The quality of host larvae was optimal since food plant N content was improved compared to unfertilized plants;
-
2.
The presence of pollen beetles provoked an increase of infochemicals orienting parasitoids to the attacked plants. In particular, GLS compound emissions as a proportion of total emission increased.
More research is needed to test if pollen beetles and their parasitoids react to repellents and attractants revealed in current study.
Data availability
The datasets used and/or analysed during the current study are partly available from Supplementary materials and the rest from corresponding author on reasonable request.
Change history
26 December 2022
The original online version of this Article was revised: In the original version of this Article the Supplementary Information file contained tracked changes, these have now been removed.
References
Poelman, E. H., van Loon, J. J. A. & Dicke, M. Consequences of variation in plant defense for biodiversity at higher trophic levels. Trends Plant Sci. 13, 534–541 (2008).
Degenhardt, J. et al. Restoring a maize root signal that attracts insect-killing nematodes to control a major pest. Proc. Natl. Acad. Sci. USA 106, 13213–13218 (2009).
Dicke, M. Behavioural and community ecology of plants that cry for help. Plant. Cell Environ. 32, 654–665 (2009).
Himanen, S. J. et al. Effects of elevated carbon dioxide and ozone on volatile terpenoid emissions and multitrophic communication of transgenic insecticidal oilseed rape (Brassica napus). New Phytol. 181, 174–186 (2009).
Girling, R. D. et al. Parasitoids select plants more heavily infested with their caterpillar hosts: A new approach to aid interpretation of plant headspace volatiles. Proc. Biol. Sci. 278, 2646–2653 (2011).
Tamiru, A. et al. Maize landraces recruit egg and larval parasitoids in response to egg deposition by a herbivore. Ecol. Lett. 14, 1075–1083 (2011).
Njihia, T. N. et al. Identification of kairomones of second instar nymphs of the variegated coffee bug Antestiopsis thunbergii (Heteroptera: Pentatomidae). Chemoecology 27, 239–248 (2017).
Becker, C. et al. Effects of abiotic factors on HIPV-mediated interactions between plants and parasitoids. BioMed. Res. Int. 2015, 1–18 (2015).
Brilli, F., Loreto, F. & Baccelli, I. Exploiting plant volatile organic compounds (VOCs) in agriculture to improve sustainable defense strategies and productivity of crops. Front. Plant. Sci. 10, 264 (2019).
Aoun, W. B., El Akkari, M., Flénet, F., Jacquet, F. & Gabrielle, B. Recommended fertilization practices improve the environmental performance of biodiesel from winter oilseed rape in France. J. Cleaner Prod. 139, 242–249 (2016).
Micha, E., Roberts, W., O’ Sullivan, L., O’ Connell, K. & Daly, K. Examining the policy-practice gap: the divergence between regulation and reality in organic fertiliser allocation in pasture based systems. Agric. Syst. 179, 102708 (2020).
Dudareva, N., Klempien, A., Muhlemann, J. K. & Kaplan, I. Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol. 198, 16–32 (2013).
Ormeño, E. & Fernandez, C. Effect of soil nutrient on production and diversity of volatile terpenoids from plants. Curr. Bioact. Compd. 8, 71–79 (2012).
Hu, B. et al. VOC emissions and carbon balance of two bioenergy plantations in response to nitrogen fertilization: A comparison of Miscanthus and Salix. Environ. Pollut. 237, 205–217 (2018).
Olson, D. M., Cortesero, A. M., Rains, G. C., Potter, T. & Lewis, W. J. Nitrogen and water affect direct and indirect plant systemic induced defense in cotton. Biol. Control. 49, 239–244 (2009).
Rosatto, L., Lainé, P. & Ourry, A. Nitrogen storage and remobilization in Brassica napus L. during the growth cycle: Nitrogen fluxes within the plant and changes in soluble protein patterns. J Exp Bot 52, 1655–1663 (2001).
Yoneyama, T., Ito, O. & Engelaar, W. M. H. G. Uptake, metabolism and distribution of nitrogen in crop plants traced by enriched and natural 15N: Progress over the last 30 years. Phytochem. Rev. 2, 121–132 (2003).
Fahey, J. W., Zalcmann, A. T. & Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 56, 5–51 (2001).
Mithen, R. F. Glucosinolates and their degradation products. Adv. Bot. Res. 35, 213–262 (2001).
García-Coronado, H. et al. Analysis of a suppressive subtractive hybridization library of Alternaria alternata resistant to 2-propenyl isothiocyanate. Electron. J. Biotechnol. 18, 320–326 (2015).
Renwick, J. A. A., Haribal, M., Gouinguené, S. & Städler, E. Isothiocyanates stimulating oviposition by the diamondback moth, Plutella xylostella. J. Chem. Ecol. 32, 755–766 (2006).
Awmack, C. S. & Leather, S. R. Host plant quality and fecundity in herbivorous insects. Annu. Rev. Entomol. 47, 817–844 (2002).
Behmer, S. T. Insect herbivore nutrient regulation. Annu. Rev. Entomol. 54, 165–187 (2009).
Butler, J., Garratt, M. P. D. & Leather, S. R. Fertilisers and insect herbivores: a meta-analysis. Ann. Appl. Biol. 161, 223–233 (2012).
Soufbaf, M., Fathipour, Y., Zalucki, M. P. & Hui, C. Importance of primary metabolites in canola in mediating interactions between a specialist leaf-feeding insect and its specialist solitary endoparasitoid. Arthropod-Plant Interact. 6, 241–250 (2012).
De Vries, S. C., van de Ven, G. W. J., van Ittersum, M. K. & Giller, K. E. Resource use efficiency and environmental performance of nine major biofuel crops, processed by first-generation conversion techniques. Biomass Bioenergy 34, 588–601 (2010).
Hegewald, H., Koblenz, B., Wensch-Dorendorf, M. & Christen, O. Impacts of high intensity crop rotation and N management on oilseed rape productivity in Germany. Crop Pasture sci. 67, 439–449 (2016).
Jankowski, K. J., Budzyński, W. S., Załuski, D., Hulanicki, P. S. & Dubis, B. Using a fractional factorial design to evaluate the effect of the intensity of agronomic practices on the yield of different winter oilseed rape morphotypes. Field. Crop. Res. 188, 50–61 (2016).
Chakwizira, E. et al. Effects of nitrogen rate on nitrate-nitrogen accumulation in forage kale and rape crops. Grass. Forage Sci. 70, 268–282 (2015).
Rathke, G. W., Behrens, T. & Diepenbrock, W. Integrated nitrogen management strategies to improve seed yield, oil content and nitrogen efficiency of winter oilseed rape (Brassica napus L.): A review. Agric. Ecosyst. Environ. 117, 80–108 (2006).
Henke, J., Breustedt, G., Sieling, K. & Kage, H. Impact of uncertainty on the optimum nitrogen fertilization rate and agronomic, ecological and economic factors in an oilseed rape based crop rotation. J. Agric. Sci. 145, 455–468 (2007).
Eurostat. Agriculture, Forestry and Fishery Statistics (Publications Office of the European Union, 2020). https://doi.org/10.2785/143455.
Zapata, N., Vargas, M., Reyes, J. F. & Belmar, G. Quality of biodiesel and press cake obtained from Euphorbia lathyris, Brassica napus and Ricinus communis. Ind. Crops Prod. 38, 1–5 (2012).
Alford, D. V., Nilsson, C. & Ulber, B. Insect pests of oilseed rape crops. In Biocontrol of Oilseed Rape Pests (ed. Alford, D. V.) 9–42 (Blackwell Science, 2003).
Veromann, E., Luik, E., Metspalu, L. & Williams, I. Key pests and their parasitoids on spring and winter oilseed rape in Estonia. Entomol. Fennica 17, 4 (2006).
Meier, U. (ed.) Growth Stages of Mono-and Dicotyledonous Plants: BBCH Monograph (Blackwell Wissenschaft, 1997).
Lancashire, P. D. et al. A uniform decimal code for growth stages of crops and weeds. Ann. Appl. Biol. 119, 561–601 (1991).
Williams, I. H. The major insect pests of oilseed rape in Europe and their management: An overview. In Biocontrol-Based Integrated Management of Oilseed Rape Pests (ed. Williams, I. H.) 1–43 (Springer, 2010).
Williams, I. H. & Free, J. B. The feeding and mating behaviour of pollen beetles (Meligethes aeneus Fab.) and seed weevils (Ceutorhynchus assimilis Payk.) on oil-seed rape (Brassica napus L.). J. Agric. Sci. 91, 453–459 (1978).
Ekbom, B. & Borg, A. Pollen beetle (Meligethes aeneus) oviposition and feeding preference on different host plant species. Entomol. Exp. Appl. 78, 291–299 (1996).
Kaasik, R. et al. Meligethes aeneus oviposition preferences, larval parasitism rate and species composition of parasitoids on Brassica nigra, Raphanus sativus and Eruca sativa compared with on Brassica napus. Biol. Control 69, 65–71 (2014).
Thieme, T., Heimbach, U. & Müller, A. Chemical control of insect pests and insecticide resistance in oilseed rape. In Biocontrol-based integrated management of oilseed rape pests (ed. Williams, I. H.) 313–335 (Springer, 2010). https://doi.org/10.1007/978-90-481-3983-5_12.
Slater, R. et al. Pyrethroid resistance monitoring in European populations of pollen beetle (Meligethes spp.): A coordinated approach through the Insecticide Resistance Action Committee (IRAC). Pest. Manag. Sci. 67, 633–638 (2011).
Zimmer, C. T., Köhler, H. & Nauen, R. Baseline susceptibility and insecticide resistance monitoring in European populations of Meligethes aeneus and Ceutorhynchus assimilis collected in winter oilseed rape. Entomol Exp Appl 150, 279–288 (2014).
Mota-Sanchez, D., Whalon, M. E., Hollingworth, R. M. & Xue, Q. 2008. Documentation of pesticide resistance in arthropods. In Global Pesticide Resistance in Arthropods (eds Whalon, M. E. et al.) 32–39 (Cromwell Press, Berlin, 2008).
Willow, J., Silva, A., Veromann, E. & Smagghe, G. Acute effect of low-dose thiacloprid exposure synergised by tebuconazole in a parasitoid wasp. PLoS ONE 14, e0212456 (2019).
Osborne, P. Observations on the natural enemies of Meligethes aeneus (F.) and M. viridescens (F.) [Coleoptera: Nitidulidae]. Parasitology 50, 91–110 (1960).
Büchi, R. Mortality of pollen beetle (Meligethes spp.) larvae due to predators and parasitoids in rape fields and the effect of conservation strips. Agric. Ecosyst. Environ. 90, 255–263 (2002).
Veromann, E., Saarniit, M., Kevväi, R. & Luik, A. Effect of crop management on the incidence of Meligethes aeneus Fab. and their larval parasitism rate in organic and conventional winter oilseed rape. Agronomy Res. 7, 548–554 (2009).
Veromann, E. et al. Effects of nitrogen fertilization on insect pests, their parasitoids, plant diseases and volatile organic compounds in Brassica napus. Crop Prot 43, 79–88 (2013).
Kovács, G. et al. Effects of land use on infestation and parasitism rates of cabbage seed weevil in oilseed rape. Pest Manag Sci 75, 658–666 (2019).
Kaasik, R., Kovács, G., Toome, M., Metspalu, L. & Veromann, E. The relative attractiveness of Brassica napus, B. rapa, B. juncea and Sinapis alba to pollen beetles. Bio. Control. 59, 19–28 (2014).
Lucas-Barbosa, D. et al. Endure and call for help: strategies of black mustard plants to deal with a specialized caterpillar. Funct. Ecol. 31, 325–333 (2017).
Toome, M. et al. Leaf rust induced volatile organic compounds signalling in willow during the infection. Planta 232, 235–243 (2010).
Kännaste, A., Copolovici, L. & Niinemets, Ü. Gas chromatography–mass spectrometry method for determination of biogenic volatile organic compounds emitted by plants. Methods Mol. Biol. 1153, 161–169. https://doi.org/10.1007/978-1-4939-0606-2_11 (2014).
Kask, K., Kännaste, A., Talts, E., Copolovici, L. & Niinemets, Ü. How specialized volatiles respond to chronic and short-term physiological and shock heat stress in Brassica nigra. Plant Cell Environ. 39, 2027–2042 (2016).
Niinemets, Ü. et al. Estimations of isoprenoid emission capacity from enclosure studies: measurements, data processing, quality and standardized measurement protocols. Biogeosciences 8, 2209–2246 (2011).
Copolovici, L., Kännaste, A., Remmel, T., Vislap, V. & Niinemets, Ü. Volatile emissions from Alnus glutionosa induced by herbivory are quantitatively related to the extent of damage. J. Chem. Ecol. 37, 18–28 (2011).
Peck, J. E. In Multivariate Analysis for Ecologists: Step-by-Step 2nd edn (ed. Peck, J. E.) (MjM Software Design, 2016).
Narits, L. Effect of nitrogen rate and application time to yield and quality of winter oilseed rape (Brassica napus L. var. oleifera subvar. biennis). Agron. Res. 8, 671–686 (2010).
Naderi, R. & Ghadiri, H. Competition of wild mustard (Sinapis arvense L.) densities with rapeseed (Brassica napus L.) under different levels of nitrogen fertilizer. J. Agr. Sci. Technol. 13, 45–51 (2011).
Grzebisz, W., Łukowiak, R. & Kotnis, K. Evaluation of nitrogen fertilization systems based on the in-season variability in the nitrogenous growth factor and soil fertility factors—A case of winter oilseed rape (Brassica napus L.). Agronomy 10, 1701 (2020).
He, H. et al. Genotypic variation in nitrogen utilization efficiency of oilseed rape (Brassica napus) under contrasting N supply in pot and field experiments. Front. Plant. Sci. 8, 1825 (2017).
Pashalidou, F. G., Lucas-Barbosa, D., van Loon, J. J. A., Dicke, M. & Fatouros, N. E. Phenotypic plasticity of plant response to herbivore eggs: Effects on resistance to caterpillars and plant development. Ecology 94, 702–713 (2013).
Lucas-Barbosa, D., Loon van, J. J. A., Gols, R., Beek van, T. A. & Dicke, M. Reproductive escape: annual plant responds to butterfly eggs by accelerating seed production. Funct. Ecol. 27, 245–254 (2013).
Milchunas, D. G. & Noy-Meir, I. Grazing refuges, external avoidance of herbivory and plant diversity. Oikos 99, 113–130 (2002).
Williams, I. H. & Free, J. B. Compensation of oil-seed rape (Brassica napus L.) plants after damage to their buds and pods. J. Agric. Sci. 92, 53–59. https://doi.org/10.1017/S0021859600060494 (1979).
Tatchell, G. Compensation in spring-sown oil-seed rape (Brassica napus L.) plants in response to injury to their flower buds and pods. J. Agric. Sci. 101, 565–573. https://doi.org/10.1017/S0021859600038594 (1983).
Tiffin, P. Mechanisms of tolerance to herbivore damage: What do we know?. Evol. Ecol. 14, 523–536. https://doi.org/10.1023/A:1010881317261 (2000).
Pinet, A., Mathieu, A. & Jullien, A. Floral bud damage compensation by branching and biomass allocation in genotypes of Brassica napus with different architecture and branching potential. Front. Plant Sci 6, 70. https://doi.org/10.3389/fpls.2015.00070 (2015).
Muzika, R. M. & Pregitzer, K. S. Effect of nitrogen fertilization on leaf phenolic production of grand fir seedlings. Trees 6, 241–244 (1992).
Kesselmeier, J. Exchange of short-chain oxygenated volatile organic compounds (VOCs) between plants and the atmosphere: A compilation of field and laboratory studies. J. Atmos. Chem. 39, 219–233 (2001).
Karl, T., Curtis, A. J., Rosenstiel, T. N., Monson, R. K. & Fall, R. Transient releases of acetaldehyde from tree leaves—Products of a pyruvate overflow mechanism?. Plant. Cell Environ. 25, 1121–1131 (2002).
Szczepaniak, W., Grzebisz, W., Potarzycki, J., Łukowiak, R. & Przygocka-Cyna, K. Nutritional status of winter oilseed rape in cardinal stages of growth as the yield indicator. Plant Soil Environ. 61, 291–296 (2015).
Anjum, N. A. et al. Improving growth and productivity of Oleiferous brassicas under changing environment: Significance of nitrogen and sulphur nutrition, and underlying mechanisms. Scientific World J. 2012, 657808 (2012).
Okereke, C. N., Liu, B., Kaurilind, E. & Niinemets, Ü. Heat stress resistance drives coordination of emissions of suites of volatiles after severe heat stress and during recovery in five tropical crops. Environ. Exp. Bot. 184, 104375 (2021).
Kanagendran, A., Pazouki, L. & Niinemets, Ü. Differential regulation of volatile emission from Eucalyptus globulus leaves upon single and combined ozone and wounding treatments through recovery and relationships with ozone uptake. Environ. Exp. Bot. 145, 21–38 (2018).
Robertson, G. W. et al. A comparison of the flower volatiles from hawthorn and four raspberry cultivars. Phytochemistry 33, 1047–1053 (1993).
Robertson, G. W., Griffiths, D. W., Smith, W. M. & Butcher, R. D. The application of thermal desorption-gas chromatography-mass spectrometry to the analyses of flower volatiles from five varieties of oilseed rape (Brassica napus spp. oleifera). Phytochem. Anal. 4, 152–157 (1993).
Kos, M. et al. Effects of glucosinolates on a generalist and specialist leaf-chewing herbivore and an associated parasitoid. Phytochemistry 77, 162–170 (2012).
Niinemets, Ü., Kännaste, A. & Copolovici, L. Quantitative patterns between plant volatile emissions induced by biotic stresses and the degree of damage. Front. Plant. Sci. 4, 262. https://doi.org/10.3389/fpls.2013.00262 (2013).
Shannon, R. W. R. et al. Something in the air? The impact of volatiles on mollusc attack of oilseed rape seedlings. Ann. Bot. 117, 1073–1082 (2016).
Ruther, J., Reinecke, A. & Hilker, M. Plant volatiles in the sexual communication of Melolontha hippocastani: Response towards time-dependent bouquets and novel function of (Z)-3-hexen-1-ol as a sexual kairomone. Ecol. Entomol. 27, 76–83 (2002).
Khan, Z. R., Pickett, J. A., Berg, J. V. D., Wadhams, L. J. & Woodcock, C. M. Exploiting chemical ecology and species diversity: Stem borer and striga control for maize and sorghum in Africa. Pest. Manag. Sci. 56, 957–962 (2000).
Jayanthi, P. D. K. et al. Specific volatile compounds from mango elicit oviposition in gravid Bactrocera dorsalis females. J. Chem. Ecol. 40, 259–266 (2014).
Hu, Z. et al. Aldehyde volatiles emitted in succession from mechanically damaged leaves of poplar cuttings. J. Plant. Biol. 51, 269–275 (2008).
Giacomuzzi, V., Mattheis, J. P., Basoalto, E., Angeli, S. & Knight, A. L. Survey of conspecific herbivore-induced volatiles from apple as possible attractants for Pandemis pyrusana (Lepidoptera: Tortricidae). Pest. Manag. Sci. 73, 1837–1845 (2017).
Torrens-Spence, M. P. et al. Structural basis for independent origins of new catalytic machineries in plant AAAD proteins. BioRxiv 404970 (2018)
Birkett, M. A. et al. The role of volatile semiochemicals in mediating host location and selection by nuisance and disease-transmitting cattle flies. Med. Vet. Entomol. 18, 313–322 (2004).
Brodmann, J. et al. Orchids mimic green-leaf volatiles to attract prey-hunting wasps for pollination. Curr. Biol. 18, 740–744 (2008).
Hervé, M. R. et al. Oviposition behavior of the pollen beetle (Meligethes aeneus): A functional study. J. Insect. Behav. 28, 107–119 (2015).
Hilker, M. & Meiners, T. Plants and insect eggs: How do they affect each other?. Phytochemistry 72, 1612–1623 (2011).
Ibanez, S., Gallet, C. & Després, L. Plant insecticidal toxins in ecological networks. Toxins 4, 228–243 (2012).
Acknowledgements
This study was supported by the Institutional Research Funding project IUT36-2, and Personal Research Funding projects no. PRG1056 and PRG947 of the Estonian Research Council. We thank Gabriella Kovács for the assistance during plant and beetle collection.
Author information
Authors and Affiliations
Contributions
V.Z. carried out the experiment, collected the data, participated in data analysis and led the writing of the manuscript; T.R., E.V. conceived the ideas and designed methodology, collected and analysed the data; R.K. collected the data; Ü.N., A.K. designed the V.O.C. collection methodology, and provided input to V.O.C. and statistical analysis; All authors contributed to the subsequent drafts and approved the final version of the M.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zolotarjova, V., Remmel, T., Kännaste, A. et al. Pollen beetle offspring is more parasitized under moderate nitrogen fertilization of oilseed rape due to more attractive volatile signal. Sci Rep 12, 14294 (2022). https://doi.org/10.1038/s41598-022-18030-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-18030-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.