Abstract
Heloniadeae (Melanthiaceae) presents an East Asia–North America disjunct distribution. Different molecular and morphological data nevertheless support the tribe as a monophyletic group. However, their phylogenetic relationships and biogeographic history, together with the character evolution, are not clear. Therefore, we constructed a Bayesian phylogenetic tree for Heloniadeae using cpDNA and inferred the historical biogeography and floral character evolution. The results revealed that Heloniadeae was distributed in high-latitudes of East Asia and North America, originating since 22.2 mya. The East Asia clade migrated into southwest China, and subsequently colonized the Korean Peninsula, Taiwan, the Ryukyus, and spread northward to Japan and southern Sakhalin. The evolution of the inflorescence and number of flowers were phylogenetically conserved, associated with the historical biogeography of Heloniadeae. The inflorescences transferred from raceme to sub-umbel, and the number of flowers decreased during the dispersal process, which may be accompanied by changes in the breeding system. Besides, the anthesis period was more affected by the habitat environment than phylogenetic constraints. The flowering temperature of Heloniadeae was below 20 °C in most species, except H. kawanoi. Such a low temperature might not be conductive to pollinator activities, but it could be compensated by sustaining seed production with long-lasting flowers.
Similar content being viewed by others
Introduction
Disjunct distribution is a discontinuous distribution pattern of organisms, and many types of disjunct distributions have been reported1,2. The intercontinental disjunction between North America and East Asia is one of the most prominent disjunctions, and had been long studied, over 150 years3,4,5,6,7,8,9,10,11,12. Till now, many taxa have been reported to display such a distribution pattern, such as Sassafras13, Pogonia14, Chamaecyparis15, Maianthemum16, Cornus17, Pseudotsuga18, and Phryma19. Generally, biogeographic studies attributed such distribution patterns to relics of Tertiary temperate forests, especially the disjunction between eastern North America and East Asia20,21,22,23,24.
Heloniadeae, a tribe of Melanthiaceae, comprises three genera which form the classic disjunct distribution between eastern North America (i.e., Helonias L.) and East Asia (i.e., Heloniopsis A.Gray, and Ypsilandra Franch)25,26. Among them, Helonias is a monotypic genus with He. bullata L. (Helonias was abbreviated as He after here) distributed in eastern North America27; in contrast, Heloniopsis has five species distributed in China, Japan, Korea, and Taiwan28; Ypsilandra has five species in China and the Himalayas28. These genera have perennial herbs with rosette leaves, scapose inflorescences, tepals with various colors, and seeds with caudate appendages on both ends. They also inhabit a similar habitat of the understory, shady and moist places, except that Heloniopsis kawanoi (Koidz.) Honda had been reported to also be epiphytic on trunks29. These genera are well known for their highly varying floral characters. The diagnostic characters for species delimitation rely on floral traits, such as inflorescence type, flower number per inflorescence, color of the tepals, stamen adnation, anther type, and style and stigma morphology28,30,31. Helonias has a compact raceme and three styluli. In contrast, Heloniopsis and Ypsilandra have relatively sparse racemes or umbels, and capitate or tri-lobed stigmas. The stamen of Heloniopsis is adnate to the opposite tepal, and the anther is dorsifixed with two locules or nearly so. On the other hand, the stamen of Ypsilandra is free from the tepals, and the anther is basifixed with one locule.
Previous studies on Heloniadeae were mainly focusing on taxonomy30,31,32,33,34,35,36,37, morphology38,39,40,41, life history29,42, genetic structure43,44, species maintenance mechanism45, and pollination biology46,47. The phylogeny of Heloniadeae has been reported in Tanaka31,48, Fuse and Tamura45, Fuse et al.49,50, and Kim et al.26,51. Among them, Fuse and Tamura50 reconstructed a phylogenetic tree from five plastid loci and discussed the generic classification of Heloniadeae. Although Tanaka31,48, Fuse et al.49,50, and Kim et al.26 discussed the biogeographic history, the results were merely based on morphological and phenetic aspects31,39, considered Heloniadeae lumped in the higher rank of Melanthiaceae51, or just had limited discussions49.
These studies have provided a wealth of data for the morphology and phylogeny of these genera, showing some trends in distribution and flower characteristics. For example, raceme inflorescence was mainly in continental species, e.g. He. bullata, Ypsilandra, and H. koreana and H. tubiflora, in contrast, the sub-umbel inflorescence was only found in the insular species of Heloniopsis. According to the phylogeny of Fuse and Tamura45 and the biogeographic inference of Tanaka31, the spreading of Heloniadeae was most probably from continental to the insular environment. Therefore, the inflorescence type change might be associated with biogeography. Based on these previous morphological and molecular findings, this study aims to reconstruct the historical biogeography of Heloniadeae from a phylogenetic perspective, focusing on the origin and spreading of Heloniopsis, by the model-based Bayesian evolutionary analysis. As a result, a hypothesis of the disjunct distribution was proposed, and the reconstruction of ancestral state and trait evolution, especially the floral parts, were also explored in this study.
Results
Phylogenetic analysis
The substitution models selected for the phylogenetic analysis were HKY for atpB-rbcL and trnK, and HKY + G for the other sequences (Table 1). With Chionographis (C. chinensis and C. japonica) and Chamaelirium luteum as the outgroups, the phylogenetic tree confirms the monophyly of Heloniadeae that comprises a total of 11 taxa and one putative species from Taiwan (Fig. 1). In this phylogenetic tree, the genus Helonias diverged first from the monophyletic group of Ypsilandra and Heloniopsis. Each genus forms a highly supported monophyletic group [posterior probability (pp) = 1] (Fig. 1), as does the clade of two Ypsilandra species. Heloniopsis consisted of three highly to moderately supported clades. Among them, two Korean species (H. koreana Fuse, N.S.Lee & M.N.Tamura and H. tubiflora Fuse, N.S.Lee & M.N.Tamura) formed a highly supported clade (pp = 1) sister to the others. Among the remaining species, H. umbellata, H. leucantha (Koidz.) Honda, and Heloniopsis sp. formed a robust clade (pp = 1), and H. leucantha was sister to the others. The other taxa formed a highly supported clade (pp = 0.90), which consists of two subclades: one is comprised of two varieties of H. orientalis (Thunb.) Tanaka [var. breviscapa (Maxim.) Ohwi and var. flavida (Nakai) Ohwi], and another is composed of its autonym and H. kawanoi. However, the latter had only moderate support (pp = 0.87).
Miocene origin with vicariant diversification of Heloniadeae
The divergence time estimated in BEAST indicated an origin of Heloniadeae during the Miocene, 22.2 mya [95% highest posterior density (HPD): 29.5–11.45 mya]. The age of Ypsilandra was estimated as 5.75 mya (95% HPD 3.0–13.0 mya), and that of Heloniopsis was 2.9 mya (95% HPD 5.7–1.5 mya). The two Korean endemic species, H. koreana and H. tubiflora, can be dated to 1.25 mya (95% HPD 2.8–0.2 mya). The subtending Taiwan + Ryukyu clades diversified at 0.1 mya (95% HPD 0.25–0 mya) and 0.5 mya (95% HPD 1.3–0.05 mya), respectively. The clade of H. orioentalis + H. kawanoi dated to 1.65 mya (95% HPD 3.75–0.8 mya) (Fig. 1).
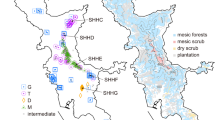
The best model for our dataset selected by BioGeoBears was DIVALIKE + J (AICc_wt = 0.51) (Supplementary Table S1). The ancestral area reconstruction suggests an ambiguous distribution for Heloniadeae, and the North America–East Asia disjunction might have been formed by a vicariance event (Fig. 2.) (Supplementary Tables S2, S3, Supplementary Fig. S4). The East Asia lineage experienced in situ cladogenesis and vicariance, resulting in two lineages, Ypsilandra and Heloniopsis. Heloniopsis further dispersed into the Ryukyus and Taiwan and separated into two lineages (viz. China + Korea and the Ryukyus + Taiwan) due to a vicariance event. The southern lineage of the Ryukyus + Taiwan further spread northward to Japan and became separate lineages in the two areas (Japan and the Ryukyus). The former one underwent in situ cladogenesis and vicariance resulting in the three present taxa. The latter one experienced cladogenesis and colonized southward to the Ryukyus again. After the vicariance event, it diversified to H. orientalis in Japan and H. kawanoi in the Ryukyus (Fig. 3).
Dispersal routes of Heloniadeae in East Asia, with the dating of each event. Double slashes mean vicariance, and the clade symbol (besides Ypsilandra) means cladogenesis. The map was created using QGIS ver. 3.16 (https://www.qgis.org), and the map source was from GADM (https://gadm.org/index.html).
Ancestral state reconstruction of selected characters
Ancestral states of the floral and phenological characters were inferred on the reconstructed phylogeny. Different character revealed dissimilar state change patterns:
-
1.
Inflorescence: The ancestral state of Heloniadeae and Ypsilandra + Heloniopsis was raceme, changing to sub-umbel in Heloniopsis. However, this state changed to raceme again in the Ryukyus + Taiwan and China + Korea clade (Fig. 4).
-
2.
Flower color: The state change of this character was more complex and with more ambiguous states than in inflorescence. The ancestral state of flower color in Heloniadeae, Ypsilandra + Heloniopsis, and Heloniopsis was ambiguous. However, the ancestral state was white in the clade of Japan + the Ryukyus + Taiwan, and it was ambiguous in the Japan clade, and white in the clade of H. orientalis var. flavida + H. orientalis var. breviscapa (Fig. 4).
-
3.
Stigma: The ancestral state in Ypsilandra + Heloniopsis was reconstructed as capitate, and the 3-styluli stigma of He. bullata was an autapomorphy. The state of Ypsilandra was trifid, and all clades of Heloniopsis were reconstructed as capitate (Fig. 4).
-
4.
Anthesis period: The ancestral state of Heloniadeae was spring to summer; however, this state became ambiguous in Heloniopsis. The state of the non-Korean species was reconstructed as summer to fall and switched to winter to spring in the Ryukyus + Taiwan clade, to spring to summer in the remaining species, and further reversely changed back to summer to fall in the clade of H. kawanoi + H. orientalis var. orientalis (Fig. 4).
-
5.
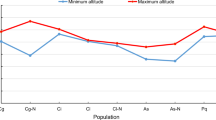
Anthesis temperature: According to the suitable mean monthly temperature (mmt) of Heloniadeae, the species could be divided into three groups (Table 2, Fig. 5). The first group (group a) comprised only H. kawanoi, which the anthesis mmt was higher than 20 °C. In the second group, comprising H. tubiflora, H. umbellata, and Heloniopsis sp., the anthesis mmt was around or below 10 °C; the third group had an anthesis mmt between 10 and 20 °C, comprised He. bullata, H. koreana, H. leucantha, H. orientalis and its varieties, Y. thibetica Franch., and Y. yunnanensis W.W.Sm. & Jeffrey (Fig. 5).
-
6.
Flower number: Flower number: The result showed reducing flower numbers in Heloniadeae over time. A reversal event was only observed in the clade of H. leucantha + Heloniopsis sp. + H. umbellata (Fig. 6).
Correlation of inflorescence change and biogeography
The result of Pagel's test between inflorescence types and biogeography was marginal significant (For the sub-umbel and raceme coding in H. koreana and H. tubiflora, results were both marginally significant with P-value = 0.051 or 0.048, respectively) for alternative coding of inflorescence types.
Discussion
In the study of Tanaka31, Heloniadeae was suggested as a member of the “Arcto-Tertiary Geoflora,” a group of deciduous broad-leaved plant communities in northern circumpolar regions52,53. The ancestor of Heloniadeae was widely distributed in the high latitudinal area of the Northern Hemisphere and dispersed southward to eastern Asia and eastern North America31. However, this inference was not well supported by the ambiguous inference of ancestral distribution of Heloniadeae in our BioGeoBEARS analysis. An analysis of plant taxa with the same (eastern North America–East Asia) distribution pattern revealed that all these taxa belonged to relic elements of temperate forest from the Tertiary23. Therefore, considering the Miocene origin and disjunct distribution pattern, we presumed that the tribe Heloniadeae might originate under the similar evolutionary pattern.
For the dating of Heloniadeae, Kim et al.51 suggested that the origin of Heloniadeae could be traced back to 27.3 mya, similar to our result (22.2 mya). Due to the lack of a fossil record in Melanthiaceae, Kim et al.51 calibrated the ages by applying fossil records of six outgroup clades. Here, we used a different strategy: an uncorrelated relaxed clock model but without fossil calibration but with an evolutionary rate of the chloroplast genes based on Wolfe et al.54. In our timeline, the divergence time between the North American and the Asian Heloniadeae is approximately Miocene (22.2 mya), which could be explained by the Beringia connection in the Miocene55. This divergence pattern was also found in some other species, e.g. Phryma19, Pseudotsuga18, Meehania56, and Deutzia57. The dating was similar to Kim et al.'s51 inference without ingroup fossils, and we expected the discovery of dated fossils in Heloniadeae or Melanthiaceae to validate this dating.
Two diversification hypotheses for the East Asian Heloniopsis have been proposed: one presumes that Heloniopsis originated in Sakhalin and spread southward into Japan and Korea49; the alternative presumed it originated in the southern part of East Asia, i.e., Taiwan and the Ryukyus, and migrated into Japan, Korea, and Sakhalin31. Generally, these two hypotheses are similar. They presume Heloniopsis originated on the islands of East Asia, spreading unidirectionally among these islands and the Korean peninsula. However, our result supports neither the inference of Fuse et al.49 nor Tanaka31. Our result shows that Heloniopsis possibly originated from southern China 5.75 mya, with a subsequent vicariance event (Fig. 2). Later, one lineage spread into the Korean Peninsula, and another colonized Taiwan and Ryukyus at approximately 2.9 mya. The colonization time roughly coincides with the formation of the islands of Taiwan and the Ryukyus58,59,60. After that, dispersal was northward to Japan and Sakhalin starting 1.65 mya, in the Pleistocene. If the Korean species spread from Sakhalin or the Ryukyus as proposed by Fuse et al.49 and Tanaka31, respectively, we would suppose that Heloniopsis inhabited areas surrounding the Korea Strait. However, populations of Heloniopsis are absent from Jeju Island, the southernmost part of the Korean peninsula, Shimayama island, and the Gotō archipelago. Besides, the dispersal of island Heloniopsis may not be unidirectional. Despite a general trend of dispersal from the southern (Taiwan + the Ryukyus) to the northern part (Sakhalin), one lineage had colonized the Ryukyus from major island of Japan, deriving H. kawanoi at approximately 1.25 mya.
States of most floral traits changed multiple times, including reversals, indicating a more complicated character evolution than Tanaka's31 inference. For the inflorescences, raceme was the primitive state, and sub-umbel was a derived one, and the flower number decreased from more than 50 flowers in He. bullata to the solitary flower of H. kawanoi, i.e. taxa with a sub-umbel usually had fewer flowers than those with a raceme. Tanaka31 supposed that the inflorescence change was pollinator- and habit-related, which improved the pollination efficiency. However, according to observations of the pollinator of He. bullata61,62 and H. orientalis46, the various flower visitors seem to be not diversified, and the flower morphology and the color under UV light also revealed a non-specialized pollination syndrome of Heloniadeae. Therefore, the shift in inflorescence type might not be attributed to the pollinator.
An extension of Baker's Law63 would be an alternative hypothesis for uniparental reproduction (self-compatibility) in plants on islands compared to their continental counterparts. The rationale is that the small population size after long-distance dispersal decreases the successful reproduction of obligate biparental populations, decreasing their fitness relative to uniparental ones at the beginning of colonization. However, many studies demonstrated the increasing dioecy on islands. This contrasting pattern was interpreted as in-situ diversification secondarily after colonization, when the selection against dioecy was relaxed through population expansion. As time goes by, Baker's Law would be obscured by subsequent local adaptation or genetic drift.
In terms of the breeding system, the dioecy–monoecy spectrum is connected with outcrossing and selfing, respectively. Besides dioecy and monoecy, floral display such as flower number and inflorescence are also related to the breeding system via pollination64,65,66,67,68. Several studies indicated that more flowers in an inflorescence could facilitate the mating opportunities; however, the self-pollination among different flowers in an inflorescence also increased, causing pollen discounting66,69,70. That is, the selfing rate would decrease by simplifying the inflorescence and reducing the flower number.
Among the Heloniadeae, Godt et al.43 found that the continental species He. bullata harbored low genetic variation due to frequent inbreeding; according to the results, a significant proportion of the seeds (20%) was produced in this way. A bagging experiment yielded seeds in 77% bagged flowers in this species61, revealing that autogamous pollination could occur under natural conditions. In contrast, a study of the island species, H. orientalis revealed a decreased selfing rate and increased outbreeding and total seed number with late flowering dates47. The bagging experiment with H. orientalis resulted in an extremely low seed: ovule ratio, revealing that autogamous pollination of this species was very limited47.
Tanaka31 also mentioned the change in the breeding system following the change in floral traits, especially the relative position between stamen and style. Based on Baker’s Law together with its connection with plant pollination and breeding, we hypothesize that the extant island species (i.e., H. leucantha and H. umbellata) with few but more aggregated flowers (i.e., sub-umbel) were evolved secondarily from their ancestors with abundant but more dispersed flowers (i.e., raceme). The more aggregated and decreased number of flowers would more easily attract pollinators and avoid inbreeding, outcompeting the early colonizing ancestors postceding the relaxation of selection against outcrossing.
Considerable flower-color variation is the characteristic of Heloniadeae31,50. However, the evolution of the flower color of Heloniadeae was less discussed. Here we show that the probability of each flower-color state was less than 50% on all nodes, revealing ambiguous patterns of ancestral states (Fig. 5). Only in the Ryukyu + Taiwan clade (H. leucantha, H. umbellata, and Heloniopsis sp.) and the clade composed of H. orientalis var. flavida and H. orientalis var. orientalis are dominated in white. All parts of the flowers of H. orientalis showed strong UV absorption without special patterns for insect vision46. The flower color variation of Heloniadeae might not act as nectary guilds like some other species71,72. Alternatively, it might provide a corresponding light color visual attraction to the pollinators under the dark understory environment29. Therefore, the flower color variation might be more influenced by habitat environments than by phylogenetic constraints. Further studies on the pollination of Heloniadeae were necessary to elucidate the flower color and pollinator interaction.
Compared to the other characters, the change of stigma states was relatively simple, both for Heloniadeae and Heloniopsis. The ancestral state of these taxa is capitate, whereas the tri-lobed one is a derived state. Except for the case of Y. thibetica, the only Ypsilandra species bearing a capitate or discoid stigma31, there was consensus about the stigma type within each genus. Although the evolution of floral organs is often tied with pollination syndrome, this seems not to be the case for the stigma of Heloniadeae.
The anthesis period reflects the flowering schedule, in which three anthesis periods were defined, viz. late spring to summer, late summer to autumn, and late winter to spring. The first period was suggested as the primitive state in Heloniadeae in this study, and the other two periods were derived ones. The spring–summer species (Heloniadeae excluding H. leucantha, H. kawanoi, H. umbellata, and Heloniopsis sp.) were all distributed in temperate or high-altitude areas; in contrast, the others are in the subtropics. Therefore, the evolution of this character might be related to adaptation to the local climate instead of phylogenetic constraints. The late winter to summer anthesis period of most Heloniadeae taxa implied most members were vernal flowering species, and the flowering phenology was susceptible to temperature change73,74.
Although there might be geographical bias caused by the rare collection record of some species, the analysis still revealed the anthesis temperature pattern of Heloniadeae. Our analysis of the mmt of the anthesis period indicated all species except H. kawanoi had an anthesis mmt below 20 °C. Furthermore, species could be classified into three groups according to their mmt, and the mmt of most temperate species were between 10 and 20 °C. For H. tubiflora and H. koreana, these closely related species are endemic to the Korean peninsula and distributed sympatrically, but have a different anthesis mmt. Such differences in phenological characteristics might affect pollinator behavior and hence maintain reproductive isolation. However, the anthesis mmt of the subtropical species, viz. H. kawanoi, H. leucantha, H. umbellata, and Heloniopsis sp., classified them into different groups. In the monophyletic group of H. leucantha, H. umbellata, and Heloniopsis sp., mmt of H. leucantha was 15–20 °C, and that of the others was around 10 °C. Such differences might be due to the distributional differences in altitude: H. leucantha is distributed in the Ryukyus, while H. umbellata and Heloniopsis sp. are endemic to the medium-altitude mountains (1500–2000 m) in Taiwan. Lower anthesis temperature was unfavorable for the pollinator. However, the long anthesis period in each flower (7–14 days)47, and the aggregated flowers, light flower color, with unspecialized pollinators still improved the pollination efficiency and resulted in mass production of seeds.
This study formulates an evolutionary hypothesis for the origin and dispersal of Heloniadeae from molecular data. The migration route of this tribe in East Asia was generally from continental to insular areas and then from south to north. Simultaneously, the inflorescence type became shorter and the number of flowers decreased during the dispersal process of Heloniopsis from North America to the continent and islands of East Asia, and accompanying the breeding system alteration. Along with the changes in the number of flowers, the evolutionary trend of these floral structures guarantees effective sexual reproduction. Our study provides a detailed biogeographic inference of Heloniadeae in East Asia and proposes a new model for the anthesis phenology of vernal species.
Methods
Source of materials
In this study, we integrated previous studies on Heloniadeae to analyze of the historical biogeography and character evolution. Therefore, we gathered morphological data from Chen et al.28, Tanaka31,75, Utech27, Fuse et al.49, Hsu et al.76, and Fuse30, and by visual examination of herbarium sheets or via online websites. The following herbaria or their websites were examined: HAST, KYO, P, TAI, TAIF, TCF, TNM, herbarium acronyms according to Thiers77. Over 100 herbarium sheets were examined for this study.
All taxa of Heloniadeae were included in this study; for the scientific names of all taxa Fuse and Tamura50 was followed. Chamaelirium luteum, Chionographis chinensis, and Chionographis japonica were selected as outgroups following the studies of Kim et al.26, and Fuse and Tamura50. Most sequences used in this study were adopted from Fuse and Tamura50 and downloaded from NCBI databases.
For new sequencing, young leaves of H. umbellata and Heloniopsis sp. were collected from Taiwan. Leaves were preserved in silica gel for DNA extraction. The newly collected taxa were not listed as protected species in the Cultural Heritage Preservation Act, the plant protection law in Taiwan. The collection location was neither the protected and reserved areas nor private land. No permission was required for the new collection of H. umbellata and Heloniopsis sp. The first author (C.T.C.) undertook formal identification of these taxa. Collection information for these materials is listed in Table 3. Voucher specimens were deposited in the herbarium of the Taiwan Forestry Research Institute (TAIF).
DNA extraction and PCR protocols
Samples of H. umbellata and Heloniopsis sp. from Taiwan were included here. Accession numbers of all sequences are listed in Supplementary Table S5. Total genomic DNA was extracted by the modified CTAB method78. Five cpDNA fragments (atpB-rbcL, matK, trnG, trnK, and trnL-F) were applied for phylogeny reconstruction. The primers of all cpDNA loci used for the polymerase chain reaction (PCR) are listed in Table 179,80,81,82. PCR amplification was conducted by the following protocol: 3 min at 94 °C for enzyme activation, and 35 cycles of 94 °C for 30 s, 57 °C for 30 s, and 72 °C for 1 min, followed by a final extension at 72 °C for 5 min. Quality and quantity of the PCR products were checked by gel electrophoresis and then they were sequenced using an ABI PRISMH3730XL DNA Sequencer (Perkin-Elmer, Foster City, CA, USA). All experiments were performed following the relevant guidelines and regulations.
Phylogeny reconstruction
The sequences (including the downloaded and newly sequenced ones) were aligned in BioEdit 7.2.583. Substitution models for the five cpDNA fragments were selected with MEGA 7.084; the models with the lowest Akaike and Bayesian Information Criterion (AIC and BIC) values were applied in the following analyses.
The five chloroplast fragments, viz. atpB-rbcL, matK, trnG, trnK, and trnL-F, were concatenated to reconstruct the phylogenetic tree. The phylogenetic tree was reconstructed using BEAST v.1.8.085. The xml file was generated in BEAUti v.1.8.085, and the substitution models selected as mentioned above were applied. The clock model was set as a strict clock with a constant rate of 2 × 10−9 per site per year54. The length of the MCMC chain reaction was set as 5 × 108 generations sampled every 50,000 generations; thus, a total of 10,000 trees were kept. Tracer v.1.686 was used to check that the values of mean and ESS in the log file were over 200. TreeAnnotator v.1.8.085 was applied to construct the consensus tree and it was visualized using FigTree v.1.4.087.
Character coding
For the biogeographic and character evolutionary analysis, distribution and selected morphological characters were coded manually. The distributional range was divided into four areas, namely North America (A), Taiwan and the Ryukyus (B), Korea and China (C), and Japan (excluding the Ryukyus) (D). These areas were selected according to the patterns of endemism and the distribution of each taxon.
Regarding the morphological analysis, we mainly chose characters, especially the floral parts, that were commonly used in the species diagnosis of Heloniadeae. Character states of each taxon were determined from herbarium specimens, living plants, and the studies of Hsu et al.76, Tanaka31,75, Utech27, Fuse et al.49, and Fuse30. The following characters were applied in this study: inflorescence type, flower color, anthesis period, flower number, and stigma type. The final list of characters is presented in Supplementary Table S1, together with their distribution. The anthesis period was classified into three categories according to the season of anthesis, namely spring to summer, summer to autumn, and winter to spring.
Biogeographic inference
RASP 4.288 was used for historical biogeographic inference. The biogeographic models, including jump dispersal events (+ J), were selected using the R package BioGeoBEARS89. The maximum distribution range was set to two areas, and the following combination of areas was excluded from the analysis, i.e. North America + Taiwan + Ryukyus (AB). The model with the highest AICc_wt was regarded as the best one for our dataset (Supplementary Table S6).
Character evolution
Ancestral states of discrete characters were inferred with BayesTraits90 of RASP 4.288 with default settings. The character with a possibility higher than 50% was labeled on the clade. Because non-discrete data cannot be applied in BayesTraits, the ancestral state of flower number was determined using the package phytools of R91,92.
In order to test the correlation between inflorescence change and biogeographic pattern, we used the Pagel's test of package phytools of R91,92. However, because only binary character state data could be tested in Pagel's test, we re-coded each taxon's distribution and inflorescence. For the distribution, we re-coded the states as continental and insular distribution; the inflorescence types were coded as raceme and sub-umbel. Some taxa (e.g., H. koreana and H. tubiflora) with both inflorescences were tested in alternative codings reciprocally.
Anthesis temperature analysis
One of the characters we were interested in was the anthesis period. The records revealed that most Heloniadeae taxa bloom between late winter and early summer and are typical cases of vernal flowering plants. The anthesis of these species is often related to vernalization73,93. Thus, we collected and organized the distribution and flowering period of each species of Heloniadeae from the Global Biodiversity Information Facility (GBIF), iNaturalist (www.inaturalist.org), naturing.net (www.naturing.net), science museum net (science-net.kahaku.go.jp), and related references28,30,49,50. Further, we used the distribution points and the CHELSA Timeseries data, a high-resolution (~ 1 km) climate database for 1979–201394,95, to extract the long-term mean monthly temperature of habitats during each species' flowering period. One-way ANOVA was used to test the similarity of the mean monthly temperature of each species' flowering period, and the Scheffe test was employed to determine significant differences (α = 0.05) among groups. To avoid bias caused by outliers, the first and third quantiles were used to describe the suitable anthesis temperature of each species. The analyzed location and their mean monthly temperature were provided in Supplementary Tables S7, S8, and S9.
Change history
27 January 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41598-022-05908-2
References
Raven, P. H. Plant species disjunctions: A summary. Ann. Mo. Bot. Gard. 59, 234–246 (1972).
Thorne, R. F. Major disjunctions in the geographic ranges of seed plants. Q. Rev. Biol. 47, 365–411 (1972).
Xiang, Q. Y., Soltis, D. E. & Soltis, P. S. The Eastern Asian and Eastern and Western North American floristic disjunction: Congruent phylogenetic patterns in seven diverse genera. Mol. Phylogenet. Evol. 10, 178–190 (1998).
Gray, A. Diagnostic characters of new species of phanerogamous plants collected in Japan by Charles Wright, Botanists of the U. S. North Pacific Exploring Expedition, with observations upon the relationship of the Japanese flora to that of North America and of other parts of the northern Temperate zone. Mem. Am. Acad. Arts 6, 377–453 (1859).
Gray, A. Forest geography and archaeology. A lecture delivered before the Harvard University Natural History Society. Am. J. Sci. Arts Ser. 3 16, 85–94, 183–196 (1878).
Hu, H. H. Late Cretaceous and Cenozoic vegetation in China, emphasizing their connections with North America. Ann. Mo. Bot. Gard. 70, 490–508 (1935).
Chaney, R. W. Tertiary centers and migration routes. Ecol. Monogr. 17, 139–148 (1947).
Li, H. L. Floristic relationships between eastern Asia and eastern North America. Trans. Am. Philos. Soc. 42, 371–429 (1952).
Koyama, T. & Kawano, S. Critical taxa of grasses with North American and eastern Asiatic distribution. Can. J. Bot. 42, 859–884 (1964).
Wolfe, J. A. Some aspects of plant geography of the Northern Hemisphere during the late Cretaceous and Tertiary. Ann. Mo. Bot. Gard. 62, 264–279 (1975).
Hsu, J. Late Cretaceous and Cenozoic vegetation in China, emphasizing their connections with North America. Ann. Mo. Bot. Gard. 70, 490–508 (1983).
Boufford, D. E. Phylogenetic utility of the internal transcribed spacers of nuclear ribosomal DNA in plants: An example from the Compositae. Mol. Phytogenet. Evol. 1, 3–16 (1992).
Nie, Z. L., Wen, J. & Sun, H. Phylogeny and biogeography of Sassafras (Lauraceae) disjunct between eastern Asia and eastern North America. Plant Syst. Evol. 267, 191–203 (2007).
Cameron, K. M. & Chase, M. W. Phylogenetic relationships of Pogoniinae (Vanilloideae, Orchidaceae): An herbaceous example of the eastern north America-eastern Asia phytogeographic disjunction. J. Plant Res. 112, 317–329 (1999).
Liao, P. C., Lin, T. P. & Hwang, S. Y. Reexamination of the pattern of geographical disjunction of Chamaecyparis (Cupressaceae) in North America and East Asia. Bot. Stud. 51, 511–520 (2010).
Meng, Y., Wen, J., Nie, Z. L., Sun, H. & Yang, Y. P. Phylogeny and biogeographic diversification of Maianthemum (Ruscaceae: Polygonatae). Mol. Phylogenet. Evol. 49, 424–434 (2008).
Xiang, Q. Y., Thomas, D. T., Zhang, W., Manchester, S. R. & Murrell, Z. Species level phylogeny of the genus Cornus (Cornaceae) based on molecular and morphological evidence-implications for taxonomy and Tertiary intercontinental migration. Taxon 55, 9–30 (2006).
Wei, X. X., Yang, Z. Y., Li, Y. & Wang, X. Q. Molecular phylogeny and biogeography of Pseudotsuga (Pinaceae): Insights into the floristic relationship between Taiwan and its adjacent areas. Mol. Phylogenet. Evol. 55, 776–785 (2010).
Nie, Z. L., Sun, H., Beardsley, P. M., Olmstead, R. G. & Wen, J. Evolution of biogeographic disjunction between eastern Asia and eastern North America in Phryma (Phrymaceae). Am. J. Bot. 93, 1343–1356 (2006).
Graham, A. History of the vegetation: Cretaceous (Maastrichtian)-Tertiary. In Flora of North America North of Mexico vol. 1. (eds. Editorial Committee) 57–70 (Oxford University Press, 1993).
Xiang, Q. Y., Soltis, D. E., Soltis, P. S., Manchester, S. R. & Crawford, D. J. Timing the Eastern Asian-Eastern North American floristic disjunction: Molecular clock corroborates paleontological estimates. Mol. Phys. Ecol. 15, 462–472 (2000).
Milne, R. I. Northern Hemisphere plant disjunctions: A window on Tertiary land bridges and climate change? Ann. Bot. 98, 465–472 (2006).
Wen, J. Evolution of eastern Asian and eastern North American disjunct distribution in flowering plants. Annu. Rev. Ecol. Evol. Syst. 30, 421–455 (1999).
Wen, J., Ickert-Bond, S., Nie, Z. L. & Li, R. Timing and modes of evolution of Eastern Asian—North American biogeographic disjunctions in seed plants. In Darwins Heritage Today: Proceedings of the Darwin 200 Beijing International Conference. (eds. Long, M. Y., Gu, H. Y., Zhou, Z. H.) 252–269 (Higher Education Press, 2010)
Angiosperm Phylogeny Group. An updated of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot. J. Linn. Soc. 181, 1–20 (2016).
Kim, S. C., Kim, J. S., Chase, M. W., Fay, M. F. & Kim, J. H. Molecular phylogenetic relationships of Melanthiaceae (Liliales) based on plastid DNA sequences. Bot. J. Linn. Soc. 181, 567–584 (2016).
Utech, F. H. Liliaceae. In Flora of North America North of Mexico vol. 26. (eds. Editorial Committee) 50–347 (Oxford University Press, 2002).
Chen, X. Q., Liang, S. G., Xui, J. M. & Tamura, M. N. Liliaceae. In Flora of China vol. 24. (eds. Wu, Z. Y. & Raven, P. H.) 73–263 (Missouri Botanical Garden Press and Beijing: Science Press, 2000).
Kawano, S., Masuda, J. & Utech, F. Life-history of Japanese plants. 9: Helonias orientalis (Thunb.) N. Tanaka (Liliaceae). Plant Species Biol. 22, 231–237 (2007).
Fuse, S. Heloniopsis. In Flora of Japan vol. IVb. (eds. Boufford, D. E. et al.) 88–91 (Kodansha, 2016).
Tanaka, N. Taxonomy, evolution and phylogeography of the genus Helonias (Melanthiaceae) revisited. Phytotaxa 390, 1–84 (2019).
Koidzumi, G. Contributiones ad cognitionem florae Asiae orientalsi. Bot. Mag. (Tokyo) 39, 299–318 (1925).
Koidzumi, G. New species of Sugerokia from the Ryukyus. Acta Phytotaxon. Geobot. 6, 46–47 (1937) (Japanese title translated by the authors).
Koidzumi, G. Contributiones ad cognitionem florae Asiae orientalis. Acta Phytotaxon. Geobot. 8, 50–61 (1939).
Kitamura, S. & Murata, G. New names and new conceptions adopted in Kitamura, Murata, and Koyama, Coloured IIIustrations of Herbaceous Plants of Japan III (Monocotyledoneae). Acta Phytotaxon. Geobot. 22, 65–74 (1966).
Tanaka, N. Phylogenetic and taxonomic studies on Helonias, Ypsilandra and Heloniopsis III. Taxonomic revision. J. Jpn. Bot. 73, 102–115 (1998).
Tanaka, N. Taxonomic treatments for two taxa of Ypsilandra (Melanthiaceae) from the Sino-Vietnamese border. J. Jpn. Bot. 84, 188–191 (2009).
Tanaka, N. Taxonomic significance of some floral characters in Helonias and Ypsilandra (Liliaceae). J. Jpn. Bot. 72, 110–116 (1997).
Tanaka, N. Evolutionary significance of the variation of the floral structure of Heloniopsis. J. Jpn. Bot. 72, 131–138 (1997).
Tanaka, N. Phylogenetic and taxonomic studies on Helonias, Ypsilandra and Heloniopsis I. Comparison of character states (1). J. Jpn. Bot. 72, 221–228 (1997).
Tanaka, N. Phylogenetic and taxonomic studies on Helonias, Ypsilandra and Heloniopsis I. Comparison of character states (2). J. Jpn. Bot. 72, 286–292 (1997).
Kawano, S. & Masuda, J. The productive and reproductive biology of flowering plants VII. Resources allocation and reproductive capacity in wild populations of Heloniopsis orientalis (Thunb.) C. Tanaka (Liliaceae). Oecologia 45, 307–318 (1980).
Godt, M. J. W., Hamrick, J. L. & Bratton, S. Genetic diversity in a threatened wetland species, Helonias bullata (Liliaceae). Conserv. Biol. 9, 596–604 (1995).
Miyazaki, Y. & Isagi, Y. Pollen flow and the intrapopulation genetic structure of Heloniopsis orientalis on the forest floor as determined using microsatellites markers. Theor. Appl. Genet. 101, 718–723 (2000).
Fuse, S. & Tamura, M. N. Investigation of maintaining mechanisms of species in case of the genus Heloniopsis (Melanthiaceae, Liliaceae sensu lato). Bunrui 3, 33–38 (2003).
Takahashi, H. The pollination biology of Heloniopsis orientalis (Thunb.) C. Tanaka (Liliaceae). Plant Species Biol. 3, 117–123 (1988).
Morinaga, S. I., Tsuji, K. & Sakai, S. Consequences of differences in flowering date on seed production in Heloniopsis orientalis (Liliaceae). Am. J. Bot. 90, 1153–1158 (2003).
Tanaka, N. Phylogenetic and taxonomic studies on Helonias, Ypsilandra and Heloniopsis II. Evolution and geographical distribution. J. Jpn. Bot. 72, 329–336 (1997).
Fuse, S., Lee, N. S. & Tamura, M. N. Biosystematic studies on the genus Heloniopsis (Melanthiaceae) II. Two new species from Korea based on morphological and molecular evidence. Taxon 53, 949–958 (2004).
Fuse, S. & Tamura, M. N. Biosystematic studies on the genus Heloniopsis (Melanthiaceae) I. Phylogeny inferred from plastid DNA sequences and taxonomic implications. Nord. J. Bot. 34, 584–595 (2016).
Kim, C., Kim, S. C. & Kim, J. H. Historical biogeography of Melanthiaceae: A case of out-of-North America through the Bering land bridge. Front. Plant Sci. 10, 396 (2019).
Shotwell, J. A. Late Tertiary biogeography of horses in the Northern Great Basin. J. Paleontol. 35, 203–217 (1961).
Grímsson, F. et al. Fagaceae pollen from the early Cenozoic of West Greenland: Revisiting Engler’s and Chaney’s Arcto-Tertiary hypotheses. Plant Syst. Evol. 301, 809–832 (2015).
Wolfe, K. H., Li, W. H. & Sharp, P. M. Rates of nucleotide substitution vary greatly among plant mitochondrial, chloroplast, and nuclear DNAs. Proc. Natl. Acad. Sci. USA 88, 9054–9058 (1987).
Tiffney, B. H. Perspectives on the origin of the floristic similarity between eastern Asia and eastern North America. J. Arnold Arbor. 66, 73–94 (1985).
Deng, T. et al. Does the Arcto-Tertiary biogeographic hypothesis explain the disjunct distribution of Northern Hemisphere herbaceous plants? The case of Meehania (Lamiaceae). PLoS One 10, e0117171. https://doi.org/10.1371/journal.pone.0117171 (2015).
Kim, C., Deng, T., Wen, J., Nie, Z. L. & Sun, H. Systematics, biogeography, and character evolution of Deutzia (Hydrangeaceae) inferred from nuclear and chloroplast DNA sequences. Mol. Phylogenet. Evol. 87, 91–104 (2015).
Chi, W. R., Namson, J. & Suppe, J. Stratigraphic record of plate interactions in the Coastal Range of eastern Taiwan. Geol. Soc. China Mem. 4, 155–194 (1981).
Teng, L. S. Extensional collapse of the northern Taiwan mountain belt. Geology 24, 949–952 (1996).
Wang, C. S. et al. Formation of the Taiwan island as a solitary wave along the Eurasian continental plate margin: Magnetic and seismological evidence. Terr. Atmos. Ocean. Sci. 13, 339–354 (2002).
Sutter, R. D. The status of Helonias bullata (Liliaceae) in the Southern Appalachians. Castanea 49, 9–16 (1984).
U.S. Fish and Wildlife Service. Swamp Pink (Helonias bullata) Recovery Plan. (eds Fish and Wildlife Reference Service) 1–39 (Fish and Wildlife Reference Service, 1991).
Pannell, J. R. et al. The scope of Baker’s law. New Phytol. 208, 656–667 (2015).
Wyatt, R. Inflorescence architecture: How flower number, arrangement and phenology affect pollination and fruit-set. Am. J. Bot. 69, 585–594 (1982).
Schmid-Hempel, P. & Speiser, B. Effects of inflorescence size on pollination in Epilobium angustifolium. Oikos 53, 98–104 (1988).
Harder, L. D., Jordan, C. Y., Gross, W. E. & Routley, M. B. Beyond floricentrism: The pollination function of inflorescences. Plant Species Biol. 19, 137–148 (2004).
Ohara, M. & Higashi, S. Effects of inflorescence size on visits from pollinators and seed set of Corydalis ambigua (Papaveraceae). Oecologia 98, 25–30 (1994).
Ohashi, K. & Yahara, T. Effects of variation in flower number on pollinator visits in Cirsium purpuratum (Asteraceae). Am. J. Bot. 85, 219–224 (1998).
Klinkhamer, P. G. L. & de Jong, T. J. Attractiveness to pollinators: A plant’s dilemma. Oikos 66, 180–184 (1993).
Suetsugu, K., Naito, R. S., Fukushima, S., Kawaita, A. & Kato, M. Pollination system and the effect of inflorescence size on fruit set in the deceptive orchid Cephalanthera falcata. J. Plant Res. 128, 585–594 (2015).
Leonard, A. S. & Papaj, D. R. ‘X’ marks the spot: The possible benefits of nectar guides to bees and plants. Funct. Ecol. 25, 1293–1301 (2011).
Hansen, D. M., der Niet, T. V. & Johnson, S. D. Floral signposts: Testing the significance of visual ‘nectar guides’ for pollinator behaviour and plant fitness. Proc. R. Soc. B 279, 634–639 (2012).
Schemske, D. W. et al. Flowering ecology of some spring woodland herbs. Ecology 59, 351–366 (1978).
Jackson, M. T. Effects of microclimate on spring flowering phenology. Ecology 47, 407–415 (1966).
Tanaka, N. Helonias. In Flora of Taiwan 2nd edition vol. 5 (eds Editorial Committee of the Flora of Taiwan) 47–49 (Department of Botany, National Taiwan University, 2000).
Hsu, T. C., Kono, Y., Chiang, T. Z. & Peng, C. I Ypsilandra (Melanthiaceae; Liliaceae sensu lato), a new generic record for Taiwan. Bot. Stud. 52, 99–104 (2011).
Thiers, B. Index Herbariorum: A global directory of public herbaria and associated staff. New York Botanical Garden. http://bluegum.nybg.org/science/index.php. (Accessed 1 July 2019). (2019).
Doyle, J. J. & Doyle, J. L. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem. Bull. 19, 11–15 (1987).
Chiang, T. Y., Schaal, B. A. & Peng, C. I Universal primers for amplification and sequencing a noncoding spacer between the atpB and rbcL genes of chloroplast DNA. Bot. Bull. Acad. Sin. 39, 245–250 (1998).
Shaw, J. et al. The tortoise and the hare II: Relative utility of 21 noncoding chloroplast DNA sequences for phylogenetic analysis. Am. J. Bot. 92, 142–166 (2005).
Litson, A. & Kadereit, J. W. Chloroplast DNA evidence for introgression and long distance dispersal in the desert annual Senecio flavus (Asteraceae). Plant Syst. Evol. 197, 33–41 (1995).
Taberlet, P., Gielly, L., Pantou, G. & Bouvert, J. Universal primers for amplification of three none-coding regions of chloroplast DNA. Plant Mol. Biol. 17, 1105–1109 (1991).
Hall, T. A. BioEdit 7.2: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 41, 95–98. https://bioedit.software.informer.com/7.2/. (Accessed 1 July 2019). (1999).
Kumar, S., Stecher, G. & Tamura, K. MEGA 7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 33, 1870–1874. https://www.megasoftware.net/. (Accessed 1 July 2019). (2016).
Drummond, A. J., Suchard, M. A., Xie, D. & Rambaut, A. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol. Biol. Evol. 29, 1969–1973. https://github.com/beast-dev/beast-mcmc. (Accessed 1 July 2019). (2012).
Rambaut, A., Drummond, A. J., Xie, D., Baele, G. & Suchard, M. A. Posterior Summarization in Bayesian Phylogenetics Using Tracer 1.7. Syst. Biol. 67, 901–904. https://github.com/beast-dev/tracer/releases/tag/v1.7.2. (Accessed 1 July 2019). (2018).
Rambaut, A. FigTree v. 1.4.0. University of Oxford, Oxford, U. K. http://tree.bio.ed.ac.uk/software/figtree/. (Accessed 1 July 2019). (2012).
Yu, Y., Blair, C. & He X. J. RASP 4: Ancestral state reconstruction tool for multiple genes and characters. Mol. Biol. Evol. 37, 604–606. http://mnh.scu.edu.cn/soft/blog/RASP/index.html. (Accessed 1 July 2019). (2020).
Matzke, N. J. Model selection in historical biogeography reveals that founder-event speciation is a crucial process in island clades. Syst. Biol. 63, 951–970 (2014).
Meade, A. & Pagel, M. BayesTraits V3.0 March 2017. http://www.evolution.rdg.ac.uk/BayesTraitsV3/BayesTraitsV3.html. (Accessed 1 July 2019). (2017).
R Core Team. R: A Language and Environment for Statistical Computing. (R Foundation for Statistical Computing, 2019). https://www.R-project.org. (Accessed 1 July 2019).
Revell, L. J. Phytools: An R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223. http://www.phytools.org/. (Accessed 1 July 2019). (2012).
Sheldon, C. C., Rouse, D. T., Finnegan, E. J., Peacock, W. J. & Dennis, E. S. The molecular basis of vernalization: The central role of flowering locus c (flc). Proc. Natl. Acad. Sci. USA 97, 3753–3758 (2000).
Karger, D. N. et al. Climatologies at high resolution for the earth’s land surface areas. Sci. Data 4, 170122. https://doi.org/10.1038/sdata.2017.122 (2017).
Karger, D. N. et al. Data from: Climatologies at high resolution for the earth’s land surface areas. Dryad Digit. Repos. https://doi.org/10.5061/dryad.kd1d4 (2017).
Acknowledgements
The authors are grateful for Dr. Nagamasu Hidetoshi for examination of herbarium sheets of KYO, and Mr. Joonhyung Jung, for examined herbarium sheets of Korean species. The authors also thank Dr. Hubert Turner, an editor with Peerwith, for his professional science editing of the manuscript. This study was supported by the project Grants no. MOST 108-2628-B-003-001 to Pei-Chun Liao, and no. MOST 108-2811-B-003-505 to Chien-Ti Chao.
Author information
Authors and Affiliations
Contributions
C.T.C. and P.C.L. designed the study and wrote the manuscript. C.C.K., J.T.C., and M.W.C. helped with data analysis and reviewed the draft. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error in the Abstract. “The flowering temperature of was below 20 °C in most species, except H. kawanoi.” now reads: “The flowering temperature of Heloniadeae was below 20 °C in most species, except H. kawanoi.”
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chao, CT., Kuo, CC., Chang, JT. et al. Evolution of floral characters and biogeography of Heloniadeae (Melanthiaceae): an example of breeding system shifts with inflorescence change. Sci Rep 11, 21494 (2021). https://doi.org/10.1038/s41598-021-01049-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-01049-0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.