Abstract
Coleoid cephalopods possess numerous complex, species-specific morphological and behavioural adaptations, e.g., a uniquely structured nervous system that is the largest among the invertebrates. The Hawaiian bobtail squid (Euprymna scolopes) is one of the most established cephalopod species. With its recent publication of the chromosomal-scale genome assembly and regulatory genomic data, it also emerges as a key model for cephalopod gene regulation and evolution. However, the latest genome assembly has been lacking a native gene model set. Our manuscript describes the generation of new long-read transcriptomic data and, made using this combined with a plethora of publicly available transcriptomic and protein sequence data, a new reference annotation for E. scolopes.
Similar content being viewed by others
Background & Summary
Coleoid cephalopods (octopus, squid, cuttlefish) comprise a molluscan clade characterised by an abundance of complex morphological and behavioural adaptations. For instance, they possess a uniquely structured nervous system that is the largest among invertebrates, enabling exceptional camouflaging ability1,2,3,4. Many cephalopod clades also evolved a multitude of novel organs such as the light organ in the bobtail squids5,6,7. The genetic basis behind these innovations remains understudied due to the lack of high-quality genomes and gene annotations. So far, only a few chromosomal-scale genomes of cephalopods have been published8,9,10,11 and, due to their large size (about 3 Gb in octopus and over 5 Gbp in many squid or cuttlefish species10,12), the gene annotation has been lagging behind.
The Hawaiian bobtail squid Euprymna scolopes has been at the centre of cephalopod molecular research, primarily as a model for symbiotic association studies for over 30 years13,14. This symbiosis entails an association of the bioluminescent bacterium Vibrio fischeri with the light organ of the squid host. Origin of the light organ is estimated to be relatively recent (within the past 80 million years15) and specific to this lineage of the bobtail squids.
More recently, E. scolopes has also become a central model for genome evolution research8,9,10,16 These studies have identified genome-wide rearrangement events10 and putatively novel regulatory landscape associated with them8. These recent genomic insights pave the way for further understanding of coleoid cephalopod gene regulation and genomic evolutionary trends that have been hypothesised to be associated with some key coleoid innovations.
Moreover, E. scolopes pioneered bobtail squids in general as emerging fruitful model systems for molecular biology thanks to their small body size, relatively easy maintenance protocols17,18 and emerging transgenic approaches19.
As such, the recently published chromosomal-scale genome of E. scolopes8 was a big step forward to making this model more broadly accessible. However, the main persisting bottleneck in this resource has been the lack of proper gene models. Gene annotation was initially published in the original publication of scaffold-level E. scolopes genome12 and this annotation has been transferred to the HiC-scaffolded genome in the most recent publication8, however, no new gene annotation was performed on this assembly.
This manuscript describes an ongoing effort to alleviate this bottleneck by creating and refining gene annotation in the E. scolopes genome using a plethora of publicly available transcriptomic and protein sequence data12,16 with newly generated long-read transcriptomic sequencing (Table 1). PacBio Iso-Seq sequencing yielded 195,212 reads, with at least 98% of reads mapped to the genome per sample. BRAKER2 predicted 39,008 gene models and 40,590 transcripts in total, which is considerably more than the previous annotation with 24,378 models (Table 2). Further comparative analyses between closely related bobtail squid genomes20,21 will help validate them.
The new annotation provided many improvements of individual loci. Examples of improvements to the gene annotation as seen on the E. scolopes genome browser are presented in Fig. 1. The main advantage of the latest annotation is also the addition of UTRs to the gene models. In total, 18,296 and 18,890 genes and 19,611 and 20,276 transcripts have 5’ UTR and 3’ UTR tags assigned to them, respectively. The average length of the 5’ UTRs and 3’ UTRs was 1842 and 1785 bp respectively. While this is likely to be an underestimate of the real UTR length, this annotation provides for an important improvement to help increase the quantification of scRNA-seq in cephalopod22,23,24 as well as regulatory genomics studies8, through proper identification of transcription start sites.
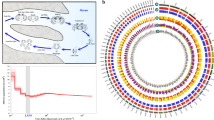
Screenshots of the previous and new gene annotations and PacBio Iso-Seq data from the Euprymna scolopes genome browser. Red lines separate tracks on the genome browser: Top; Belcaid et al. (2019) gene annotation, middle; Rogers et al. (2023) gene models55, bottom; new PacBio Iso-Seq data. Scale bars in bp are at the top of each screenshot. Yellow indicates exons and blue in the Rogers et al. (2023) annotation represents UTRs. The new gene models shown here have the following annotations according to NCBI BLASTP56 g6901; sodium bicarbonate transporter-like protein 11 isoform X2, g15477; phosphorylase b kinase regulatory subunit alpha (skeletal muscle isoform), g26475; E3 ubiquitin-protein ligase MGRN1, g15183; CDK5 and ABL1 enzyme substrate. The Euprymna genome browser can be found at: http://metazoa.csb.univie.ac.at:8000/euprymna/jbrowse.
Methods
Biological materials
All adult animal experiments were conducted in compliance with protocol number A18–029 approved by the Institutional Animal Care and Use Committee, University of Connecticut. Adult E. scolopes were collected from Maunalua Bay, Oahu, Hawaii (21°16’51.42”N, 157°43’33.07”W), and were transported to the University of Connecticut where they were maintained in recirculating artificial seawater. Animals were euthanized and tissues were sampled for RNA as described below.
RNA extraction and sequencing
Animals were anaesthetised using 2% ethanol, organs were dissected and submerged in TRIzol™. Samples were then flash frozen in liquid nitrogen and stored at −80 °C. RNA was extracted within a week of flash freezing. RNA was extracted from the hectocotylus (A1), testes, skin, left optic lobe, and central brain from one male individual of E. scolopes. Additionally, RNA was extracted from the left white body of a different E. scolopes male. Samples were processed using the TRIzol™ manufacturer’s protocol, and homogenised in 1 ml TRIzol™ in a freestanding 2 mL bead-beating tube with 0.1 mm Zirconia/Silica beads using a Qiagen PowerLyzer. The final RNA pellet was washed three times with 75% ethanol at 4 °C and resuspended in 30 µL of nuclease-free water. Next, the samples were treated with Ambion’s Turbo DNA-free kit, and their quality was assessed using an Agilent 5300 Fragment Analyzer system. RIN scores and electropherograms for extracted RNA used for PacBio Iso-Seq can be seen in supplementary Figure S1 and Table S1. Libraries were prepared using an oligo dT primer to transcribe only the polyA-mRNA and then sequenced using the PacBio Iso-Seq Sequel II 30hrs mode on one SMRTcell at the Vienna Biocenter Core Facility.
Processing and mapping of PacBio Iso-Seq data
PacBio reads were filtered for bq (barcode call quality) less than 45, reads were demultiplexed and primers were removed using Lima v.2.7.125. The PacBio Iso-Seq data was then processed according to the bulk Iso-Seq workflow found at: https://isoseq.how/getting-started.html. Here, PolyA tails and concatemers were identified and removed and hierarchical clustering was performed using IsoSeq. 3 v.3.8.225 with standard parameters. Next, reads were aligned to the E. scolopes reference genome (BioProject number PRJNA6616848) using the pbmm2 align command with default parameters in pb-assembly v.0.0.825. Bam files for each sample were then merged in Samtools v.1.726 to use for gene modelling. The merged files were then collapsed in IsoSeq. 3 in order to view them on the E. scolopes genome browser (http://metazoa.csb.univie.ac.at:8000/euprymna/jbrowse).
Gene modelling and annotation
Gene modelling was performed using BRAKER227,28,29,30,31,32,33,34,35,36,37,38,39 on the softmasked E. scolopes reference genome8. The PacBio Iso-Seq data for E. scolopes newly generated here, published Illumina RNA-seq data (mapped as in16) for E. scolopes, and publicly available protein hints files from Doryteuthis pealeii10, Octopus bimaculoides10, Nautilus pompilius40, Pecten maximus41 and Branchiostoma floridae42 were used for training (Table 1). Both Illumina RNA-seq and PacBio Iso-Seq were inputted into BRAKER2 using the —bam option, whilst protein files were specified with the–prot_seq option. Note all Illumina RNA-seq and samples inputted into BRAKER2 were male except for one female gonad sample. Once BRAKER2 had finished, untranslated regions (UTRs) were then added by running BRAKER2 again with and the Iso-Seq and RNA-seq, as well as the –addUTR = on and –skipAllTraining parameters, pointing to the augustus.hints.gtf file in the first BRAKER2 run using –AUGUSTUS_hints_preds27,28,29,35,36,43,44,45. The output of the second BRAKER2 run, gushr.gtf, was formatted for downstream analyses using the TSEBRA scripts fix_gtf_ids.py and rename_gtf.py46 and a custom perl script. We then sought to complement this with the previously available mapping of transcripts12. For this, we used GMAP version 2023–07–20 to map available Belcaid et al.12 CDS sequences to the genome. Next, bedtools v2.30.047 was used to intersect CDS regions of gushr.gtf models with the mapped Belcaid et al.12 CDS regions. We then selected Belcaid et al.12 models with two or more coding exons and that had at least 75% of their coding exons not matching BRAKER2 models and added these to the gushr.gtf annotation using a custom perl script. Lastly, CDS and exon lines were added to the GTF using another perl script.
Generation of coding sequence, protein sequence and protein annotation files
Protein sequence and coding sequence files were generated by running gffread from GffRead v0.12.748 on the reformatted gushr.gtf annotation file. Interproscan v5.62–94.049 with default parameters was used to perform annotation of the protein sequence file.
Quality checking of gene models
The previous12 and new gene annotations for E. scolopes were assessed for completeness using BUSCO v.5.4.550 with metazoa_odb10 in protein mode and OMArk v.0.3.051 with the ancestral clade Lophotrochozoa. OrthoFinder v.2.5.552 was used to count the number of orthogroups shared between each annotation and Doryteuthis pealeii10, Octopus bimaculoides10 and Pecten maximus41. The number of single- and multi-exon genes with and without protein annotation was calculated using a custom perl script along with the interproscan.tsv output file from Interproscan.
Data Records
The raw, demultiplexed PacBio Iso-Seq data underlying these analyses have been deposited in the NCBI database under Bioproject PRJNA9948253,54. The gene annotation, coding sequence, protein sequence and protein annotation files can be found on GitHub under: https://github.com/TheaFrances/E.scolopes-V2.2-BRAKER2-gene-annotation54 and Dryad under: https://doi.org/10.5061/dryad.nk98sf7xz55.
Technical Validation
The crucial improvement over the previous annotation12 was the addition of de-novo gene models on the latest chromosomal-scale assembly10 including UTR prediction and detection of many isoforms. In terms of protein coding content, our current annotation is, as expected, not substantially exceeding the BUSCO scores of the previous one12 (Tables 2 and 3). However, the OMArk results show improvement in all categories (Table 4). Additionally, the new annotation presents less missing BUSCOs compared to the previous gene annotation, highlighting the benefit of de novo gene modelling on the latest chromosomal-scale assembly. We further note that manual inspection of missing BUSCOs has yielded many loci that are present in single copies in E. scolopes genome and represented in the gene model set, but are highly divergent at the sequence level. Such genes may encode for proteins with accelerated evolutionary rates in coleoid cephalopod genomes. Further construction of an accurate coleoid cephalopod-focused single copy orthology dataset will thus be needed to properly assess genome completeness in these genomes. Note that the increase in the number of duplicated BUSCO and OMArk scores is a result of the addition of transcripts (isoforms) per gene present in the new annotation.
The number of orthogroups shared between the new annotation and D. pealeii, and shared between the new annotation and O. bimaculoides, increased compared to the orthogroups shared with the old annotation and these species. There were fewer orthogroups shared between P. maximus and the updated annotation compared with P. maximus and Belcaid et al.12 (Table 2). We find that 30,766 models were multi-exon genes, and 9,824 models were single-exon. While it is possible that single-exon models were false-positive predictions, we still were able to annotate 4,811 of them with Interproscan (compared to 25,413 in the multi-exon gene set), and thus decided to retain them in our prediction set.
The current chromosomal-scale reference genome contains many gaps (over 30%, genome assembly statistics reported in Supplementary Table 1 from Schmidbaur et al.8). BUSCO scores for the genome assembly, using metazoa_odb10 are as follows: complete 83.4%, (single: 82.9%, duplicated: 0.5%), fragmented: 10.2%, missing: 6.4%. Parallel efforts are yielding an almost gap-free reference assembly, on which the gene models presented in this paper will be transferred and improved further, potentially including the missing exons and decreasing the “missing” BUSCO count even more.
Code availability
List of commands run and scripts used are available on GitHub under: https://github.com/TheaFrances/E.scolopes-V2.2-BRAKER2-gene-annotation54.
References
Hanlon, R. T. & Messenger, J. B. Cephalopod Behaviour. (Cambridge University Press, 2018).
Shigeno, S., Andrews, P. L. R., Ponte, G. & Fiorito, G. Cephalopod Brains: An Overview of Current Knowledge to Facilitate Comparison With Vertebrates. Front. Physiol. 9, 952 (2018).
Wang, Z. Y., Ragsdale, C. W. Cephalopod nervous system organization. in Oxford Research Encyclopedia of Neuroscience (Oxford University Press, 2019).
Hanlon, R. Cephalopod dynamic camouflage. Curr. Biol. 17, R400–4 (2007).
McFall-Ngai, M. J. Giving microbes their due–animal life in a microbially dominant world. J. Exp. Biol. 218, 1968–1973 (2015).
Nyholm, S. V., Stewart, J. J., Ruby, E. G. & McFall-Ngai, M. J. Recognition between symbiotic Vibrio fischeri and the haemocytes of Euprymna scolopes. Environ. Microbiol. 11, 483–493 (2009).
Kerwin, A. H. & Nyholm, S. V. Symbiotic bacteria associated with a bobtail squid reproductive system are detectable in the environment, and stable in the host and developing eggs. Environ. Microbiol. 19, 1463–1475 (2017).
Schmidbaur, H. et al. Emergence of novel cephalopod gene regulation and expression through large-scale genome reorganization. Nat. Commun. 13, 2172 (2022).
Albertin, C. B. et al. The octopus genome and the evolution of cephalopod neural and morphological novelties. Nature 524, 220–224 (2015).
Albertin, C. B. et al. Genome and transcriptome mechanisms driving cephalopod evolution. Nat. Commun. 13, 2427 (2022).
Destanović, D. et al. A chromosome-level reference genome for the common octopus, Octopus vulgaris (Cuvier, 1797). bioRxiv https://doi.org/10.1101/2023.05.16.540928 (2023).
Belcaid, M. et al. Symbiotic organs shaped by distinct modes of genome evolution in cephalopods. Proc. Natl. Acad. Sci. USA 116, 3030–3035 (2019).
Nyholm, S. V. & McFall-Ngai, M. J. A lasting symbiosis: how the Hawaiian bobtail squid finds and keeps its bioluminescent bacterial partner. Nat. Rev. Microbiol. 19, 666–679 (2021).
Visick, K. L., Stabb, E. V. & Ruby, E. G. A lasting symbiosis: how Vibrio fischeri finds a squid partner and persists within its natural host. Nat. Rev. Microbiol. 19, 654–665 (2021).
Sanchez, G. et al. Phylogenomics illuminates the evolution of bobtail and bottletail squid (order Sepiolida). Commun Biol 4, 819 (2021).
Rouressol, L. et al. Emergence of novel genomic regulatory regions associated with light-organ development in the bobtail squid. iScience 26, 107091 (2023).
Jolly, J. et al. Lifecycle, culture, and maintenance of the emerging cephalopod models Euprymna berryi and Euprymna morsei. Frontiers in Marine Science 9, (2022).
A-review-of-the-laboratory-maintenance-rearing-and-culture-of-cephalopod-molluscs.pdf.
Crawford, K. et al. Highly Efficient Knockout of a Squid Pigmentation Gene. Curr. Biol. 30, 3484–3490.e4 (2020).
McKenna, V. et al. The Aquatic Symbiosis Genomics Project: probing the evolution of symbiosis across the tree of life. Wellcome Open Res 6, 254 (2021).
Baden, T. et al. Cephalopod-omics: Emerging Fields and Technologies in Cephalopod Biology. Integr. Comp. Biol. https://doi.org/10.1093/icb/icad087 (2023).
Gavriouchkina, D. et al. A single-cell atlas of bobtail squid visual and nervous system highlights molecular principles of convergent evolution. bioRxiv https://doi.org/10.1101/2022.05.26.490366 (2022).
Styfhals, R. et al. Cell type diversity in a developing octopus brain. Nat. Commun. 13, 7392 (2022).
Songco-Casey, J. O. et al. Cell types and molecular architecture of the Octopus bimaculoides visual system. Curr. Biol. 32, 5031–5044.e4 (2022).
pbbioconda: PacBio Secondary Analysis Tools on Bioconda. (Github).
Danecek, P. et al. Twelve years of SAMtools and BCFtools. Gigascience 10, (2021).
Hoff, K. J., Lange, S., Lomsadze, A., Borodovsky, M. & Stanke, M. BRAKER1: Unsupervised RNA-Seq-Based Genome Annotation with GeneMark-ET and AUGUSTUS. Bioinformatics 32, 767–769 (2016).
Brůna, T., Hoff, K. J., Lomsadze, A., Stanke, M. & Borodovsky, M. BRAKER2: automatic eukaryotic genome annotation with GeneMark-EP+ and AUGUSTUS supported by a protein database. NAR Genom Bioinform 3, lqaa108 (2021).
Hoff, K. J., Lomsadze, A., Borodovsky, M. & Stanke, M. Whole-Genome Annotation with BRAKER. Methods Mol. Biol. 1962, 65–95 (2019).
Brůna, T., Lomsadze, A. & Borodovsky, M. GeneMark-EP+: eukaryotic gene prediction with self-training in the space of genes and proteins. NAR Genom Bioinform 2, lqaa026 (2020).
Lomsadze, A., Ter-Hovhannisyan, V., Chernoff, Y. O. & Borodovsky, M. Gene identification in novel eukaryotic genomes by self-training algorithm. Nucleic Acids Res. 33, 6494–6506 (2005).
Buchfink, B., Xie, C. & Huson, D. H. Fast and sensitive protein alignment using DIAMOND. Nat. Methods 12, 59–60 (2015).
Gotoh, O. A space-efficient and accurate method for mapping and aligning cDNA sequences onto genomic sequence. Nucleic Acids Res. 36, 2630–2638 (2008).
Iwata, H. & Gotoh, O. Benchmarking spliced alignment programs including Spaln2, an extended version of Spaln that incorporates additional species-specific features. Nucleic Acids Res. 40, e161 (2012).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Barnett, D. W., Garrison, E. K., Quinlan, A. R., Strömberg, M. P. & Marth, G. T. BamTools: a C++ API and toolkit for analyzing and managing BAM files. Bioinformatics 27, 1691–1692 (2011).
Lomsadze, A., Burns, P. D. & Borodovsky, M. Integration of mapped RNA-Seq reads into automatic training of eukaryotic gene finding algorithm. Nucleic Acids Res. 42, e119 (2014).
Stanke, M., Diekhans, M., Baertsch, R. & Haussler, D. Using native and syntenically mapped cDNA alignments to improve de novo gene finding. Bioinformatics 24, 637–644 (2008).
Stanke, M., Schöffmann, O., Morgenstern, B. & Waack, S. Gene prediction in eukaryotes with a generalized hidden Markov model that uses hints from external sources. BMC Bioinformatics 7, 62 (2006).
Huang, Z. et al. Genomic insights into the adaptation and evolution of the nautilus, an ancient but evolving ‘living fossil’. Mol. Ecol. Resour. 22, 15–27 (2022).
Zeng, Q. et al. High-quality reannotation of the king scallop genome reveals no ‘gene-rich’ feature and evolution of toxin resistance. Comput. Struct. Biotechnol. J. 19, 4954–4960 (2021).
Simakov, O. et al. Deeply conserved synteny resolves early events in vertebrate evolution. Nat Ecol Evol 4, 820–830 (2020).
Keilwagen, J., Hartung, F. & Grau, J. GeMoMa: Homology-Based Gene Prediction Utilizing Intron Position Conservation and RNA-seq Data. in Gene Prediction: Methods and Protocols (ed. Kollmar, M.) 161–177 (Springer New York, 2019).
Keilwagen, J. et al. Using intron position conservation for homology-based gene prediction. Nucleic Acids Res. 44, e89 (2016).
Keilwagen, J., Hartung, F., Paulini, M., Twardziok, S. O. & Grau, J. Combining RNA-seq data and homology-based gene prediction for plants, animals and fungi. BMC Bioinformatics 19, 189 (2018).
Gabriel, L., Hoff, K. J., Brůna, T., Borodovsky, M. & Stanke, M. TSEBRA: transcript selector for BRAKER. BMC Bioinformatics 22, 566 (2021).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Pertea, G. & Pertea, M. GFF Utilities: GffRead and GffCompare. F1000Res. 9, 304 (2020).
Jones, P. et al. InterProScan 5: genome-scale protein function classification. Bioinformatics 30, 1236–1240 (2014).
Manni, M., Berkeley, M. R., Seppey, M., Simão, F. A. & Zdobnov, E. M. BUSCO Update: Novel and Streamlined Workflows along with Broader and Deeper Phylogenetic Coverage for Scoring of Eukaryotic, Prokaryotic, and Viral Genomes. Mol. Biol. Evol. 38, 4647–4654 (2021).
Nevers, Y. et al. Multifaceted quality assessment of gene repertoire annotation with OMArk. bioRxiv https://doi.org/10.1101/2022.11.25.517970 (2022).
Emms, D. M. & Kelly, S. OrthoFinder: phylogenetic orthology inference for comparative genomics. Genome Biol. 20, 238 (2019).
NCBI Sequence Read Archive https://identifiers.org/ncbi/insdc.sra:SRP449515 (2023).
GitHub https://github.com/TheaFrances/E.scolopes-V2.2-BRAKER2-gene-annotation (2023).
Rogers, T. Data from: Gene modelling and annotation for the Hawaiian bobtail squid, Euprymna scolopes. Dryad https://doi.org/10.5061/dryad.nk98sf7xz (2023).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Acknowledgements
We thank Koto Kon-Nanjo, Tetsuo Kon and Darrin Shultz for advice on the gene modelling. Generation of PacBio Iso-Seq data was funded by an AGA Ecology, Evolution and Conservation Genomics Research Award 2021 to T.F.R. T.F.R, G. Y. and O.S. were also supported by the European Research Council’s Horizon 2020: European Union Research and Innovation Programme, grant no. 945026. S.V.N. was supported by the Gordon and Betty Moore Foundation and the University of Connecticut. T.F.R., J.B., N.V., S.V.N. and O.S. were further supported by the Whitman Fellowship at the Marine Biological Laboratory (funded by Kuffler Research Awards, Spiegel Research Awards Fund, and L. & A. Colwin Summer Research Fellowship).
Author information
Authors and Affiliations
Contributions
T.F.R. designed and led the study, processed the PacBio Iso-Seq data and did the gene modelling. T.F.R. and O.S. wrote the manuscript under the input of all authors. G.Y. made Fig. 1 and J.B., N.V., and S.V.N. provided the samples and J.B. and N.V. did the RNA extractions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rogers, T.F., Yalçın, G., Briseno, J. et al. Gene modelling and annotation for the Hawaiian bobtail squid, Euprymna scolopes. Sci Data 11, 40 (2024). https://doi.org/10.1038/s41597-023-02903-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-023-02903-8