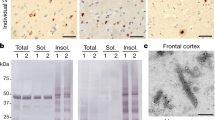
Abstract
Accumulation of abnormally phosphorylated TDP-43 (pTDP-43) is the main pathology in affected neurons of people with amyotrophic lateral sclerosis (ALS) and frontotemporal lobar degeneration (FTLD). Morphological diversity and neuroanatomical distribution of pTDP-43 accumulations allowed classification of FTLD cases into at least four subtypes, which are correlated with clinical presentations and genetic causes. To understand the molecular basis of this heterogeneity, we developed SarkoSpin, a new method for biochemical isolation of pathological TDP-43. By combining SarkoSpin with mass spectrometry, we revealed proteins beyond TDP-43 that become abnormally insoluble in a disease subtype–specific manner. We show that pTDP-43 extracted from brain forms stable assemblies of distinct densities and morphologies that are associated with disease subtypes. Importantly, biochemically extracted pTDP-43 assemblies showed differential neurotoxicity and seeding that were correlated with disease duration of FTLD subjects. Our data are consistent with the notion that disease heterogeneity could originate from alternate pathological TDP-43 conformations, which are reminiscent of prion strains.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Mass spectrometry raw files were uploaded to the ProteomeXchange Consortium via the PRIDE partner repository. Data set identifier: PXD007873. The data that support the findings of this study are available from the corresponding author upon request.
References
Ling, S. C., Polymenidou, M. & Cleveland, D. W. Converging mechanisms in ALS and FTD: disrupted RNA and protein homeostasis. Neuron 79, 416–438 (2013).
Neumann, M. et al. Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314, 130–133 (2006).
Arai, T. et al. TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem. Biophys. Res. Commun. 351, 602–611 (2006).
Buratti, E. et al. Nuclear factor TDP-43 and SR proteins promote in vitro and in vivo CFTR exon 9 skipping. EMBO J. 20, 1774–1784 (2001).
Polymenidou, M. et al. Long pre-mRNA depletion and RNA missplicing contribute to neuronal vulnerability from loss of TDP-43. Nat. Neurosci. 14, 459–468 (2011).
Tollervey, J. R. et al. Characterizing the RNA targets and position-dependent splicing regulation by TDP-43. Nat. Neurosci. 14, 452–458 (2011).
Afroz, T. et al. Functional and dynamic polymerization of the ALS-linked protein TDP-43 antagonizes its pathologic aggregation. Nat. Commun. 8, 45 (2017).
Jiang, L. L. et al. The N-terminal dimerization is required for TDP-43 splicing activity. Sci. Rep. 7, 6196 (2017).
Gu, J. et al. Transactive response DNA-binding protein 43 (TDP-43) regulates alternative splicing of tau exon 10: Implications for the pathogenesis of tauopathies. J. Biol. Chem. 292, 10600–10612 (2017).
Ederle, H. & Dormann, D. TDP-43 and FUS en route from the nucleus to the cytoplasm. FEBS Lett. 591, 1489–1507 (2017).
Dewey, C. M. et al. TDP-43 is directed to stress granules by sorbitol, a novel physiological osmotic and oxidative stressor. Mol. Cell. Biol. 31, 1098–1108 (2011).
Alami, N. H. et al. Axonal transport of TDP-43 mRNA granules is impaired by ALS-causing mutations. Neuron 81, 536–543 (2014).
Gopal, P. P., Nirschl, J. J., Klinman, E. & Holzbaur, E. L. Amyotrophic lateral sclerosis-linked mutations increase the viscosity of liquid-like TDP-43 RNP granules in neurons. Proc. Natl Acad. Sci. USA 114, E2466–E2475 (2017).
Kato, M. et al. Cell-free formation of RNA granules: low complexity sequence domains form dynamic fibers within hydrogels. Cell 149, 753–767 (2012).
Molliex, A. et al. Phase separation by low complexity domains promotes stress granule assembly and drives pathological fibrillization. Cell 163, 123–133 (2015).
Igaz, L. M. et al. Enrichment of C-terminal fragments in TAR DNA-binding protein-43 cytoplasmic inclusions in brain but not in spinal cord of frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Am. J. Pathol. 173, 182–194 (2008).
Neumann, M. et al. Phosphorylation of S409/410 of TDP-43 is a consistent feature in all sporadic and familial forms of TDP-43 proteinopathies. Acta Neuropathol. 117, 137–149 (2009).
Lashley, T., Rohrer, J. D., Mead, S. & Revesz, T. Review: an update on clinical, genetic and pathological aspects of frontotemporal lobar degenerations. Neuropathol. Appl. Neurobiol. 41, 858–881 (2015).
Neary, D. et al. Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology 51, 1546–1554 (1998).
Mackenzie, I. R. & Neumann, M. Reappraisal of TDP-43 pathology in FTLD-U subtypes. Acta Neuropathol. 134, 79–96 (2017).
Lee, E. B. et al. Expansion of the classification of FTLD-TDP: distinct pathology associated with rapidly progressive frontotemporal degeneration. Acta Neuropathol. 134, 65–78 (2017).
Nonaka, T. et al. Prion-like properties of pathological TDP-43 aggregates from diseased brains. Cell Rep. 4, 124–134 (2013).
Tsuji, H. et al. Molecular analysis and biochemical classification of TDP-43 proteinopathy. Brain 135, 3380–3391 (2012).
Laferrière, F. et al. Quaternary structure of pathological prion protein as a determining factor of strain-specific prion replication dynamics. PLoS Pathog. 9, e1003702 (2013).
Polymenidou, M. et al. Coexistence of multiple PrPSc types in individuals with Creutzfeldt-Jakob disease. Lancet Neurol. 4, 805–814 (2005).
Carra, S. et al. Alteration of protein folding and degradation in motor neuron diseases: Implications and protective functions of small heat shock proteins. Prog. Neurobiol. 97, 83–100 (2012).
Neumann, M. et al. Absence of heterogeneous nuclear ribonucleoproteins and survival motor neuron protein in TDP-43 positive inclusions in frontotemporal lobar degeneration. Acta Neuropathol. 113, 543–548 (2007).
Kametani, F. et al. Mass spectrometric analysis of accumulated TDP-43 in amyotrophic lateral sclerosis brains. Sci. Rep. 6, 23281 (2016).
Ingre, C. et al. A novel phosphorylation site mutation in profilin 1 revealed in a large screen of US, Nordic, and German amyotrophic lateral sclerosis/frontotemporal dementia cohorts. Neurobiol. Aging 34, 1708.e1–1708.e6 (2013).
Wu, C. H. et al. Mutations in the profilin 1 gene cause familial amyotrophic lateral sclerosis. Nature 488, 499–503 (2012).
Zhou, J. et al. Spinal muscular atrophy associated with progressive myoclonic epilepsy is caused by mutations in ASAH1. Am. J. Hum. Genet. 91, 5–14 (2012).
Xu, G., Stevens, S. M. Jr., Moore, B. D., McClung, S. & Borchelt, D. R. Cytosolic proteins lose solubility as amyloid deposits in a transgenic mouse model of Alzheimer-type amyloidosis. Hum. Mol. Genet. 22, 2765–2774 (2013).
McGurk, L. et al. Poly-A binding protein-1 localization to a subset of TDP-43 inclusions in amyotrophic lateral sclerosis occurs more frequently in patients harboring an expansion in C9orf72. J. Neuropathol. Exp. Neurol. 73, 837–845 (2014).
Kerman, A. et al. Amyotrophic lateral sclerosis is a non-amyloid disease in which extensive misfolding of SOD1 is unique to the familial form. Acta Neuropathol. 119, 335–344 (2010).
Robinson, J. L. et al. TDP-43 skeins show properties of amyloid in a subset of ALS cases. Acta Neuropathol. 125, 121–131 (2013).
Lin, W. L. & Dickson, D. W. Ultrastructural localization of TDP-43 in filamentous neuronal inclusions in various neurodegenerative diseases. Acta Neuropathol. 116, 205–213 (2008).
Guenther, E. L. et al. Atomic structures of TDP-43 LCD segments and insights into reversible or pathogenic aggregation. Nat. Struct. Mol. Biol. 25, 463–471 (2018).
Aguzzi, A., Heikenwalder, M. & Polymenidou, M. Insights into prion strains and neurotoxicity. Nat. Rev. Mol. Cell Biol. 8, 552–561 (2007).
Yagi, H. et al. Zonisamide enhances neurite elongation of primary motor neurons and facilitates peripheral nerve regeneration in vitro and in a mouse model. PLoS One 10, e0142786 (2015).
Danzer, K. M., Krebs, S. K., Wolff, M., Birk, G. & Hengerer, B. Seeding induced by alpha-synuclein oligomers provides evidence for spreading of alpha-synuclein pathology. J. Neurochem. 111, 192–203 (2009).
Polymenidou, M. & Cleveland, D. W. Biological spectrum of amyotrophic lateral sclerosis prions. Cold Spring Harb. Perspect. Med. 7, a024133 (2017).
Polymenidou, M. & Cleveland, D. W. The seeds of neurodegeneration: prion-like spreading in ALS. Cell 147, 498–508 (2011).
Sanders, D. W. et al. Distinct tau prion strains propagate in cells and mice and define different tauopathies. Neuron 82, 1271–1288 (2014).
Peelaerts, W. et al. α-Synuclein strains cause distinct synucleinopathies after local and systemic administration. Nature 522, 340–344 (2015).
Meyer-Luehmann, M. et al. Exogenous induction of cerebral beta-amyloidogenesis is governed by agent and host. Science 313, 1781–1784 (2006).
Leek, J. T., Johnson, W. E., Parker, H. S., Jaffe, A. E. & Storey, J. D. The sva package for removing batch effects and other unwanted variation in high-throughput experiments. Bioinformatics 28, 882–883 (2012).
Ling, S. C. et al. ALS-associated mutations in TDP-43 increase its stability and promote TDP-43 complexes with FUS/TLS. Proc. Natl Acad. Sci. USA 107, 13318–13323 (2010).
Acknowledgements
We are grateful to all the subjects and their families for donating tissues for scientific research; this work would not have been possible without their generosity and foresightedness. We thank S. Saberi and M.J. Rodriguez from UCSD for preparing and shipping autopsy material from San Diego to Zurich; A. Käch and the Center for Microscopy and Image Analysis of the University of Zurich for help and technical support in all electron microscopy experiments; H. Leske, K. Frontzek, E. Rushing and A. Aguzzi (Institute of Neuropathology, University Hospital of Zurich) for helpful advice and discussions; J. Luedke and J. Weber for technical help; and S. Sahadevan M.K. and A. Zbinden for critical input on the manuscript. We thank M. Gstaiger (Institute of Molecular Systems Biology, ETH Zurich, Switzerland) for sharing the Flp-In T-REx HEK293 cell line and the pOG44 plasmid. This work was supported by a Swiss National Science Foundation Professorship (PP00P3_144862) and a Human Frontier Science Program Career Development Award (CDA-00058/2012) to M.P. and a UCL/ZNZ Neuroscience Collaboration Grant to M.P and A.M.I funded through UCL’s Wellcome Trust Institutional Strategic Support Fund Investing in Excellent Researchers (105604/Z/14/Z). F.L. and M.H.P. are both recipients of the Milton-Safenowitz fellowship from the ALS Association (15-IIP-208 and 16-PDF-247, respectively). F.L. received a Postdoc Award and Z.M. a Candoc Award (Forschungskredit) from the University of Zurich. T.L. is funded by an Alzheimer’s Research UK senior fellowship. Y.T.A. was supported by the Leonard Wolfson Centre for experimental neurology. A.I. is the recipient of a European Research Council consolidator grant (648716-C9ND). The Queen Square Brain Bank is supported by the Reta Lila Weston Institute for Neurological Studies and the Progressive Supranuclear Palsy (Europe) Association.
Author information
Authors and Affiliations
Contributions
F.L. developed and performed the biochemical experiments with the help of Z.M. and M.P. conceived and directed the study. M.P.-B. generated and characterized the stable cell line with inducible TDP-43-HA and performed cell toxicity experiments. M.H.-P., L.G., E-M.H., and T.A. helped with performing gradients and developing the SarkoSpin method. P.B. and P.P. performed mass spectrometry, which was bioinformatically analyzed by U.W. G.B. performed electron microscopy experiments. A.A.-A., A.L. and H.S. gave critical input on the structural analysis of extracted aggregates by electron microscopy and performed preliminary cryo-electron microscopy and correlative light-electron microscopy experiments. Autopsy material and associated clinical and neuropathological information was provided by J.R., T.L. and A.I., while S.C.F. carried out the pTDP-43 immunohistochemistry and immunofluorescence and Y.T.A. sampled the cases from the Queen Square Brain Bank. F.D.G., F.I. and E.B. performed and analyzed inoculations on primary neurons. F.L., Z.M. and M.P wrote the manuscript. All authors read, edited and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Characteristics of disease subtypes and correlation to disease duration.
a. Phosphorylated TDP-43 immunohistochemical staining of temporal cortex from FTLD-TDP subtypes, highlighting differences in their pathological hallmarks. Neuronal cytoplasmic inclusions (NCI) (arrows) and short dystrophic neurites (DN) (arrowhead) were observed in FTLD-TDP-A without (top panel) or with (middle panel) C9orf72 repeat expansions. In FTLD-TDP-C, long twisted neurites were observed (lower panel, arrowhead). Scale bar 25 μm. This experiment was repeated two independent times with similar results. b-c. Age at death (b) and disease duration (c) of patients from the different disease groups used in the study (error bars show means and SD). FTLD-TDP-C patients showed significantly longer disease duration. n = 16, 25 and 11 individual patients for ALS, FTLD-TDP-A and FTLD-TDP-C respectively. One-way ANOVAs followed by Tukey’s multiple-comparison tests gave the indicated p values.
Supplementary Figure 2 Benzonase treatment releases physiological TDP-43 complexes from SarkoSpin pellets.
a. Schematic representation of sequential insolubility assay. Brain homogenates are solubilized with indicated detergents (without Benzonase) and centrifuged after each solubilization step. Each time, pellets are resuspended in buffer with increasing detergent stringency until obtaining an SDS-insoluble fraction. b. Sequential insolubility assay fractions from control (black, left panels) and ALS (red, right panels) were subjected to SDS-PAGE followed by immunolabeling of TDP-43 (top) and hnRNPA1 (bottom). Both of these proteins are present in SDS fraction, in any health or disease case, showing the importance of using Benzonase for assessing the insolubility of nucleic acid binding proteins. c. Biochemical analysis of sarkosyl-soluble and insoluble fractions extracted from human brain samples, with or without Benzonase treatment as noted. Supernatants and pellets were subjected to SDS-PAGE followed by silver staining of total proteins (upper panel), or immunoblotted against total TDP-43 (middle panel) or phosphorylated at position S409/410 TDP-43 (pTDP-43) (lower panel). Physiological TDP-43 in the pellet of the control sample (black arrow) is released upon Benzonase treatment (white arrow). b-c Experiments were independently repeated 3 times with similar results.
Supplementary Figure 3 Immunoblots of SarkoSpin fractions from all groups of patients show consistent isolation of pTDP-43.
Brain from control (black, C), ALS (red), FTLD-TDP type A (green, F-A), type B (yellow, F-B) and type C (blue, F-C) motor cortex (a, b) and frontal cortex (c) were subjected to SarkoSpin. The supernatant and pellet fractions were analyzed by SDS-PAGE and immunoblotted against TDP-43, pTDP-43, hnRNPA1 and SOD1 antibodies. Posttranslational modifications related to pathological TDP-43 are marked with asterisks: poly-ubiquitination smear (***), hyper-phosphorylation (**) and C-terminal fragmentation (*). Ssupernatant, P – pellet, L – ladder.
Supplementary Figure 4 SarkoSpin captures native pathological TDP-43 assemblies.
Native PAGE immunoblots of SarkoSpin supernatants (a.,b.) and pellets (c.,d.) extracted from different disease types were immunoblotted against total TDP-43 (a.,c.) or pTDP-43 (pS409/410, b.,d.). The indicative molecular weights were established by NativeMark protein ladder, and material retained in the stacking gels was considered of more than 10 MDa. This experiment was independently repeated twice with similar results.
Supplementary Figure 5 SarkoSpin extracts from different ALS and FTLD samples have distinct protein compositions beyond TDP-43.
Abundance levels of selected proteins that were most consistently different in patient samples of human TDP-43 proteinopathies, FTLD-Tau (FTLD-T) and controls. We applied the idealized vector method described in the Methods section for control and pooled TDP-43 proteinopathy samples (ALS, FTLD-TDP-A, FTLD-TDP-B and FTLD-TDP-C): a vector of the same number of 0s as control samples and the same number of 1s patient samples was created. For each protein, we measured the correlation of this idealized vector to the vector of normalized and batch-corrected protein abundance values of control and patient samples. The top 50 correlating proteins were sorted based on these correlation values, which are shown as a bar plot on the right. Strong correlation values (r > 0.5) were separated from medium and weak correlation values by the red dashed line.
Supplementary Figure 6 Proteins enriched in SarkoSpin pellets of ALS brains.
Abundance levels of selected proteins that were most consistently different in ALS brain samples compared to controls. We applied the idealized vector method described in Methods section and Supplementary Figure 5 legend to control and ALS samples. The top 50 correlating proteins were sorted based on these correlation values, which are given as a bar plot on the right. Strong correlation values (r > 0.5) were separated from medium and weak correlation values by the red dashed line.
Supplementary Figure 7 Proteins enriched in SarkoSpin pellets of FTLD-TDP-A brains.
Abundance levels of selected proteins that were most consistently different in FTLD-TDP-A brain samples compared to controls. We applied the idealized vector method described in Methods section and Supplementary Figure 5 legend to control and FTLD-TDP-A samples. The top 50 correlating proteins were sorted based on these correlation values, which are given as a bar plot on the right. Strong correlation values (r > 0.5) were separated from medium and weak correlation values by the red dashed line.
Supplementary Figure 8 Proteins enriched in SarkoSpin pellets of FTLD-TDP-B brains.
Abundance levels of selected proteins that were most consistently different in FTLD-TDP-B brain samples compared to controls. We applied the idealized vector method described in Methods section and Supplementary Figure 5 legend to control and FTLD-TDP-B samples. The top 50 correlating proteins were sorted based on these correlation values, which are given as a bar plot on the right. Strong correlation values (r > 0.5) were separated from medium and weak correlation values by the red dashed line.
Supplementary Figure 9 Proteins enriched in SarkoSpin pellets of FTLD-TDP-C brains.
Abundance levels of selected proteins that were most consistently different in FTLD-TDP-C brain samples compared to controls. We applied the idealized vector method described in Methods section and Supplementary Figure 5 legend to control and FTLD-TDP-C samples. The top 50 correlating proteins were sorted based on these correlation values, which are given as a bar plot on the right. Strong correlation values (r > 0.5) were separated from medium and weak correlation values by the red dashed line.
Supplementary Figure 10 Further characterization on the protein contents in SarkoSpin soluble and insoluble fractions in different neuropathological subtypes.
a. Representative immunoblots on SarkoSpin soluble fractions corresponding to immunoblots on insoluble material in Fig. 2b. b. Fullsize immunoblots on SarkoSpin supernatant and pellet fractions probed against PFN1, showing unspecific bands above 30 kDa (**) in addition to the specific Profilin band at 15 kDa (*). c. Immunoblot on SarkoSpin pellets probed against total TDP-43 d. Correlation dot plot of pTDP-43 and total TDP-43 quantified from (c) (Pearson’s correlation method). n = 4 biologically independent human brain samples for all disease groups (ALS, FTLD-TDP-A and FTLD-TDP-C) e. Representative co-immunofluorescence of pTDP-43 and ubiquitin in control, FTLD-TDP-A and FTLD-TDP-C brain sections corresponding to merged images in Fig. 2f. Experiments shown in a.-c. and e. were repeated three independent times with similar results.
Supplementary Figure 11 Co-immunofluorescence of pTDP-43 with mass spectrometry hits, ASAH1, and TXNL1.
Representative images of co-immunofluorescence of pTDP-43 with selected mass spectrometry hits ASAH1 (a) and TXNL1 (b) in control, FTLD-TDP-A and FTLD-TDP-C post mortem brain sections. On the right side, high magnification images are shown, with examples of partial co-localization of pathological pTDP-43 aggregates with ASAH1 (1) and TXNL1 (2) in FTLD-TDP-A and with TXNL1 (3) in FTLD-TDP-C. All images are overlaid with DAPI nuclear staining in blue. These experiments were repeated three independent times with similar results.
Supplementary Figure 12 FBXO2 upregulation in astrocytes in FTLD-TDP-A patients but not FTLD-TDP-C patients.
a. Representative immunohistochemistry images showing FBXO2 immunoreactivity in control, FTLD-TDP-A and FTLD-TDP-C brain sections. b. Representative co-immunofluorescence of GFAP and FBXO2 in control, FTLD-TDP-A and FTLD-TDP-C brain sections corresponding to merged images in Fig. 2h. All images are overlaid with DAPI nuclear staining in blue. These experiments were repeated three independent times with similar results.
Supplementary Figure 13 Distinct size distributions of TDP-43 aggregates isolated from different types of TDP-43 proteinopathies.
Brain homogenates from control (black, n = 6), FTLD-Tau (grey, n = 2), ALS (red, n = 4), FTLD-TDP-A (green, n = 11), FTLD-TDP-B (yellow, n = 3) and FTLD-TDP-C (blue, n = 9) patients were subjected to SarkoSpin solubilization and velocity sedimentation by ultracentrifugation on a continuous iodixanol gradient. The collected fractions (numbered from top to bottom of the gradient) were analyzed for TDP-43 (left panel) and pTDP-43 (right panel) content by dot blot. These experiments were repeated twice independently with similar results.
Supplementary Figure 14 Immunoblots representing the distribution of TDP-43, pTDP-43, FUS, SOD1, and hnRNPA1 on velocity sedimentation gradients.
a-b. Representative immunoblots of the velocity sedimentation profiles of TDP-43 (a) and pTDP-43 (b). SarkoSpin solubilized brain homogenates from frontal and motor cortex of control (black), FTLD-TDP-A (green), and FTLD-TDP-C (blue), were fractionated by velocity and the collected fractions (numbered from top to bottom of the gradient) were analyzed for TDP-43 (a) and pTDP-43 (b) by western blotting. These experiments were repeated three independent times with similar results. c. Representative immunoblots of the distribution of FUS, hnRNPA1 and SOD1 on velocity sedimentation gradients. SarkoSpin-solubilized brain homogenates from frontal and motor cortex of control (black), ALS (red), FTLD-TDP-A (green), and FTLD-TDP-C (blue) were fractionated by velocity and the collected fractions (numbered from top to bottom of the gradient) were analyzed for FUS (top), hnRNPA1 (middle) and SOD1 (bottom) content by western blotting. These experiments were repeated four independent times with similar results.
Supplementary Figure 15 Distinct density profiles of TDP-43 aggregates isolated from different types of TDP-43 proteinopathies.
SarkoSpin-solubilized brain homogenates from control (black, n = 6), FTLD-Tau (grey, n = 2), ALS (red, n = 4), FTLD-TDP type A (green, n = 11), B (yellow, n = 3) and C (blue, n = 9) patients were fractionated by density upon isopycnic equilibrium floatation gradients. The collected fractions (numbered from top to bottom of the gradient) were analyzed for TDP-43 (a) and pTDP-43 (b) content by dot blot. The density values calculated by refractometry are indicated on the top of the gradients. These experiments were repeated twice independently with similar results.
Supplementary Figure 16 Immunoblots representing the distribution of TDP-43 and pTDP-43, and verification of the size of TDP-43 aggregates segregated by density-floatation gradients.
a-b. Representative immunoblots of the distribution of TDP-43 (a) and pTDP-43 (b) on velocity gradients. SarkoSpin-solubilized brain homogenates from frontal and motor cortex of control (black), FTLD-TDP-A (green), and FTLD-TDP-C (blue) were fractionated by density and the collected fractions (numbered from top to bottom of the gradient) were analyzed for pTDP-43 and TDP-43 content by western blotting. The experiments shown (two independent biological samples and fractionations) were repeated in total four times independently with similar results. c. Representative Native PAGE immunoblots of TDP-43 (left panels), and pTDP-43 (right panels) on density fractionations from control (black), FTLD-TDP-A (green), and FTLD-TDP-C (blue) patient brain samples. Only fractions 1 to 14 were loaded due to available wells on native PAGE gels. Density values calculated by refractometry are indicated on the top of the graph. pTDP-43-positive pathological aggregates are seen at >10 MDa for FTLD-TDP-A and FTLD-TDP-C (green and blue arrowheads respectively), whereas oligomeric TDP-43 assemblies are observed in all samples between 66 kDa and 10 MDa (black brackets). These experiments were repeated twice independently with similar results.
Supplementary Figure 17 Further structural analysis of pathological TDP-43 isolated from different disease subtypes.
a. TEM images of negative control, without the use of primary antibody, of immunolabeled FTLD-TDP-A insoluble material corresponding to TEM images from Fig. 5a-b, showing no unspecific binding of the gold-conjugated secondary antibody. Sample in panel on the left was prepared including the methylcellulose (MC) preservation step, and the sample represented on the right without the MC step. b. Upper panel: TEM image of immunolabeled FTLD-TDP-A aggregates (red arrows) with lipid contaminants (yellow arrows) surrounding the protein assemblies. Lower panel: TEM image of immunolabeled FTLD-TDP-A sample purified over sucrose cushion during SarkoSpin extraction, which efficiently removes the lipid contaminants (red arrows point to examples of gold-positive structures). a. and b. experiments were independently repeated three times with similar results. c. Representative immunoblots of SarkoSpin pellets extracted from control, ALS, FTLD-TDP-A and FTLD-TDP-C brains after proteolytic digestion. Brain homogenates were digested by different proteases for 30 minutes at 37 °C during SarkoSpin solubilization step. SarkoSpin pellets were subjected to SDS-PAGE and immunoblotted against total TDP-43 (top panels) and pTDP-43 (bottom panels). N – not treated; P – treated with 0.5 mg/ml Proteinase K; T – treated with 0.1 mg/ml Trypsin; C – treated with 0.01 mg/ml Chymotrypsin. d. Full size representation of the immunoblots on Chymotrypsinized and Trypsinized SarkoSpin pellets shown in Fig. 5c. c. and d. experiments were repeated twice independently with similar results.
Supplementary Figure 18 Inoculation of GFP-pTDP-43-HA pellets in an inducible TDP-43-HA HEK 293 cell line, and verification of TDP-43, pTDP-43, and total protein amounts inoculated to primary cortical neurons.
a. Schematic representation of the pcDNA5 constructs used to generate the stable HEK 293 cell line and pellets containing GFP-pTDP-43-HA. b-c. Sensitivity of the inducible system was tested by Western blot (b) and immunofluorescence (c) with an HA-tag specific antibody. d. SarkoSpin-isolated TDP-43 aggregates in NSC-34 cells transiently transfected with the GFP-TDP-43-HA construct for 48 hours. e. TDP-43-HA-expressing HEK 293 cells internalized lipofected SarkoSpin-insoluble GFP-TDP-43-HA. 4 hours after inoculation, cells were trypsinized and collected by centrifugation before lysis in order to remove all GFP-TDP-43-HA aggregates adhered to the cell surface and only visualize those that have truly entered the cells. L2000: Lipofectamine 2000. f. Immunocytochemistry of TDP-43-HAexpressing cells lipofected with GFP-TDP-43-HA aggregates. Side views of the z-stack (yz plane) are depicted in 1 and 2, show examples of internalized GFP-TDP-43-HA aggregates. g. Immunoblots on homogenates for SarkoSpin from TDP-43-HA HEK 293 cells inoculated with SarkoSpin pellets from control, FTLD-TDP-A or FTLD-TDP-C patients. h. Full-size immunoblots of SarkoSpin supernatant and pellet fractions from TDP-43-HA expressing cells inoculated with SarkoSpin pellets from control, FTLD-TDP-A or FTLD-TDP-C patients, corresponding to Fig. 6c. i. Each primary neuron inoculum was loaded on dot blot and immunolabeled with antibodies against TDP-43 (top), pTDP-43 pS409/410 (middle) and actin (bottom). Experiments were repeated independently three times with similar results. b.-i. Experiments were repeated three independent times with similar results.
Supplementary information
Supplementary Table 1
Summary of patient information of the autopsy material used in this study.
Supplementary Table 2
List of antibodies used in this study, and related information.
Rights and permissions
About this article
Cite this article
Laferrière, F., Maniecka, Z., Pérez-Berlanga, M. et al. TDP-43 extracted from frontotemporal lobar degeneration subject brains displays distinct aggregate assemblies and neurotoxic effects reflecting disease progression rates. Nat Neurosci 22, 65–77 (2019). https://doi.org/10.1038/s41593-018-0294-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-018-0294-y