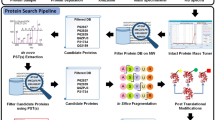
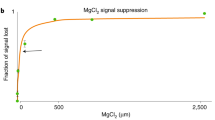
Abstract
The combination of native electrospray ionization with top-down fragmentation in mass spectrometry (MS) allows simultaneous determination of the stoichiometry of noncovalent complexes and identification of their component proteoforms and cofactors. Although this approach is powerful, both native MS and top-down MS are not yet well standardized, and only a limited number of laboratories regularly carry out this type of research. To address this challenge, the Consortium for Top-Down Proteomics initiated a study to develop and test protocols for native MS combined with top-down fragmentation of proteins and protein complexes across 11 instruments in nine laboratories. Here we report the summary of the outcomes to provide robust benchmarks and a valuable entry point for the scientific community.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The raw MS data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD047341 (refs. 72,73). The protein sequences can be accessed by the respective identifier (Supplementary Table 4) via https://www.uniprot.org/.
References
Smith, L. M., Kelleher, N. L. & The Consortium for Top Down Proteomics Proteoform: a single term describing protein complexity. Nat. Methods 10, 186–187 (2013).
Aebersold, R. et al. How many human proteoforms are there? Nat. Chem. Biol. 14, 206–214 (2018).
Smith, L. M. & Kelleher, N. L. Proteoforms as the next proteomics currency. Science 359, 1106–1107 (2018).
Dang, X. et al. The first pilot project of the consortium for top-down proteomics: a status report. Proteomics 14, 1130–1140 (2014).
Chen, B., Brown, K. A., Lin, Z. & Ge, Y. Top-down proteomics: ready for prime time? Anal. Chem. 90, 110–127 (2018).
Donnelly, D. P. et al. Best practices and benchmarks for intact protein analysis for top-down mass spectrometry. Nat. Methods 16, 587–594 (2019).
Srzentic, K. et al. Interlaboratory study for characterizing monoclonal antibodies by top-down and middle-down mass spectrometry. J. Am. Soc. Mass Spectrom. 31, 1783–1802 (2020).
Habeck, T. & Lermyte, F. Seeing the complete picture: proteins in top-down mass spectrometry. Essays Biochem. 67, 283–300 (2023).
Brown, K. A., Melby, J. A., Roberts, D. S. & Ge, Y. Top-down proteomics: challenges, innovations, and applications in basic and clinical research. Expert Rev. Proteom. 17, 719–733 (2020).
Smith, L. M. et al. The Human Proteoform Project: defining the human proteome. Sci. Adv. 7, eabk0734 (2021).
Leney, A. C. & Heck, A. J. Native mass spectrometry: what is in the name? J. Am. Soc. Mass Spectrom. 28, 5–13 (2017).
Robinson, C. V. Mass spectrometry: from plasma proteins to mitochondrial membranes. Proc. Natl Acad. Sci. USA 116, 2814–2820 (2019).
Tamara, S., den Boer, M. A. & Heck, A. J. R. High-resolution native mass spectrometry. Chem. Rev. 122, 7269–7326 (2022).
Bennett, J. L., Nguyen, G. T. H. & Donald, W. A. Protein-small molecule interactions in native mass spectrometry. Chem. Rev. 122, 7327–7385 (2022).
Rogawski, R. & Sharon, M. Characterizing endogenous protein complexes with biological mass spectrometry. Chem. Rev. 122, 7386–7414 (2022).
Skinner, O. S. et al. Top-down characterization of endogenous protein complexes with native proteomics. Nat. Chem. Biol. 14, 36–41 (2018).
Ro, S. Y. et al. Native top-down mass spectrometry provides insights into the copper centers of membrane-bound methane monooxygenase. Nat. Commun. 10, 2675 (2019).
Gault, J. et al. Combining native and ‘omics’ mass spectrometry to identify endogenous ligands bound to membrane proteins. Nat. Methods 17, 505–508 (2020).
Vimer, S. et al. Comparative structural analysis of 20S proteasome ortholog protein complexes by native mass spectrometry. ACS Cent. Sci. 6, 573–588 (2020).
Schachner, L. F. et al. Decoding the protein composition of whole nucleosomes with Nuc-MS. Nat. Methods 18, 303–308 (2021).
Melani, R. D. et al. Mapping proteoforms and protein complexes from king cobra venom using both denaturing and native top-down proteomics. Mol. Cell Proteom. 15, 2423–2434 (2016).
Lermyte, F., Tsybin, Y. O., O’Connor, P. B. & Loo, J. A. Top or middle? Up or down? Toward a standard lexicon for protein top-down and allied mass spectrometry approaches. J. Am. Soc. Mass Spectrom. 30, 1149–1157 (2019).
Zhou, M. et al. Higher-order structural characterisation of native proteins and complexes by top-down mass spectrometry. Chem. Sci. 11, 12918–12936 (2020).
VanAernum, Z. L. et al. Rapid online buffer exchange for screening of proteins, protein complexes and cell lysates by native mass spectrometry. Nat. Protoc. 15, 1132–1157 (2020).
McCabe, J. W. et al. Implementing digital-waveform technology for extended m/z range operation on a native dual-quadrupole FT-IM-orbitrap mass spectrometer. J. Am. Soc. Mass Spectrom. 32, 2812–2820 (2021).
LeDuc, R. D. et al. ProForma: a standard proteoform notation. J. Proteome Res 17, 1321–1325 (2018).
Schachner, L. F. et al. Standard proteoforms and their complexes for native mass spectrometry. J. Am. Soc. Mass Spectrom. 30, 1190–1198 (2019).
Smith, L. M. et al. A five-level classification system for proteoform identifications. Nat. Methods 16, 939–940 (2019).
Iacobucci, C. et al. First community-wide, comparative cross-linking mass spectrometry study. Anal. Chem. 91, 6953–6961 (2019).
Masson, G. R. et al. Recommendations for performing, interpreting and reporting hydrogen deuterium exchange mass spectrometry (HDX-MS) experiments. Nat. Methods 16, 595–602 (2019).
Allison, T. M. et al. Software requirements for the analysis and interpretation of native ion mobility mass spectrometry data. Anal. Chem. 92, 10881–10890 (2020).
Allison, T. M. et al. Computational strategies and challenges for using native ion mobility mass spectrometry in biophysics and structural biology. Anal. Chem. 92, 10872–10880 (2020).
Gabelica, V. et al. Recommendations for reporting ion mobility mass spectrometry measurements. Mass Spectrom. Rev. 38, 291–320 (2019).
Konijnenberg, A., Butterer, A. & Sobott, F. Native ion mobility-mass spectrometry and related methods in structural biology. Biochim. Biophys. Acta 1834, 1239–1256 (2013).
Konermann, L., Ahadi, E., Rodriguez, A. D. & Vahidi, S. Unraveling the mechanism of electrospray ionization. Anal. Chem. 85, 2–9 (2013).
Hall, Z., Politis, A., Bush, M. F., Smith, L. J. & Robinson, C. V. Charge-state dependent compaction and dissociation of protein complexes: insights from ion mobility and molecular dynamics. J. Am. Chem. Soc. 134, 3429–3438 (2012).
Rolland, A. D., Biberic, L. S. & Prell, J. S. Investigation of charge-state-dependent compaction of protein ions with native ion mobility-mass spectrometry and theory. J. Am. Soc. Mass Spectrom. 33, 369–381 (2022).
Sobott, F., Hernandez, H., McCammon, M. G., Tito, M. A. & Robinson, C. V. A tandem mass spectrometer for improved transmission and analysis of large macromolecular assemblies. Anal. Chem. 74, 1402–1407 (2002).
Sobott, F., McCammon, M. G., Hernandez, H. & Robinson, C. V. The flight of macromolecular complexes in a mass spectrometer. Philos. Trans. A Math. Phys. Eng. Sci. 363, 379–389 (2005).
Rose, R. J., Damoc, E., Denisov, E., Makarov, A. & Heck, A. J. High-sensitivity Orbitrap mass analysis of intact macromolecular assemblies. Nat. Methods 9, 1084–1086 (2012).
van de Waterbeemd, M. et al. High-fidelity mass analysis unveils heterogeneity in intact ribosomal particles. Nat. Methods 14, 283–286 (2017).
Fort, K. L. et al. Expanding the structural analysis capabilities on an Orbitrap-based mass spectrometer for large macromolecular complexes. Analyst 143, 100–105 (2017).
McGee, J. P. et al. Voltage rollercoaster filtering of low-mass contaminants during native protein analysis. J. Am. Soc. Mass Spectrom. 31, 763–767 (2020).
Snijder, J., Rose, R. J., Veesler, D., Johnson, J. E. & Heck, A. J. Studying 18 MDa virus assemblies with native mass spectrometry. Angew. Chem. Int. Ed. Engl. 52, 4020–4023 (2013).
Behnke, J. S. & Urner, L. H. Emergence of mass spectrometry detergents for membrane proteomics. Anal. Bioanal. Chem. 415, 3897–3909 (2023).
Borysik, A. J., Hewitt, D. J. & Robinson, C. V. Detergent release prolongs the lifetime of native-like membrane protein conformations in the gas-phase. J. Am. Chem. Soc. 135, 6078–6083 (2013).
Reading, E. et al. The role of the detergent micelle in preserving the structure of membrane proteins in the gas phase. Angew. Chem. Int. Ed. Engl. 54, 4577–4581 (2015).
Ives, A. N. et al. Using 10,000 fragment ions to inform scoring in native top-down proteomics. J. Am. Soc. Mass Spectrom. 31, 1398–1409 (2020).
Lantz, C. et al. Native top-down mass spectrometry with collisionally activated dissociation yields higher-order structure information for protein complexes. J. Am. Chem. Soc. 144, 21826–21830 (2022).
Paizs, B. & Suhai, S. Fragmentation pathways of protonated peptides. Mass Spectrom. Rev. 24, 508–548 (2005).
Skinner, O. S. et al. Fragmentation of integral membrane proteins in the gas phase. Anal. Chem. 86, 4627–4634 (2014).
Horn, D. M., Zubarev, R. A. & McLafferty, F. W. Automated reduction and interpretation of high resolution electrospray mass spectra of large molecules. J. Am. Soc. Mass Spectrom. 11, 320–332 (2000).
Zamdborg, L. et al. ProSight PTM 2.0: improved protein identification and characterization for top down mass spectrometry. Nucleic Acids Res. 35, W701–W706 (2007).
Mayampurath, A. M. et al. DeconMSn: a software tool for accurate parent ion monoisotopic mass determination for tandem mass spectra. Bioinformatics 24, 1021–1023 (2008).
Li, L. & Tian, Z. Interpreting raw biological mass spectra using isotopic mass-to-charge ratio and envelope fingerprinting. Rapid Commun. Mass Spectrom. 27, 1267–1277 (2013).
Liu, X. et al. Deconvolution and database search of complex tandem mass spectra of intact proteins: a combinatorial approach. Mol. Cell Proteom. 9, 2772–2782 (2010).
Compton, P. D., Zamdborg, L., Thomas, P. M. & Kelleher, N. L. On the scalability and requirements of whole protein mass spectrometry. Anal. Chem. 83, 6868–6874 (2011).
Guner, H. et al. MASH Suite: a user-friendly and versatile software interface for high-resolution mass spectrometry data interpretation and visualization. J. Am. Soc. Mass Spectrom. 25, 464–470 (2014).
Cai, W. et al. MASH Suite Pro: a comprehensive software tool for top-down proteomics. Mol. Cell Proteom. 15, 703–714 (2016).
Wu, Z. et al. MASH Explorer: a universal software environment for top-down proteomics. J. Proteome Res 19, 3867–3876 (2020).
McIlwain, S. J. et al. Enhancing top-down proteomics data analysis by combining deconvolution results through a machine learning strategy. J. Am. Soc. Mass Spectrom. 31, 1104–1113 (2020).
Larson, E. J. et al. MASH Native: a unified solution for native top-down proteomics data processing. Bioinformatics 39, btad359 (2023).
Kafader, J. O. et al. Multiplexed mass spectrometry of individual ions improves measurement of proteoforms and their complexes. Nat. Methods 17, 391–394 (2020).
Kafader, J. O. et al. Individual ion mass spectrometry enhances the sensitivity and sequence coverage of top-down mass spectrometry. J. Proteome Res. 19, 1346–1350 (2020).
Worner, T. P. et al. Resolving heterogeneous macromolecular assemblies by Orbitrap-based single-particle charge detection mass spectrometry. Nat. Methods 17, 395–398 (2020).
McGee, J. P. et al. Isotopic resolution of protein complexes up to 466 kDa using individual ion mass spectrometry. Anal. Chem. 93, 2723–2727 (2021).
Marty, M. T. et al. Bayesian deconvolution of mass and ion mobility spectra: from binary interactions to polydisperse ensembles. Anal. Chem. 87, 4370–4376 (2015).
Reid, D. J. et al. MetaUniDec: high-throughput deconvolution of native mass spectra. J. Am. Soc. Mass Spectrom. 30, 118–127 (2019).
Marty, M. T. A universal score for deconvolution of intact protein and native electrospray mass spectra. Anal. Chem. 92, 4395–4401 (2020).
Park, J. et al. Informed-proteomics: open-source software package for top-down proteomics. Nat. Methods 14, 909–914 (2017).
Zhou, M., Pasa-Tolic, L. & Stenoien, D. L. Profiling of histone post-translational modifications in mouse brain with high-resolution top-down mass spectrometry. J. Proteome Res 16, 599–608 (2017).
Deutsch, E. W. et al. The ProteomeXchange consortium in 2020: enabling ‘big data’ approaches in proteomics. Nucleic Acids Res. 48, D1145–D1152 (2020).
Perez-Riverol, Y. et al. The PRIDE database resources in 2022: a hub for mass spectrometry-based proteomics evidences. Nucleic Acids Res. 50, D543–D552 (2022).
Acknowledgements
We are grateful to all members of the Board of Directors of the CTDP for valuable discussions. We also thank B. Krichel and S. McIlwain (University of Wisconsin-Madison) and C. Hake (TU Darmstadt) for assistance with database searching. The CTDP, a 501c3 nonprofit corporation, is grateful for the generous support from Seer, ThermoFisher Scientific, Bruker, SCIEX, Lilly, Pfizer, Newomics and Agilent. M.T.M. thanks A. Makarov and K. Fort at ThermoFisher Scientific for support on the UHMR Q Exactive HF instrument. This work was supported by National Institutes of Health (NIH)/National Institute of General Medical Sciences (NIGMS) grant R35 GM128624 to M.T.M, NIH grant R01 GM125085 and S10 OD018475 to Y.G. and NIH/NIGMS grant P41 GM108569 to N.L.K., as well as NIH grant R01GM103479, R35GM145286, S10RR028893 and US Department of Energy grant DEFC02-02ER63421 to J.A.L. We acknowledge project award (10.46936/intm.proj.2019.51141/60006696) from the Environmental Molecular Sciences Laboratory, a Department of Energy Office of Science User Facility sponsored by the Biological and Environmental Research program under Contract No. DE-AC05-76RL01830 to M.Z. and J.W.W. and the Ruth L. Kirschstein National Research Service Award Program at NIH (GM007185) to C.L. We are grateful for funding by the Hessian Ministry for Science and Arts via the LOEWE project ‘TRABITA’ and by the Deutsche Forschungsgemeinschaft (grant nos. 461372424 and 524226614) to F.L. The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
F.L., M.Z., B.D.S., J.A.L, C.L. and K.A.B. conceptualized the study. F.L., M.Z., B.D.S., C.L., K.A.B., Y.G., L.P.-T., W.S., J.A.L., J.N.A., N.L.K., M.T.M., H.L. and P.O.D. developed the methodology. T.H., K.A.B., B.D.S., C.L., M.Z., M.d.A.H., N.A., W.J., J.E.K., M.V., J.W.W. and Y.Y. performed the investigations. F.L. and T.H. performed formal analysis. F.L. and T.H. wrote the original draft of the manuscript. F.L., T.H., Y.G., L.P.-T., W.S., J.A.L., J.N.A., N.L.K., M.T.M., H.L. and P.O.D. contributed to the writing, review and editing of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
N.L.K. is involved in entrepreneurial activities in top-down proteomics and consults for ThermoFisher Scientific. P.O.D. is the founder and principal of Eastwoods Consulting, providing business advisory services to life science companies. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Methods thanks Alexander Leitner and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available. Primary Handling Editor: Arunima Singh, in collaboration with the Nature Methods team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Supplementary information
Supplementary Information
Supplementary Figs. 1–11, Tables 1–7, Protocol 1 and Survey 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Habeck, T., Brown, K.A., Des Soye, B. et al. Top-down mass spectrometry of native proteoforms and their complexes: a community study. Nat Methods (2024). https://doi.org/10.1038/s41592-024-02279-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41592-024-02279-6