Abstract
Chromatin remodellers include diverse enzymes with distinct biological functions, but nucleosome-sliding activity appears to be a common theme1,2. Among the remodelling enzymes, Snf2 serves as the prototype to study the action of this protein family. Snf2 and related enzymes share two conserved RecA-like lobes3, which by themselves are able to couple ATP hydrolysis to chromatin remodelling. The mechanism by which these enzymes couple ATP hydrolysis to translocate the nucleosome along the DNA remains unclear2,4,5,6,7,8. Here we report the structures of Saccharomyces cerevisiae Snf2 bound to the nucleosome in the presence of ADP and ADP-BeFx. Snf2 in the ADP-bound state adopts an open conformation similar to that in the apo state, and induces a one-base-pair DNA bulge at superhelix location 2 (SHL2), with the tracking strand showing greater distortion than the guide strand. The DNA distortion propagates to the proximal end, leading to staggered translocation of the two strands. The binding of ADP-BeFx triggers a closed conformation of the enzyme, resetting the nucleosome to a relaxed state. Snf2 shows altered interactions with the DNA in different nucleotide states, providing the structural basis for DNA translocation. Together, our findings suggest a fundamental mechanism for the DNA translocation that underlies chromatin remodelling.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Atomic coordinates and electron microscopy maps are deposited in the Electron Microscopy Data Bank (EMDB) and Protein Data Bank (PDB) under accession codes EMD-6883, PBD code 5Z3V (‘601’, ADP-BeFx, SHL-2 complex); EMD-6882, PDB code 5Z3U (“601”, ADP-BeFx, SHL2 complex); EMD-6880, PBD code 5Z3O (“601”, ADP, SHL2 complex); EMD-6879, PBD code 5Z3L (‘601’, apo, SHL2 complex); EMD-9748, PBD code 6IY2 (‘MMTV’, ADP, SHL2 complex); and EMD-9749, PBD code 6IY3 (‘MMTV’, ADP, SHL−2 complex).
References
Clapier, C. R. & Cairns, B. R. The biology of chromatin remodeling complexes. Annu. Rev. Biochem. 78, 273–304 (2009).
Clapier, C. R., Iwasa, J., Cairns, B. R. & Peterson, C. L. Mechanisms of action and regulation of ATP-dependent chromatin-remodelling complexes. Nat. Rev. Mol. Cell Biol. 18, 407–422 (2017).
Xia, X., Liu, X., Li, T., Fang, X. & Chen, Z. Structure of chromatin remodeler Swi2/Snf2 in the resting state. Nat. Struct. Mol. Biol. 23, 722–729 (2016).
Becker, P. B. & Hörz, W. ATP-dependent nucleosome remodeling. Annu. Rev. Biochem. 71, 247–273 (2002).
Flaus, A. & Owen-Hughes, T. Mechanisms for nucleosome mobilization. Biopolymers 68, 563–578 (2003).
Saha, A., Wittmeyer, J. & Cairns, B. R. Chromatin remodelling: the industrial revolution of DNA around histones. Nat. Rev. Mol. Cell Biol. 7, 437–447 (2006).
Bowman, G. D. Mechanisms of ATP-dependent nucleosome sliding. Curr. Opin. Struct. Biol. 20, 73–81 (2010).
Narlikar, G. J., Sundaramoorthy, R. & Owen-Hughes, T. Mechanisms and functions of ATP-dependent chromatin-remodeling enzymes. Cell 154, 490–503 (2013).
Liu, X., Li, M., Xia, X., Li, X. & Chen, Z. Mechanism of chromatin remodelling revealed by the Snf2-nucleosome structure. Nature 544, 440–445 (2017).
Zofall, M., Persinger, J., Kassabov, S. R. & Bartholomew, B. Chromatin remodeling by ISW2 and SWI/SNF requires DNA translocation inside the nucleosome. Nat. Struct. Mol. Biol. 13, 339–346 (2006).
Saha, A., Wittmeyer, J. & Cairns, B. R. Chromatin remodeling through directional DNA translocation from an internal nucleosomal site. Nat. Struct. Mol. Biol. 12, 747–755 (2005).
Luger, K., Mäder, A. W., Richmond, R. K., Sargent, D. F. & Richmond, T. J. Crystal structure of the nucleosome core particle at 2.8 Å resolution. Nature 389, 251–260 (1997).
Makde, R. D., England, J. R., Yennawar, H. P. & Tan, S. Structure of RCC1 chromatin factor bound to the nucleosome core particle. Nature 467, 562–566 (2010).
Richmond, T. J. & Davey, C. A. The structure of DNA in the nucleosome core. Nature 423, 145–150 (2003).
Winger, J., Nodelman, I. M., Levendosky, R. F. & Bowman, G. D. A twist defect mechanism for ATP-dependent translocation of nucleosomal DNA. eLife 7, e34100 (2018).
Suto, R. K. et al. Crystal structures of nucleosome core particles in complex with minor groove DNA-binding ligands. J. Mol. Biol. 326, 371–380 (2003).
Deindl, S. et al. ISWI remodelers slide nucleosomes with coordinated multi-base-pair entry steps and single-base-pair exit steps. Cell 152, 442–452 (2013).
Harada, B. T. et al. Stepwise nucleosome translocation by RSC remodeling complexes. eLife 5, e10051 (2016).
Yan, L., Wu, H., Li, X., Gao, N. & Chen, Z. Structure of the ISWI-nucleosome complex reveals a conserved mechanism of chromatin remodeling. Nat. Struct. Mol. Biol. (in press).
Frouws, T. D., Duda, S. C. & Richmond, T. J. X-ray structure of the MMTV-A nucleosome core. Proc. Natl Acad. Sci. USA 113, 1214–1219 (2016).
Ngo, T. T., Zhang, Q., Zhou, R., Yodh, J. G. & Ha, T. Asymmetric unwrapping of nucleosomes under tension directed by DNA local flexibility. Cell 160, 1135–1144 (2015).
Tan, S. & Davey, C. A. Nucleosome structural studies. Curr. Opin. Struct. Biol. 21, 128–136 (2011).
Hu, M. et al. A particle-filter framework for robust cryoEM 3D reconstruction. Nat. Methods 15, 1083–1089 (2018).
Farnung, L., Vos, S. M., Wigge, C. & Cramer, P. Nucleosome–Chd1 structure and implications for chromatin remodelling. Nature 550, 539–542 (2017).
Nodelman, I. M. et al. Interdomain communication of the chd1 chromatin remodeler across the DNA gyres of the nucleosome. Mol. Cell 65, 447–459.e446 (2017).
Hall, M. A. et al. High-resolution dynamic mapping of histone–DNA interactions in a nucleosome. Nat. Struct. Mol. Biol. 16, 124–129 (2009).
Lorch, Y., Maier-Davis, B. & Kornberg, R. D. Mechanism of chromatin remodeling. Proc. Natl Acad. Sci. USA 107, 3458–3462 (2010).
Gangaraju, V. K., Prasad, P., Srour, A., Kagalwala, M. N. & Bartholomew, B. Conformational changes associated with template commitment in ATP-dependent chromatin remodeling by ISW2. Mol. Cell 35, 58–69 (2009).
Li, X., Zheng, S., Agard, D. A. & Cheng, Y. Asynchronous data acquisition and on-the-fly analysis of dose fractionated cryoEM images by UCSFImage. J. Struct. Biol. 192, 174–178 (2015).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Zhang, K. Gctf: Real-time CTF determination and correction. J. Struct. Biol. 193, 1–12 (2016).
Bharat, T. A. M., Russo, C. J., Löwe, J., Passmore, L. A. & Scheres, S. H. W. Advances in single-particle electron cryomicroscopy structure determination applied to sub-tomogram averaging. Structure 23, 1743–1753 (2015).
Yan, C., Wan, R., Bai, R., Huang, G. & Shi, Y. Structure of a yeast activated spliceosome at 3.5 Å resolution. Science 353, 904–911 (2016).
Scheres, S. H. & Chen, S. Prevention of overfitting in cryo-EM structure determination. Nat. Methods 9, 853–854 (2012).
Afonine, P. V. et al. Towards automated crystallographic structure refinement with phenix.refine. Acta Crystallogr. D 68, 352–367 (2012).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Hwang, W. L., Deindl, S., Harada, B. T. & Zhuang, X. Histone H4 tail mediates allosteric regulation of nucleosome remodelling by linker DNA. Nature 512, 213–217 (2014).
Kerssemakers, J. W. et al. Assembly dynamics of microtubules at molecular resolution. Nature 442, 709–712 (2006).
Lin, W. et al. Helicase stepping investigated with one-nucleotide resolution fluorescence resonance energy transfer. Phys. Rev. Lett. 119, 138102 (2017).
Gu, M. & Rice, C. M. Three conformational snapshots of the hepatitis C virus NS3 helicase reveal a ratchet translocation mechanism. Proc. Natl Acad. Sci. USA 107, 521–528 (2010).
Zheng, G., Lu, X. J. & Olson, W. K. Web 3DNA—a web server for the analysis, reconstruction, and visualization of three-dimensional nucleic-acid structures. Nucleic Acids Res. 37, W240–W246 (2009).
Acknowledgements
We thank J. Lei and the staff at the Tsinghua University Branch of China National Center for Protein Sciences Beijing for providing facility support. This work was supported by the Chinese MOST (2017YFA0102900), the National Natural Science Foundation of China (31825016, 31570731, 31630046) to Z.C. and (11834018) to Ming Li.
Reviewer information
Nature thanks Beat Fierz and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
Meijing Li collected the EM data with help from X. Liu and X.X.; X. Liu and X.X. prepared the proteins and performed the biochemical analyses; Meijing Li and X. Li performed the EM analysis; Y.T., Q.J., Y.L., Z.C. and Ming Li performed the smFRET analysis; Z.C. wrote the manuscript with help from all authors; Z.C. directed and supervised the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
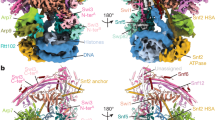
Extended Data Fig. 1 Cryo-EM structure analysis of the Snf2–nucleosome complex in the ADP-bound state.
a, Representative micrograph of cryo-EM sample. b, Fast Fourier transform (FFT) of image in a with the Thon rings extending to approximately 3.5 Å. c, 2D class averages of characteristic projection views of cryo-EM particles, Snf2–NCP complexes marked with red box were selected for further processing. d, Flow chart of cryo-EM data processing. The linker DNA is visible at low resolution during the 3D classification steps, and becomes invisible at high resolution after auto-refinements. e, The gold-standard FSC curves calculated between two halves of datasets for the Snf2–nucleosome complex in different states. f, Angular distribution of particle projections of the SHL2 complex in the ADP-bound state.
Extended Data Fig. 2 Local density maps of the Snf2–nucleosome complex in the ADP-bound state.
a–g, Electron density maps of DNA (more views in Supplementary Video 1) (a); Snf2 (b); ADP-binding pocket (c); Arg1142 (d); Ser1162, Arg164 (motif V) and Trp1185 (e); α13 and Thr1113 (motif IV) (f); and Lys900 and Lys905 (motif II) (g). h, Comparison of the map-fitting quality of the models with the 5′-tracking strand DNA pulled from the proximal end (red) and from the distal end (grey). FSCs were calculated between the corresponding models and the segmented DNA density inside the nucleosome. Dashed line denotes FSC = 0.143. A representative region near the dyad is shown on the right. The hypothetical model with DNA translocation from the distal end with led to a lower FSC between the model and the segmented DNA density, with the bases at positions 72 and 70 not fitting well into the map.
Extended Data Fig. 3 Flow chart of cryo-EM data processing of the Snf2–nucleosome complex in the ADP-BeFx-bound state.
a, Representative micrograph of cryo-EM sample. b, FFT of image in a with the Thon rings extending to approximately 3.5 Å. c, 2D class averages of characteristic projection views of cryo-EM particles. Snf2–NCP complexes marked with a red box were selected for further processing. d, Flow chart of cryo-EM data processing.
Extended Data Fig. 4 Local density maps of the Snf2–nucleosome complex in the ADP-BeFx-bound state.
a, b, Density maps of the nucleosome with ADP-BeFx-bound Snf2 at SHL2 (a) and SHL−2 (b). The boxed regions are enlarged for further analyses on the right. c, Comparison of the structures of the SHL2 (colour-coded) and SHL−2 (grey) complexes. Structures of the nucleosome are shown on the right. d, Density map of Snf2 in the ADP-BeFx-bound state. The brace-II and post-HSA (helicase-SANT-associated) helices show weak cryo-EM density. e, Density map of the catalytic pocket of Snf2 in the ADP-BeFx-bound state. BeFx is modelled by a BeF3 molecule (magenta). f, Structural comparison of the ATP-binding pocket of Snf2 (colour-coded) and NS3 helicase (grey, PDB code 3KQU40). g–j, Local density maps near Arg1142 (g), Arg1164 and Trp1185 (h), α13/Thr1113 (i), and motif II centring on Lys900 (j).
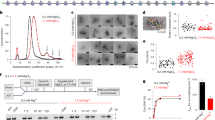
Extended Data Fig. 5 smFRET analyses of the DNA translocation induced by Snf2.
a, Labelling scheme. Nucleosomes are coloured as in Fig. 2. The boxed region is enlarged for further analysed in b. SHL2 and SHL−2 are defined based on the position relative to the fluorescent probes. The red and black arrows indicate the direction of DNA movement by Snf2 working at SHL2 and SHL−2, respectively. The distances between the Cy3 probe (at R71C of H2A) and the Cy5 probe placed at different positions are shown in the parentheses. c, d, Representative trace curves showing the Cy3 intensity (green), Cy5 intensity (red), the FRET value (left bottom) and distribution (right) of the free nucleosome with tracking strand (position 33, c) and guide strand (position −30, d) labelling. e–p, Representative trace curves showing the Cy3 intensity (green), Cy5 intensity (red), and the FRET value (black) of the nucleosome bound by Snf2 in the presence of ADP (e, f), ATP (g, h, the fluctuation of the FRET value was probably due to bidirectional DNA translocation resulting from the alternating remodelling by Snf2 at SHL2 and SHL−2), apo (i, j), ADP-BeFx (k, l), and adding Snf2 and ADP-BeFx sequentially (m–p). Left columns (c, e, g, i, k, m and o), tracking-strand labelling; right columns (d, f, h, j, l, n and p), guide-strand labelling. q, FRET peak values with the Cy5 probe placed at different positions of the free nucleosome.
Extended Data Fig. 6 Cryo-EM structure analysis of the MMTV-A nucleosome bound by Snf2 in the ADP-bound state.
a, Representative micrograph of cryo-EM sample. b, FFT of image in a with the Thon rings extending to approximately 3.5 Å. c, 2D class averages of characteristic projection views of cryo-EM particles, Snf2–MMTV-A NCP complexes marked with red box were selected for further processing. d, Flow chart of cryo-EM data processing. e, The gold-standard FSC curves calculated between two halves of datasets.
Extended Data Fig. 7 Local density maps of the MMTV-A nucleosome bound by Snf2 in the ADP-bound state.
a, Density maps of the whole SHL2 complex (left) and the MMTV-A nucleosome bound by Snf2 (right). The boxed region (SHL2) is enlarged for further analyses in b. b, Local density maps at the regions from positions 49–58 of the SHL2 complex. c, Density maps of the SHL−2 complex. The boxed region (SHL−2) is enlarged for further analyses at the bottom. d, Structural comparison of the Snf2–nucleosome complexes in the ADP-bound state. The ‘601’ nucleosome complex (SHL2) is colour coded. MMTV-A complex (SHL2) is in dark grey; MMTV-A complex (SHL-2) is in light grey. e, Local base-pair step parameters of the MMTV-A complex at SHL2. Calculation was performed with w3DNA41. f, Structural comparison of the DNA at SHL2. Free MMTV-A nucleosome (PDB code 5F99)20, cyan; Snf2-bound MMTV-A (colour-coded) and ‘601’ (grey) nucleosomes.
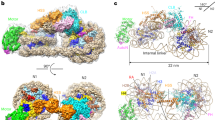
Extended Data Fig. 8 Cryo-EM structure of the Snf2–nucleosome complex in the apo state.
a, 4.31-Å-resolution cryo-EM density map (calculated with RELION) of the apo complex superimposed with the structures of Snf2 and nucleosome. Local cryo-EM density at SHL2 is shown in the middle, and local resolution analysis of the map on the right. b, 4.22-Å-resolution cryo-EM density map (calculated with THUNDER). Local cryo-EM density at SHL2 is shown in the middle, and local resolution analysis of the map on the right. c, Superposition of the models refined according to RELION (grey) and THUNDER (green). The two structures are essentially identical. d, Structural alignment of Snf2 in ADP (grey) and apo states (PDB code 5X0Y9). The apo structure is colour coded: core 1, green; core 2, cyan; brace helices, red; suppressor helix (SuppH), yellow. The nucleosomal DNA bound by the lobe 1–lobe 2 cleft is schematically illustrated as an oval. e, Structural alignment of the nucleosomal DNA bound by Snf2 in ADP (grey) and apo (colour coded) states. The Snf2 binding location is highlighted by the dotted square.
Extended Data Fig. 9 Snf2–nucleosomal DNA interactions.
a, Domain architecture of Snf2, coloured as before9. b, Comparison of the catalytic pocket of Snf2 in the ADP-bound (grey) and ADP-BeFx-bound (colour-coded) states. The bound BeFx is modelled as BeF3 (magenta). Blue arrow, movement of lobe 2 on ADP-BeFx binding. c, Comparison of the nucleosomal DNA bound by Snf2 in the presence of ADP-BeFx (colour-coded) and the unbound state (PDB code 3MVD, grey)13. The boxed region is enlarged for further analysis on the right. d, e, Details of the Snf2–DNA interactions in the ADP-BeFx-bound (d) and ADP-bound (e) states. Right, schematic of the interactions. f, Position of the gating helix (α13, magenta) in the ADP-bound complex.
Extended Data Fig. 10 Simplified remodelling cycle.
a, Remodelling cycle initiated from the apo state. In the apo state, Snf2 adopts an open conformation; the nucleosome is distorted with 1-bp bulge at SHL2, primed for the DNA translocation (stage 1). After ATP binding, Snf2 undergoes a large domain rotation to a closed conformation (stage 2), in which lobe2 pushes the DNA distortion to the exit side and the nucleosome recovers to the relaxed state, completing the translocation of 1 bp DNA. After ATP hydrolysis and phosphate release, Snf2 reopens its nucleosome-binding cleft (stage 3), causing a distortion of 1 bp DNA at the site of binding and translocation of the DNA from the proximal end into SHL2, whereby the nucleosome is primed for the next DNA translocation event. No large motion occurs after ADP release and the empty catalytic pocket of the enzyme waits from the next incoming ATP (stage 4). b, Remodelling cycle initiated from the ATP-bound state. The selection of the initial event within an ATPase cycle will not change the directionality of DNA translocation. An ATPase cycle initiated from the ATP-bound state is mechanistically equivalent to that initiated from the apo state (a).
Supplementary information
Supplementary Figure 1
This file contains the uncropped gel.
Video 1
CryoEM map of the “601” nucleosome bound by Snf2 in the ADP state.
Video 2
Morphing video of chromatin remodeling. The video is made according to the proposed model (Extended Data Fig. 10b), with the structures of the stages 1-3 as determined in this study and that of state 4 based on its counterpart in the previous ATPase cycle (stage 1) with 1 bp DNA translocated to the exit side.
Rights and permissions
About this article
Cite this article
Li, M., Xia, X., Tian, Y. et al. Mechanism of DNA translocation underlying chromatin remodelling by Snf2. Nature 567, 409–413 (2019). https://doi.org/10.1038/s41586-019-1029-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1029-2
This article is cited by
-
Energy-driven genome regulation by ATP-dependent chromatin remodellers
Nature Reviews Molecular Cell Biology (2024)
-
Functionalized graphene-oxide grids enable high-resolution cryo-EM structures of the SNF2h-nucleosome complex without crosslinking
Nature Communications (2024)
-
Molecular basis of chromatin remodelling by DDM1 involved in plant DNA methylation
Nature Plants (2024)
-
Structural insights into histone exchange by human SRCAP complex
Cell Discovery (2024)
-
Asymmetric nucleosome PARylation at DNA breaks mediates directional nucleosome sliding by ALC1
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.