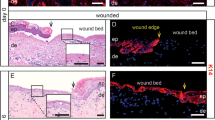
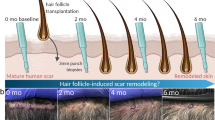
Abstract
Wound healing occurs as a response to disruption of the epidermis and dermis. It is an intricate and well-orchestrated response with the goal to restore skin integrity and function. However, in hundreds of millions of patients, skin wound healing results in abnormal scarring, including keloid lesions or hypertrophic scarring. Although the underlying mechanisms of hypertrophic scars and keloid lesions are not well defined, evidence suggests that the changes in the extracellular matrix are perpetuated by ongoing inflammation in susceptible individuals, resulting in a fibrotic phenotype. The lesions then become established, with ongoing deposition of excess disordered collagen. Not only can abnormal scarring be debilitating and painful, it can also cause functional impairment and profound changes in appearance, thereby substantially affecting patients’ lives. Despite the vast demand on patient health and the medical society, very little progress has been made in the care of patients with abnormal scarring. To improve the outcome of pathological scarring, standardized and innovative approaches are required.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gauglitz, G. G., Korting, H. C., Pavicic, T., Ruzicka, T. & Jeschke, M. G. Hypertrophic scarring and keloids: pathomechanisms and current and emerging treatment strategies. Mol. Med. 17, 113–125 (2011).
Ud-Din, S. & Bayat, A. new insights on keloids, hypertrophic scars, and striae. Dermatol. Clin. 32, 193–209 (2014).
Tan, S., Khumalo, N. & Bayat, A. Understanding keloid pathobiology from a quasi-neoplastic perspective: less of a scar and more of a chronic inflammatory disease with cancer-like tendencies. Front. Immunol. 10, 1810 (2019).
Shih, B. & Bayat, A. Genetics of keloid scarring. Arch. Dermatol. Res. 302, 319–339 (2010).
Zhang, J. et al. Recent advances in hypertrophic scar. Histol. Histopathol. 33, 27–39 (2018).
Lee, S.-S., Yosipovitch, G., Chan, Y.-H. & Goh, C.-L. Pruritus, pain, and small nerve fiber function in keloids: a controlled study. J. Am. Acad. Dermatol. 51, 1002–1006 (2004).
Guest, J. F., Fuller, G. W. & Vowden, P. Cohort study evaluating the burden of wounds to the UK’s National Health Service in 2017/2018: update from 2012/2013. BMJ Open 10, e045253 (2020).
International Surgical Outcomes Study Group. Global patient outcomes after elective surgery: prospective cohort study in 27 low-, middle- and high-income countries. Br. J. Anaesth. 117, 601–609 (2016).
Rose, J. et al. Estimated need for surgery worldwide based on prevalence of diseases: implications for public health planning of surgical services. Lancet Glob. Health 3, S13–S20 (2015).
Amici, J. M. et al. Prevalence of scars: an international epidemiological survey in adults. J. Eur. Acad. Dermatol. Venereol. 36, e799–e800 (2022).
van der Wal, M. B. A. et al. Outcome after burns: an observational study on burn scar maturation and predictors for severe scarring. Wound Repair Regen. 20, 676–687 (2012).
Zhou, C. et al. Beyond the surface: a deeper look at the psychosocial impacts of acne scarring. Clin. Cosmet. Investig. Dermatol. 16, 731–738 (2023).
Liu, S. et al. Keloid: genetic susceptibility and contributions of genetics and epigenetics to its pathogenesis. Exp. Dermatol. 31, 1665–1675 (2022).
Liu, R. et al. Risk factors associated with the progression from keloids to severe keloids. Chin. Med. J. 135, 828–836 (2022).
Ogawa, R. The most current algorithms for the treatment and prevention of hypertrophic scars and keloids: a 2020 update of the algorithms published 10 years ago. Plast. Reconstr. Surg. 149, 79e–94e (2022).
Ingrasci, G., El-Kashlan, N., Alexis, A. & Yosipovitch, G. Chronic itch in African Americans: an unmet need. Arch. Dermatol. Res. 314, 405–415 (2021).
Chung, S., Nakashima, M., Zembutsu, H. & Nakamura, Y. Possible involvement of NEDD4 in keloid formation; its critical role in fibroblast proliferation and collagen production. Proc. Jpn Acad. Ser. B Phys. Biol. Sci. 87, 563–573 (2011).
Zhao, Y. et al. NEDD4 single nucleotide polymorphism rs2271289 is associated with keloids in Chinese Han population. Am. J. Transl. Res. 8, 544–555 (2016).
Wang, H. et al. Identification and validation of HOXD3 and UNC5C as molecular signatures in keloid based on weighted gene co-expression network analysis. Genomics 114, 110403 (2022).
Wang, X. et al. Identification and characterization of four immune-related signatures in keloid. Front. Immunol. 13, 942446 (2022).
Lu, Y.-Y. et al. Risk of cancer development in patients with keloids. Sci. Rep. 11, 9390–9390 (2021).
Madu, P. & Kundu, R. V. Follicular and scarring disorders in skin of color: presentation and management. Am. J. Clin. Dermatol. 15, 307–321 (2014).
Huang, C. & Ogawa, R. Systemic factors that shape cutaneous pathological scarring. FASEB J. 34, 13171–13184 (2020).
Ibrahim, N. E., Shaharan, S. & Dheansa, B. Adverse effects of pregnancy on keloids and hypertrophic scars. Cureus 12, e12154 (2020).
Noishiki, C., Hayasaka, Y. & Ogawa, R. Sex differences in keloidogenesis: an analysis of 1659 keloid patients in Japan. Dermatol. Ther. 9, 747–754 (2019).
Ogawa, R. et al. The relationship between skin stretching/contraction and pathologic scarring: the important role of mechanical forces in keloid generation. Wound Repair Regen. 20, 149–157 (2012).
Wang, J. C., Fort, C. L. & Hom, D. B. Location propensity for keloids in the head and neck. Facial Plast. Surg. Aesthet. Med. 23, 59–64 (2021).
Arno, A. I., Gauglitz, G. G., Barret, J. P. & Jeschke, M. G. Up-to-date approach to manage keloids and hypertrophic scars: a useful guide. Burns 40, 1255–1266 (2014).
Kwon, H.-E., Ahn, H.-J., Jeong, S. J. & Shin, M. K. The increased prevalence of keloids in atopic dermatitis patients with allergic comorbidities: a nationwide retrospective cohort study. Sci. Rep. 11, 23669 (2021).
Ung, C. Y. et al. Comorbidities of keloid and hypertrophic scars among participants in UK Biobank. JAMA Dermatol. 159, 172–181 (2023).
Limandjaja, G. C., Niessen, F. B., Scheper, R. J. & Gibbs, S. Hypertrophic scars and keloids: overview of the evidence and practical guide for differentiating between these abnormal scars. Exp. Dermatol. 30, 146–161 (2021).
Butzelaar, L., Ulrich, M. M. W., Mink van der Molen, A. B., Niessen, F. B. & Beelen, R. H. J. Currently known risk factors for hypertrophic skin scarring: a review. J. Plast. Reconstr. Aesthet. Surg. 69, 163–169 (2016).
Davies, P., Cuttle, L. & Young, A. A scoping review of the methodology used in studies of genetic influences on the development of keloid or hypertrophic scarring in adults and children after acute wounding. Adv. Wound Care 10, 557–570 (2021).
Lawrence, J. W., Mason, S. T., Schomer, K. & Klein, M. B. Epidemiology and impact of scarring after burn injury. J. Burn Care Res. 33, 136–146 (2012).
Korkmaz, H. I. et al. Scar formation from the perspective of complexity science: a new look at the biological system as a whole. J. Wound Care 31, 178–184 (2022).
Cb, N., Nc, D., Jm, H., M, M. & Jg, W. Burns: pathophysiology of systemic complications and current management. J. Burn Care Res. 38, e469–e481 (2017).
WHO. Fact Sheet: Burns. WHO https://www.who.int/en/news-room/fact-sheets/detail/burns (2023).
Spronk, I. et al. Health related quality of life in adults after burn injuries: a systematic review. PLoS ONE 13, e0197507 (2018).
van Baar, M. E. in Textbook on Scar Management (eds Téot, L., Mustoe, T. A., Middelkoop, E. & Gauglitz, G. G.) 37–43 (Springer, 2020).
van de Kar, A. L., van Riessen, F., Koolbergen, D. R. & van der Horst, C. M. A. M. Influence of age on scar tissue: a retrospective study on the differences in scar tissue development between children and adults. J. Plast. Reconstr. Aesthet. Surg. 73, 1357–1404 (2020).
van der Veer, W. M. et al. Potential cellular and molecular causes of hypertrophic scar formation. Burns 35, 15–29 (2009).
Fitzpatrick, T. B. The validity and practicality of sun-reactive skin types I through VI. Arch. Dermatol. 124, 869 (1988).
Sasor, S. E. & Chung, K. C. Upper extremity burns in the developing world: a neglected epidemic. Hand Clin. 35, 457–466 (2019).
Mackenbach, J. P. et al. Socioeconomic inequalities in health in 22 European countries. N. Engl. J. Med. 358, 2468–2481 (2008).
MacKinnon, N. J. et al. Mapping health disparities in 11 high-income nations. JAMA Netw. Open 6, e2322310 (2023).
Hendriks, T. C. C. et al. The development of burn scar contractures and impact on joint function, disability and quality of life in low- and middle-income countries: a prospective cohort study with one-year follow-up. Burns 48, 215–227 (2022).
Oosterwijk, A. M. et al. Prevalence of scar contractures after burn: a systematic review. Burns 43, 41–49 (2017).
Meng, F. et al. Pediatric burn contractures in low- and lower middle-income countries: a systematic review of causes and factors affecting outcome. Burns 46, 993–1004 (2020).
Wong, V. W. et al. A mechanomodulatory device to minimize incisional scar formation. Adv. Wound Care 2, 185–194 (2013).
Bayat, A., McGrouther, D. A. & Ferguson, M. W. J. Skin scarring. BMJ 326, 88–92 (2003).
Ud-Din, S., Volk, S. W. & Bayat, A. Regenerative healing, scar-free healing and scar formation across the species: current concepts and future perspectives. Exp. Dermatol. 23, 615–619 (2014).
Shih, B., Garside, E., McGrouther, D. A. & Bayat, A. Molecular dissection of abnormal wound healing processes resulting in keloid disease. Wound Repair Regen. 18, 139–153 (2010).
Sinha, S. et al. Fibroblast inflammatory priming determines regenerative versus fibrotic skin repair in reindeer. Cell 185, 4717–4736.e25 (2022).
Bagabir, R. et al. Site-specific immunophenotyping of keloid disease demonstrates immune upregulation and the presence of lymphoid aggregates. Br. J. Dermatol. 167, 1053–1066 (2012).
Brown, J. J. & Bayat, A. Genetic susceptibility to raised dermal scarring. Br. J. Dermatol. 161, 8–18 (2009).
Hosseini, M., Brown, J., Khosrotehrani, K., Bayat, A. & Shafiee, A. Skin biomechanics: a potential therapeutic intervention target to reduce scarring. Burns Trauma 10, tkac036 (2022).
Greaves, N. S., Ashcroft, K. J., Baguneid, M. & Bayat, A. Current understanding of molecular and cellular mechanisms in fibroplasia and angiogenesis during acute wound healing. J. Dermatol. Sci. 72, 206–217 (2013).
Hodgkinson, T. & Bayat, A. Dermal substitute-assisted healing: enhancing stem cell therapy with novel biomaterial design. Arch. Dermatol. Res. 303, 301–315 (2011).
Li, Z. et al. Epidermal Notch1 recruits RORγ+ group 3 innate lymphoid cells to orchestrate normal skin repair. Nat. Commun. 7, 11394 (2016).
Rak, G. D. et al. IL-33-dependent group 2 innate lymphoid cells promote cutaneous wound healing. J. Invest. Dermatol. 136, 487–496 (2016).
Kidzeru, E. B. et al. Immune cells and associated molecular markers in dermal fibrosis with focus on raised cutaneous scars. Exp. Dermatol. 32, 570–587 (2023).
Rees, P. A., Greaves, N. S., Baguneid, M. & Bayat, A. Chemokines in wound healing and as potential therapeutic targets for reducing cutaneous scarring. Adv. Wound Care 4, 687–703 (2015).
Tanno, H. et al. Invariant NKT cells promote skin wound healing by preventing a prolonged neutrophilic inflammatory response. Wound Repair Regen. 25, 805–815 (2017).
Iqbal, S. A., Sidgwick, G. P. & Bayat, A. Identification of fibrocytes from mesenchymal stem cells in keloid tissue: a potential source of abnormal fibroblasts in keloid scarring. Arch. Dermatol. Res. 304, 665–671 (2012).
Ud-Din, S., Wilgus, T. A. & Bayat, A. Mast cells in skin scarring: a review of animal and human research. Front. Immunol. 11, 552205 (2020).
Govindaraju, P., Todd, L., Shetye, S., Monslow, J. & Puré, E. CD44-dependent inflammation, fibrogenesis, and collagenolysis regulates extracellular matrix remodeling and tensile strength during cutaneous wound healing. Matrix Biol. 75–76, 314–330 (2019).
Kidzeru, E. B. et al. Immune and associated molecular markers – their role in fibrotic dermal scarring. Exp. Dermatol. 32, 570–587 (2022).
Xiao, T., Yan, Z., Xiao, S. & Xia, Y. Proinflammatory cytokines regulate epidermal stem cells in wound epithelialization. Stem Cell Res. Ther. 11, 232 (2020).
Lau, K., Paus, R., Tiede, S., Day, P. & Bayat, A. Exploring the role of stem cells in cutaneous wound healing. Exp. Dermatol. 18, 921–933 (2009).
Moses, H. L., Roberts, A. B. & Derynck, R. The discovery and early days of TGF-β: a historical perspective. Cold Spring Harb. Perspect. Biol. 8, a021865 (2016).
Jagadeesan, J. & Bayat, A. Transforming growth factor beta (TGFβ) and keloid disease. Int. J. Surg. 5, 278–285 (2007).
Pakshir, P. et al. The myofibroblast at a glance. J. Cell Sci. 133, jcs227900 (2020).
Taylor, A. et al. Transforming growth factor beta gene signatures are spatially enriched in keloid tissue biopsies and ex vivo-cultured keloid fibroblasts. Acta Derm. Venereol. 97, 10–16 (2017).
Willenborg, S., Injarabian, L. & Eming, S. A. Role of macrophages in wound healing. Cold Spring Harb. Perspect. Biol. 14, a041216 (2022).
Wynn, T. A. & Vannella, K. M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity 44, 450–462 (2016).
Zhang, M. & Zhang, S. T cells in fibrosis and fibrotic diseases. Front. Immunol. 11, 1142 (2020).
Nguyen, J. K., Austin, E., Huang, A., Mamalis, A. & Jagdeo, J. The IL-4/IL-13 axis in skin fibrosis and scarring: mechanistic concepts and therapeutic targets. Arch. Dermatol. Res. 312, 81–92 (2020).
Ashrafi, M., Baguneid, M. & Bayat, A. The role of neuromediators and innervation in cutaneous wound healing. Acta Derm. Venereol. 96, 587–594 (2016).
Volk, S. W., Iqbal, S. A. & Bayat, A. Interactions of the extracellular matrix and progenitor cells in cutaneous wound healing. Adv. Wound Care 2, 261–272 (2013).
Syed, F. et al. Fibroblasts from the growing margin of keloid scars produce higher levels of collagen I and III compared with intralesional and extralesional sites: clinical implications for lesional site-directed therapy. Br. J. Dermatol. 164, 83–96 (2010).
Khan, U. & Bayat, A. Microarchitectural analysis of decellularised unscarred and scarred dermis provides insight into the organisation and ultrastructure of the human skin with implications for future dermal substitute scaffold design. J. Tissue Eng. 10, 2041731419843710 (2019).
Sidgwick, G. P. & Bayat, A. Extracellular matrix molecules implicated in hypertrophic and keloid scarring. J. Eur. Acad. Dermatol. Venereol. 26, 141–152 (2012).
Folkman, J. Angiogenesis. Annu. Rev. Med. 57, 1–18 (2006).
Chaudhari, N. et al. Topical application of an irreversible small molecule inhibitor of lysyl oxidases ameliorates skin scarring and fibrosis. Nat. Commun. 13, 5555 (2022).
Ud-Din, S. & Bayat, A. Classification of distinct endotypes in human skin scarring: S.C.A.R.-a novel perspective on dermal fibrosis. Adv. Wound Care 11, 109–120 (2022).
Sorg, H., Tilkorn, D. J., Hager, S., Hauser, J. & Mirastschijski, U. Skin wound healing: an update on the current knowledge and concepts. Eur. Surg. Res. 58, 81–94 (2016).
Elrefaie, A. M., Salem, R. M. & Faheem, M. H. High-resolution ultrasound for keloids and hypertrophic scar assessment. Lasers Med. Sci. 35, 379–385 (2020).
Jacobs, J. E., Birnbaum, B. A. & Siegelman, E. S. Heterotopic ossification of midline abdominal incisions: CT and MR imaging findings. Am. J. Roentgenol. 166, 579–584 (1996).
Roques, C., Téot, L., Frasson, N. & Meaume, S. PRIMOS: an optical system that produces three-dimensional measurements of skin surfaces. J. Wound Care 12, 362–365 (2003).
Deng, H. & Li-Tsang, C. W. P. Measurement of vascularity in the scar: a systematic review. Burns 45, 1253–1265 (2019).
Leszczynski, R., da Silva, C. A., Pinto, A. C. P. N., Kuczynski, U. & da Silva, E. M. Laser therapy for treating hypertrophic and keloid scars. Cochrane Database Syst. Rev. 9, CD011642 (2022).
Baryza, M. J. & Baryza, G. A. The vancouver scar scale: an administration tool and its interrater reliability. J. Burn Care Rehabil. 16, 535–538 (1995).
van de Kar, A. L. et al. Reliable and feasible evaluation of linear scars by the patient and observer scar assessment scale. Plast. Reconstr. Surg. 116, 514–522 (2005).
Duncan, J. A. L. et al. Visual analogue scale scoring and ranking: a suitable and sensitive method for assessing scar quality? Plast. Reconstr. Surg. 118, 909–918 (2006).
Ogawa, R. et al. Diagnosis and treatment of keloids and hypertrophic scars-japan scar workshop consensus document 2018. Burns Trauma 7, 39 (2019).
Scar-Keloid. Japan Scar Workshop Scar Scale 2011. Japan Scar Workshop http://www.scar-keloid.com/pdf/JSW_scar_scale_2011_EN.pdf (2011).
Scar-Keloid. Japan Scar Workshop Scar Scale 2015. Japan Scar Workshop http://www.scar-keloid.com/pdf/JSW_scar_scale_2015_EN.pdf (2015).
van Zuijlen, P. P. et al. Dermal substitution in acute burns and reconstructive surgery: a subjective and objective long-term follow-up. Plast. Reconstr. Surg. 108, 1938–1946 (2001).
Nixon, M., Outlaw, F. & Leung, T. S. Accurate device-independent colorimetric measurements using smartphones. PLoS ONE 15, e0230561 (2020).
Sanderson, R. W. et al. Smartphone-based optical palpation: towards elastography of skin for telehealth applications. Biomed. Opt. Express 12, 3117–3132 (2021).
Carmichael, S. W. The tangled web of Langer’s lines. Clin. Anat. 27, 162–168 (2014).
Harn, H. I.-C. et al. The tension biology of wound healing. Exp. Dermatol. 28, 464–471 (2019).
Ogawa, R. et al. Clinical applications of basic research that shows reducing skin tension could prevent and treat abnormal scarring: the importance of fascial/subcutaneous tensile reduction sutures and flap surgery for keloid and hypertrophic scar reconstruction. J. Nippon Med. Sch. 78, 68–76 (2011).
Smith, D. J., Thomson, P. D., Garner, W. L. & Rodriguez, J. L. Burn wounds: infection and healing. Am. J. Surg. 167, S46–S48 (1994).
Amici, J.-M. et al. Expert recommendations on supportive skin care for non-surgical and surgical procedures. J. Eur. Acad. Dermatol. Venereol. 37, 16–33 (2023).
Goutos, I., Dziewulski, P. & Richardson, P. M. Pruritus in burns: review article. J. Burn. Care Res. 30, 221–228 (2009).
Andrews, J. P., Marttala, J., Macarak, E., Rosenbloom, J. & Uitto, J. Keloids: the paradigm of skin fibrosis - pathomechanisms and treatment. Matrix Biol. 51, 37–46 (2016).
Finnerty, C. C. et al. Hypertrophic scarring: the greatest unmet challenge after burn injury. Lancet 388, 1427–1436 (2016).
O’Reilly, S., Crofton, E., Brown, J., Strong, J. & Ziviani, J. Use of tape for the management of hypertrophic scar development: a comprehensive review. Scars Burn. Heal. 7, 205951312110292 (2021).
Deflorin, C. et al. Physical management of scar tissue: a systematic review and meta-analysis. J. Altern. Complement. Med. 26, 854–865 (2020).
Ai, J.-W. et al. The effectiveness of pressure therapy (15–25 mmHg) for hypertrophic burn scars: a systematic review and meta-analysis. Sci. Rep. 7, 40185 (2017).
Finnerty, C. C. et al. Hypertrophic scarring: the greatest unmet challenge following burn injury. Lancet 388, 1427–1436 (2016).
Jeschke, M. G. et al. Burn injury. Nat. Rev. Dis. Primers 6, 11 (2020).
Thomson, W. G. Treatment of hypertrophic scarring by compression and occlusion. Proc. R. Soc. Med. 67, 256–257 (1974).
Lin, T. R., Chou, F.-H., Wang, H.-H. & Wang, R.-H. Effects of scar massage on burn scars: a systematic review and meta-analysis. J. Clin. Nurs. 32, 3144–3154 (2023).
Anzarut, A., Olson, J., Singh, P., Rowe, B. H. & Tredget, E. E. The effectiveness of pressure garment therapy for the prevention of abnormal scarring after burn injury: a meta-analysis. J. Plast. Reconstr. Aesthet. Surg. 62, 77–84 (2009).
Wiseman, J. et al. Effectiveness of topical silicone gel and pressure garment therapy for burn scar prevention and management in children 12-months postburn: a parallel group randomised controlled trial. Clin. Rehabil. 35, 1126–1141 (2021).
Zhuang, Z., Li, Y. & Wei, X. The safety and efficacy of intralesional triamcinolone acetonide for keloids and hypertrophic scars: a systematic review and meta-analysis. Burns 47, 987–998 (2021).
Gold, M. H. et al. Updated international clinical recommendations on scar management: part 1–evaluating the evidence. Dermatol. Surg. 40, 817–824 (2014).
Ogawa, R. Keloid and hypertrophic scars are the result of chronic inflammation in the reticular dermis. Int. J. Mol. Sci. 18, 606 (2017).
Ceilley, R. I. & Babin, R. W. The combined use of cryosurgery and intralesional injections of suspensions of fluorinated adrenocorticosteroids for reducing keloids and hypertrophic scars. J. Dermatol. Surg. Oncol. 5, 54–56 (1979).
Schwaiger, H., Reinholz, M., Poetschke, J., Ruzicka, T. & Gauglitz, G. Evaluating the therapeutic success of keloids treated with cryotherapy and intralesional corticosteroids using noninvasive objective measures. Dermatol. Surg. 44, 635–644 (2018).
Goutos, I. & Ogawa, R. Steroid tape: a promising adjunct to scar management. Scars Burn. Heal. 3, 2059513117690937 (2017).
Ogawa, R. in Textbook on Scar Management (eds Téot, L., Mustoe, T. A., Middelkoop, E. & Gauglitz, G. G.) 491–496 (Springer, 2020).
Nast, A. et al. German S2k guidelines for the therapy of pathological scars (hypertrophic scars and keloids). J. Dtsch Dermatol. Ges. 10, 747–760 (2012).
Gauglitz, G. Management of keloids and hypertrophic scars: current and emerging options. Clin. Cosmet. Investig. Dermatol. 6, 103–114 (2013).
O’Boyle, C. P., Shayan-Arani, H. & Hamada, M. W. Intralesional cryotherapy for hypertrophic scars and keloids: a review. Scars Burn. Heal. 3, 205951311770216 (2017).
Fitzpatrick, R. E. Treatment of inflamed hypertrophic scars using intralesional 5-FU. Dermatol. Surg. 25, 224–232 (1999).
Nanda, S. & Reddy, B. S. N. Intralesional 5-fluorouracil as a treatment modality of keloids. Dermatol. Surg. 30, 54–57 (2004).
Liu, W., Wu, X., Gao, Z. & Song, N. Remodelling of keloid tissue into normal-looking skin. J. Plast. Reconstr. Aesthet. Surg. 61, 1553–1554 (2008).
Darougheh, A., Asilian, A. & Shariati, F. Intralesional triamcinolone alone or in combination with 5-fluorouracil for the treatment of keloid and hypertrophic scars. Clin. Exp. Dermatol. 34, 219–223 (2009).
Davison, S. P. et al. Efficacy of intralesional 5-fluorouracil and triamcinolone in the treatment of keloids. Aesthet. Surg. J. 29, 40–46 (2009).
Reinholz, M., Guertler, A., Schwaiger, H., Poetschke, J. & Gauglitz, G. G. Treatment of keloids using 5‐fluorouracil in combination with crystalline triamcinolone acetonide suspension: evaluating therapeutic effects by using non‐invasive objective measures. J. Eur. Acad. Dermatol. Venereol. 34, 2436–2444 (2020).
Zhang, W., Li, X. & Li, X. Efficacy and safety of verapamil versus triamcinolone acetonide in treating keloids and hypertrophic scars: a systematic review and meta-analysis. Aesthetic Plast. Surg. 47, 473–482 (2023).
Danielsen, P. L. et al. Verapamil is less effective than triamcinolone for prevention of keloid scar recurrence after excision in a randomized controlled trial. Acta Derm. Venereol. 96, 774–778 (2016).
Paasch, U. et al. S2k guideline: laser therapy of the skin. J. Dtsch Dermatol. Ges. 20, 1248–1267 (2022).
Nast, A. et al. S2k guidelines for the therapy of pathological scars (hypertrophic scars and keloids) – Update 2020. J. Dtsch Dermatol. Ges. 19, 312–327 (2020).
Seago, M. et al. Laser treatment of traumatic scars and contractures: 2020 international consensus recommendations. Lasers Surg. Med. 52, 96–116 (2019).
Salameh, F. et al. Energy-based devices for the treatment of acne scars: 2022 International consensus recommendations. Lasers Surg. Med. 54, 10–26 (2022).
Guertler, A. et al. Objective evaluation of the efficacy of a non-ablative fractional 1565 nm laser for the treatment of deliberate self-harm scars. Lasers Med. Sci. 33, 241–250 (2018).
Alegre-Sánchez, A., Jiménez-Gómez, N. & Boixeda, P. Laser-assisted drug delivery. Actas Dermosifiliogr. 109, 858–867 (2018).
Muskat, A., Kost, Y., Balazic, E., Cohen, J. L. & Kobets, K. Laser-assisted drug delivery in the treatment of scars, rhytids, and melasma: a comprehensive review of the literature. Aesthet. Surg. J. 43, NP181–NP198 (2023).
You, H. et al. The outcome of early ablative fractional laser treatment for thyroidectomy scars. Lasers Surg. Med. 52, 721–729 (2020).
Zainib, M. & Amin, N. P. Radiation therapy in the treatment of keloids. StatPearls [online] https://www.ncbi.nlm.nih.gov/books/NBK499973/ (updated 31 July 2023).
Shin, H. W., Suk, S., Chae, S. W., Yoon, K. C. & Kim, J. Early postoperative treatment of mastectomy scars using a fractional carbon dioxide laser: a randomized, controlled, split-scar, blinded study. Arch. Plast. Surg. 48, 347–352 (2021).
Juhasz, M. L. W. & Cohen, J. L. Microneedling for the treatment of scars: an update for clinicians. Clin. Cosmet. Investig. Dermatol. 13, 997–1003 (2020).
Chopra, V. K. & Claytor, B. B. Microneedling of immature scars Is safe and improves scar esthetics. Plast. Reconstr. Surg. Glob. Open 7, 13 (2019).
Ogawa, R., Yoshitatsu, S., Yoshida, K. & Miyashita, T. Is radiation therapy for keloids acceptable? The risk of radiation-induced carcinogenesis. Plast. Reconstr. Surg. 124, 1196–1201 (2009).
Ogawa, R. Surgery for scar revision and reduction: from primary closure to flap surgery. Burns Trauma 7, 7 (2019).
Arima, J., Dohi, T., Kuribayashi, S., Akaishi, S. & Ogawa, R. Z-plasty and postoperative radiotherapy for anterior chest wall keloids: an analysis of 141 patients. Plast. Reconstr. Surg. Glob. Open 7, e2177 (2019).
Ogawa, R., Dohi, T., Tosa, M., Aoki, M. & Akaishi, S. The latest strategy for keloid and hypertrophic scar prevention and treatment: the Nippon Medical School (NMS) protocol. J. Nippon Med. Sch. 88, 2–9 (2021).
Simman, R. Wound closure and the reconstructive ladder in plastic surgery. J. Am. Coll. Certif. Wound Spec. 1, 6–11 (2009).
Willows, B. M., Ilyas, M. & Sharma, A. Laser in the management of burn scars. Burns 43, 1379–1389 (2017).
Wood, F. M. in Textbook on Scar Management (eds Téot, L., Mustoe, T. A., Middelkoop, E. & Gauglitz, G. G.) 311–316, 10.1007/978-3-030-44766-3_36 (Springer, 2020).
Lo, C. H. et al. Wound healing and dermal regeneration in severe burn patients treated with NovoSorb® Biodegradable Temporising Matrix: a prospective clinical study. Burns 48, 529–538 (2022).
Finlay, V. et al. Increased burn healing time is associated with higher Vancouver Scar Scale score. Scars Burn. Heal. 3, 2059513117696324 (2017).
Stone Ii, R. et al. Advancements in regenerative strategies through the continuum of burn care. Front. Pharmacol. 9, 672 (2018).
Zuo, K. J., Medina, A. & Tredget, E. E. Important developments in burn care. Plast. Reconstr. Surg. 139, 120e–138e (2017).
Finnerty, C. C., Mabvuure, N. T., Ali, A., Kozar, R. A. & Herndon, D. N. The surgically induced stress response. JPEN J. Parenter. Enter. Nutr. 37, 21S–29S (2013).
Asuku, M. et al. Split-thickness skin graft donor-site morbidity: a systematic literature review. Burns 47, 1525–1546 (2021).
Chogan, F., Chen, Y., Wood, F. & Jeschke, M. G. Skin tissue engineering advances in burns: a brief introduction to the past, the present, and the future potential. J. Burn Care Res. 44, S1–S4 (2022).
Wood, F. M. Skin regeneration: the complexities of translation into clinical practise. Int. J. Biochem. Cell Biol. 56, 133–140 (2014).
Ramakrishnan, R. et al. Human-derived scaffold components and stem cells creating immunocompatible dermal tissue ensuing regulated nonfibrotic cellular phenotypes. ACS Biomater. Sci. Eng. 6, 2740–2756 (2020).
Varkey, M., Ding, J. & Tredget, E. E. Advances in skin substitutes-potential of tissue engineered skin for facilitating anti-fibrotic healing. J. Funct. Biomater. 6, 547–563 (2015).
Monstrey, S. et al. Updated scar management practical guidelines: non-invasive and invasive measures. J. Plast. Reconstr. Aesthet. Surg. 67, 1017–1025 (2014).
Teot, L., Cherenfant, E., Otman, S. & Giovannini, U. M. Prefabricated vascularised supraclavicular flaps for face resurfacing after postburns scarring. Lancet 355, 1695–1696 (2000).
Rüegg, E. M. et al. Face transplantation program in france. Transplantation 93, 1166–1172 (2012).
Kildal, M., Andersson, G., Fugl-Meyer, A. R., Lannerstam, K. & Gerdin, B. Development of a brief version of the Burn Specific Health Scale (BSHS-B). J. Trauma 51, 740–746 (2001).
Meirte, J. et al. Classification of quality of life subscales within the ICF framework in burn research: identifying overlaps and gaps. Burns 40, 1353–1359 (2014).
Spronk, I. et al. Health related quality of life 5–7 years after minor and severe burn injuries: a multicentre cross-sectional study. Burns 45, 1291–1299 (2019).
Gojowy, D., Kauke, M., Ohmann, T., Homann, H.-H. & Mannil, L. Early and late-recorded predictors of health-related quality of life of burn patients on long-term follow-up. Burns 45, 1300–1310 (2019).
Bijlard, E. A systematic review on the prevalence, etiology, and pathophysiology of intrinsic pain in dermal scar tissue. Pain Physician 1, 1–13 (2017).
Balci, D. D., Inandi, T., Dogramaci, C. A. & Celik, E. DLQI scores in patients with keloids and hypertrophic scars: a prospective case control study. J. Dtsch Dermatol. Ges. 7, 688–691 (2009).
Bock, O., Schmid-Ott, G., Malewski, P. & Mrowietz, U. Quality of life of patients with keloid and hypertrophic scarring. Arch. Dermatol. Res. 297, 433–438 (2006).
Reinholz, M. et al. The dermatology life quality index as a means to assess life quality in patients with different scar types. J. Eur. Acad. Dermatol. Venereol. 29, 2112–2119 (2015).
Gieler, U., Gieler, T. & Kupfer, J. P. Acne and quality of life - impact and management. J. Eur. Acad. Dermatol. Venereol. 29, 12–14 (2015).
HAHM, B. J. et al. Changes of psychiatric parameters and their relationships by oral isotretinoin in acne patients. J. Dermatol. 36, 255–261 (2009).
Van Loey, N. E. E. & Van Son, M. J. M. Psychopathology and psychological problems in patients with burn scars. Am. J. Clin. Dermatol. 4, 245–272 (2003).
Spronk, I., Legemate, C. M., Polinder, S. & van Baar, M. E. Health-related quality of life in children after burn injuries. J. Trauma Acute Care Surg. 85, 1110–1118 (2018).
Poetschke, J. et al. Ultrapulsed fractional ablative carbon dioxide laser treatment of hypertrophic burn scars: evaluation of an in-patient controlled, standardized treatment approach. Lasers Med. Sci. 32, 1031–1040 (2017).
Boersma-van Dam, E., van de Schoot, R., Hofland, H. W. C., Engelhard, I. M. & Van Loey, N. E. E. Individual recovery of health-related quality of life during 18 months post-burn using a retrospective pre-burn measurement: an exploratory study. Qual. Life Res. 30, 737–749 (2021).
Meuli, M. et al. A cultured autologous dermo-epidermal skin substitute for full-thickness skin defects. Plast. Reconstr. Surg. 144, 188–198 (2019).
Dagher, J., Arcand, C., Auger, F. A., Germain, L. & Moulin, V. J. The self-assembled skin substitute history: successes, challenges, and current treatment indications. J. Burn Care Res. 44, S57–S64 (2023).
Cervelli, V. et al. Treatment of stable vitiligo by ReCell system. Acta Dermatovenerol. Croat. 17, 273–278 (2009).
Cervelli, V. et al. Use of a novel autologous cell-harvesting device to promote epithelialization and enhance appropriate pigmentation in scar reconstruction. Clin. Exp. Dermatol. 35, 776–780 (2010).
Armendariz-Borunda, J. et al. A controlled clinical trial with pirfenidone in the treatment of pathological skin scarring caused by burns in pediatric patients. Ann. Plast. Surg. 68, 22–28 (2012).
Knuth, C. M. et al. Single-nuclei RNA profiling reveals disruption of adipokine and inflammatory signaling in adipose tissue of burn patients. Ann. Surg. https://doi.org/10.1097/SLA.0000000000005880 (2023).
Lee, C.-C. et al. An updated review of the immunological mechanisms of keloid scars. Front. Immunol. 14, 1117630 (2023).
Poetschke, J. & Gauglitz, G. G. Current options for the treatment of pathological scarring. J. Dtsch Dermatol. Ges. 14, 467–477 (2016).
Mustoe, T. A. et al. International clinical recommendations on scar management. Plast. Reconstr. Surg. 110, 560–571 (2002).
Author information
Authors and Affiliations
Contributions
Introduction (M.G.J., F.M.W. and G.G.G.); Epidemiology (M.G.J., E.M., F.M.W. and G.G.G.); Mechanisms/pathophysiology (M.G.J., A.B. and G.G.G.); Diagnosis, screening and prevention (M.G.J., E.M., L.T., R.O. and G.G.G.); Management (M.G.J., E.M., L.T., R.O. and G.G.G.); Quality of life (M.G.J., E.M. and G.G.G.); Outlook (M.G.J., F.M.W. and G.G.G.); Overview of the Primer (M.G.J. and G.G.G.).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Disease Primers thanks D. M. Supp and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Informed consent
The authors affirm that human research participants provided informed consent for publication of the images in Fig. 6.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jeschke, M.G., Wood, F.M., Middelkoop, E. et al. Scars. Nat Rev Dis Primers 9, 64 (2023). https://doi.org/10.1038/s41572-023-00474-x
Accepted:
Published:
DOI: https://doi.org/10.1038/s41572-023-00474-x
This article is cited by
-
Injectable hydrogel with doxorubicin-loaded ZIF-8 nanoparticles for tumor postoperative treatments and wound repair
Scientific Reports (2024)
-
The clinical efficacy of single-hole punch excision combined with intralesional steroid injection for nodular keloid treatment: a self-controlled trial
Scientific Reports (2024)