Abstract
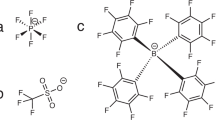
To access the full performance potential of advanced batteries, electrodes and electrolytes must be designed to facilitate ion transport at all applicable length scales. Here, we perform electrodynamic measurements on single electrode particles of ~6 nAh capacity, decouple bulk and interfacial transport from other pathways and show that Li intercalation into LiNi0.33Mn0.33Co0.33O2 (NMC333) is primarily impeded by interfacial kinetics when using a conventional LiPF6 salt. Electrolytes containing LiTFSI salt, with or without LiPF6, exhibit about 100-fold higher exchange current density under otherwise identical conditions. This anion group effect is explained using molecular dynamics simulations to identify preferred solvation structures, density functional theory calculations of their binding energies and Raman spectroscopy confirmation of solvation structure. We show that TFSI− preferentially solvates Li+ compared to PF6−, and yet its preferred solvation structures provide a lower Li+ binding energy, suggesting a lower desolvation energy consistent with ultrafast interfacial kinetics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All relevant data are included in the paper and its Supplementary Information.
References
Fredericks, W. L., Sripad, S., Bower, G. C. & Viswanathan, V. Performance metrics required of next generation batteries to electrify vertical takeoff and landing (VTOL) aircraft. ACS Energy Lett. 3, 2989–2994 (2018).
Sander, J. S., Erb, R. M., Li, L., Gurijala, A. & Chiang, Y.-M. High-performance battery electrodes via magnetic templating. Nat. Energy 1, 16099 (2016).
Delattre, B. et al. Impact of pore tortuosity on electrode kinetics in lithium battery electrodes: study in directionally freeze-cast LiNi0.8Co0.15Al0.05O2 (NCA). J. Electrochem. Soc. 165, A388–A395 (2018).
Suo, L. et al. “Water-in-salt” electrolyte enables high-voltage aqueous lithium-ion chemistries. Science 350, 938–943 (2015).
Yamada, Y. & Yamada, A. Review—superconcentrated electrolytes for lithium batteries. J. Electrochem. Soc. 162, A2406–A2423 (2015).
Yamada, Y. et al. Unusual stability of acetonitrile-based superconcentrated electrolytes for fast-charging lithium-ion batteries. J. Am. Chem. Soc. 136, 5039–5046 (2014).
Wang, J. et al. Superconcentrated electrolytes for a high-voltage lithium-ion battery. Nat. Commun. 7, 12032 (2016).
Zheng, J. et al. Electrolyte additive enabled fast charging and stable cycling lithium metal batteries. Nat. Energy 2, 17012 (2017).
Xu, K. Electrolytes and interphases in Li-ion batteries and beyond. Chem. Rev. 114, 11503–11618 (2014).
Amin, R., Ravnsbæk, D. B. & Chiang, Y.-M. Characterization of electronic and ionic transport in Li1-xNi0.8Co0.15Al0.05O2 (NCA) batteries and energy storage. J. Electrochem. Soc. 162, A1163–A1169 (2015).
Amin, R. & Chiang, Y.-M. Characterization of electronic and ionic transport in Li1-xNi0.33Mn0.33Co0.33O2 (NMC333) and Li1-xNi0.50Mn0.20Co0.30O2 (NMC523) as a function of li content batteries and energy storage. J. Electrochem. Soc. 163, A1512–A1517 (2016).
Jow, T. R., Delp, S. A., Allen, J. L., Jones, J.-P. & Smart, M. C. Factors limiting Li+ charge transfer kinetics in Li-ion batteries. J. Electrochem. Soc. 2, A361–A367 (2018).
Tsai, P.-C. et al. Single-particle measurements of electrochemical kinetics in NMC and NCA cathodes for Li-ion batteries. Energy Environ. Sci. 11, 860–871 (2018).
Kanamura, K., Yamada, Y., Annaka, K., Nakata, N. & Munakata, H. Electrochemical evaluation of active materials for lithium ion batteries by one (single) particle measurement. Electrochemistry 84, 759–765 (2016).
Ando, K. et al. Degradation analysis of LiNi0.8Co0.15Al0.05O2 for cathode material of lithium-ion battery using single-particle measurement. ACS Appl. Energy Mater. 1, 4536–4544 (2018).
Li, J., Xiao, X., Yang, F., Verbrugge, M. W. & Cheng, Y.-T. Potentiostatic intermittent titration technique for electrodes governed by diffusion and interfacial reaction. J. Phys. Chem. C. 116, 1472–1478 (2012).
Li, J., Yang, F., Xiao, X., Verbrugge, M. W. & Cheng, Y.-T. Potentiostatic intermittent titration technique (PITT) for spherical particles with finite interfacial kinetics. Electrochim. Acta 75, 56–61 (2012).
Inada, R., Kumasaka, R., Inabe, S., Tojo, T. & Sakurai, Y. Li+ insertion/extraction properties for TiNb2O7 single particle characterized by a particle-current collector integrated microelectrode. J. Electrochem. Soc. 33, A5157–A5162 (2019).
Lin, F. et al. Surface reconstruction and chemical evolution of stoichiometric layered cathode materials for lithium-ion batteries. Nat. Commun. 5, 3529 (2014).
Wang, F. et al. Hybrid aqueous/non-aqueous electrolyte for safe and high-energy Li-ion batteries. Joule 2, 927–937 (2018).
Jiao, S. et al. Stable cycling of high-voltage lithium metal batteries in ether electrolytes. Nat. Energy 3, 739–746 (2018).
Lu, Y.-C., Mansour, A. N., Yabuuchi, N. & Shao-Horn, Y. Probing the origin of enhanced stability of ‘AlPO4’ nanoparticle coated LiCoO2 during cycling to high voltages: combined XRD and XPS Studies. Chem. Mater. 21, 4408–4424 (2009).
Wagner, R. et al. Impact of selected LiPF6 hydrolysis products on the high voltage stability of lithium-ion battery cells. ACS Appl. Mater. Interfaces 8, 30871–30878 (2016).
Lebens-Higgins, Z. W. et al. Evolution of the electrode–electrolyte interface of LiNi0.8Co0.15Al0.05O2 electrodes due to electrochemical and thermal stress. Chem. Mater. 30, 958–969 (2018).
Sharova, V. et al. Comparative study of imide-based Li salts as electrolyte additives for Li-ion batteries. J. Power Sources 375, 43–52 (2018).
Ensling, D., Stjerndahl, M., Nytén, A., Gustafsson, T. & Thomas, J. O. A comparative XPS surface study of Li2FeSiO4/C cycled with LiTFSI- and LiPF6-based electrolytes. J. Mater. Chem. 19, 82–88 (2009).
Quinlan, R. A., Lu, Y.-C., Shao-Horn, Y., Mansour, A. N. & Studies, X. P. S. of Surface chemistry changes of LiNi0.5Mn0.5O2 electrodes during high-voltage cycling. J. Electrochem. Soc. 160, A669–A677 (2013).
Baggetto, L., Dudney, N. J. & Veith, G. M. Surface chemistry of metal oxide coated lithium manganese nickel oxide thin film cathodes studied by XPS. Electrochim. Acta 90, 135–147 (2013).
Chapman, N., Borodin, O., Yoon, T., Nguyen, C. C. & Lucht, B. L. Spectroscopic and density functional theory characterization of common lithium salt solvates in carbonate electrolytes for lithium batteries. J. Phys. Chem. C. 121, 2135–2148 (2017).
Morita, M., Asai, Y., Yoshimoto, N., Ishikawa, M. & Raman, A. Spectroscopic study of organic electrolyte solutions based on binary solvent systems of ethylene carbonate with low viscosity solvents which dissolve different lithium salts. J. Chem. Soc. Faraday Trans. 94, 3451–3456 (1998).
Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004).
Liu, Zhi, Huang, S. & Wang, W. A refined force field for molecular simulation of imidazolium-based ionic liquids. J. Phys. Chem. B. 108, 12978–12989 (2004).
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: an N⊕ log (N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089–10092 (1993).
Martínez, L., Andrade, R., Birgin, E. G. & Martínez, J. M. PACKMOL: a package for building initial configurations for molecular dynamics simulations. J. Comput. Chem. 30, 2157–2164 (2009).
Ong, S. P. et al. Python materials genomics (pymatgen): a robust, open-source python library for materials analysis. Comp. Mater. Sci. 68, 314–319 (2013).
Frisch, M. J. et al. Gaussian (Gaussian, Inc., 2013).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B. 37, 785–789 (1988).
Acknowledgements
This work was supported as part of the NorthEast Center for Chemical Energy Storage, an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, Basic Energy Sciences under Award no. DE-SC0012583. Z.D. and S.P.O. acknowledge computational resources provided by the Triton Shared Computing Cluster at the University of California San Diego, and the Extreme Science and Engineering Discovery Environment supported by the National Science Foundation under grant no. ACI-1053575. Z. Du from Oak Ridge National Laboratory is acknowledged for kindly providing the casted NMC333 cathodes. Z.D. also acknowledges discussions with T. Hou, UC Berkeley, and help with MD from C. Jian, York University.
Author information
Authors and Affiliations
Contributions
B.W. and Y.-M.C. initiated and designed the research. B.W. conducted the experiments and electrochemical analysis. P.-C.T. assisted the nanofabrication with FIB. Z.D. and S.P.O. performed the MD simulations as well as DFT calculations. Z.W.L.-H. and L.F.J.P. assisted with XPS analysis. All authors contributed to writing the manuscript under the supervision of Y.-M.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–13, Tables 1–15, methods and refs. 1–4.
Rights and permissions
About this article
Cite this article
Wen, B., Deng, Z., Tsai, PC. et al. Ultrafast ion transport at a cathode–electrolyte interface and its strong dependence on salt solvation. Nat Energy 5, 578–586 (2020). https://doi.org/10.1038/s41560-020-0647-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-020-0647-0
This article is cited by
-
Force-chemical coupling analysis of nanocomposite anode during charging and discharging process
Acta Mechanica (2024)
-
Ultrathin positively charged electrode skin for durable anion-intercalation battery chemistries
Nature Communications (2023)
-
Degradation mechanism analysis of LiNi0.5Co0.2Mn0.3O2 single crystal cathode materials through machine learning
npj Computational Materials (2022)
-
Ab initio Interfacial Chemical Stability of Argyrodite Sulfide Electrolytes and Layered-Structure Cathodes in Solid-State Lithium Batteries
JOM (2022)