Abstract
The ongoing climate change is triggering plant community thermophilization. This selection process ought to shift community composition towards species adapted to warmer climates but may also lead to biotic homogenization. The link between thermophilization and homogenization and the community dynamics that drive them (colonization and extinction) remain unknown but is critical for understanding community responses under rapid environmental change. We used 14,167 pairs of plots to study shifts in plant community during 10 years of rising temperature in 80 forest ecoregions of France. We computed community mean thermal optimum (thermophilization) and Δβ-diversity (homogenization) for each ecoregion and partitioned these changes into extinction and colonization dynamics of cold- and warm-adapted species. Forest understorey communities thermophilized on average by 0.12 °C per decade and up to 0.20 °C per decade in warm ecoregions. This rate was entirely driven by extinction dynamics. Extinction of cold-adapted species was a driver of homogenization but it was compensated for by the colonization of rare species and the extinction of common species, resulting in the absence of an apparent homogenization trend. Here we show a dieback of present cold-adapted species rather than an adaptation of communities via the arrival of warm-adapted species, with a mutually cancelling effect on β-diversity. These results suggest that a future loss of biodiversity and delayed biotic homogenization should be considered.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
French National Forest Inventory data are freely distributed by IGN at https://inventaire-forestier.ign.fr. The dataset and the code used to reproduce our analysis can be downloaded from GitHub https://github.com/Jeremy-borderieux/Article_thermo_beta_part.git.
Code availability
The code and workflow to run and reproduce our analysis can be downloaded from GitHub https://github.com/Jeremy-borderieux/Article_thermo_beta_part.git.
References
Franklin, J., Serra-Diaz, J. M., Syphard, A. D. & Regan, H. M. Global change and terrestrial plant community dynamics. Proc. Natl Acad. Sci. USA 113, 3725–3734 (2016).
Lenoir, J. & Svenning, J.-C. Climate-related range shifts—a global multidimensional synthesis and new research directions. Ecography 38, 15–28 (2015).
Svenning, J.-C. & Sandel, B. Disequilibrium vegetation dynamics under future climate change. Am. J. Bot. 100, 1266–1286 (2013).
Sala, O. E. et al. Global biodiversity scenarios for the year 2100. Science 287, 1770–1774 (2000).
Reu, J. C., Catano, C. P., Spasojevic, M. J. & Myers, J. A. Beta diversity as a driver of forest biomass across spatial scales. Ecology 103, e3774 (2022).
Wang, S. et al. Biotic homogenization destabilizes ecosystem functioning by decreasing spatial asynchrony. Ecology https://doi.org/10.1002/ecy.3332 (2021).
De Frenne, P. et al. Microclimate moderates plant responses to macroclimate warming. Proc. Natl Acad. Sci. USA 110, 18561–18565 (2013).
De Frenne, P. et al. Light accelerates plant responses to warming. Nat. Plants 1, 15110 (2015).
Martin, G., Devictor, V., Motard, E., Machon, N. & Porcher, E. Short-term climate-induced change in French plant communities. Biol. Lett. 15, 20190280 (2019).
Richard, B. et al. The climatic debt is growing in the understorey of temperate forests: stand characteristics matter. Glob. Ecol. Biogeogr. https://doi.org/10.1111/geb.13312 (2021).
Bertrand, R. et al. Changes in plant community composition lag behind climate warming in lowland forests. Nature 479, 517–520 (2011).
Bertrand, R. et al. Ecological constraints increase the climatic debt in forests. Nat. Commun. 7, 12643 (2016).
Cholewińska, O., Adamowski, W. & Jaroszewicz, B. Homogenization of temperate mixed deciduous forests in Białowieża Forest: similar communities are becoming more similar. Forests 11, 545 (2020).
Olden, J. D. & Rooney, T. P. On defining and quantifying biotic homogenization. Glob. Ecol. Biogeogr. 15, 113–120 (2006).
Staude, I. R. et al. Directional turnover towards larger-ranged plants over time and across habitats. Ecol. Lett. 25, 466–482 (2022).
Gottfried, M. et al. Continent-wide response of mountain vegetation to climate change. Nat. Clim. Change https://doi.org/10.1038/nclimate1329 (2012).
Boulangeat, I., Gravel, D. & Thuiller, W. Accounting for dispersal and biotic interactions to disentangle the drivers of species distributions and their abundances: the role of dispersal and biotic interactions in explaining species distributions and abundances. Ecol. Lett. 15, 584–593 (2012).
Govaert, S. et al. Rapid thermophilization of understorey plant communities in a 9 year-long temperate forest experiment. J. Ecol. 109, 2434–2447 (2021).
Ozinga, W. A. et al. Dispersal failure contributes to plant losses in NW Europe. Ecol. Lett. 12, 66–74 (2009).
Leibold, M. A. et al. The metacommunity concept: a framework for multi-scale community ecology. Ecol. Lett. 7, 601–613 (2004).
Pérez‐Navarro, M. Á. et al. Extreme drought reduces climatic disequilibrium in dryland plant communities. Oikos 130, 680–690 (2021).
Bahn, M. & Körner, Ch. in Alpine Biodiversity in Europe (eds Nagy, L. et al.) 437–441 (Springer, 2003).
Steinbauer, M. J. et al. Accelerated increase in plant species richness on mountain summits is linked to warming. Nature 556, 231–234 (2018).
Tobias, N. & Monika, W. Does taxonomic homogenization imply functional homogenization in temperate forest herb layer communities? Plant Ecol. 213, 431–443 (2012).
Xu, W.-B. et al. Regional occupancy increases for widespread species but decreases for narrowly distributed species in metacommunity time series. Nat. Commun. 14, 1463 (2023).
Zwiener, V. P., Lira-Noriega, A., Grady, C. J., Padial, A. A. & Vitule, J. R. S. Climate change as a driver of biotic homogenization of woody plants in the Atlantic Forest. Glob. Ecol. Biogeogr. 27, 298–309 (2018).
Dietz, L. et al. Windstorm-induced canopy openings accelerate temperate forest adaptation to global warming. Glob. Ecol. Biogeogr. 29, 2067–2077 (2020).
Dupouey, J.-L., Sciama, D., Dambrine, E., Rameau, J.-C. & Koerner, W. La végétation des forêts anciennes. Rev. Forest. Française https://doi.org/10.4267/2042/4940 (2002).
Fischer, A., Lindner, M., Abs, C. & Lasch, P. Vegetation dynamics in central european forest ecosystems (near-natural as well as managed) after storm events. Folia Geobot. 37, 17–32 (2002).
Crisfield, V. E., Guillaume Blanchet, F., Raudsepp‐Hearne, C. & Gravel, D. How and why species are rare: towards an understanding of the ecological causes of rarity. Ecography https://doi.org/10.1111/ecog.07037 (2024).
Socolar, J. B., Gilroy, J. J., Kunin, W. E. & Edwards, D. P. How should beta-diversity inform biodiversity conservation? Trends Ecol. Evol. 31, 67–80 (2016).
Baeten, L. et al. Distinguishing between turnover and nestedness in the quantification of biotic homogenization. Biodivers. Conserv. 21, 1399–1409 (2012).
Gosselin, F. Putting floristic thermophilization in forests into a conservation biology perspective: beyond mean trait approaches. Ann. For. Sci. https://doi.org/10.1007/s13595-015-0526-1 (2016).
Tatsumi, S., Iritani, R. & Cadotte, M. W. Temporal changes in spatial variation: partitioning the extinction and colonisation components of beta diversity. Ecol. Lett. 24, 1063–1072 (2021).
Gégout, J.-C., Coudun, C., Bailly, G. & Jabiol, B. EcoPlant: a forest site database linking floristic data with soil and climate variables. J. Veg. Sci. 16, 257–260 (2005).
Dullinger, S. et al. Extinction debt of high-mountain plants under twenty-first-century climate change. Nat. Clim. Change 2, 619–622 (2012).
Engler, R. et al. 21st century climate change threatens mountain flora unequally across Europe: climate change impacts on mountain florae. Glob. Change Biol. 17, 2330–2341 (2011).
Kuhn, E. & Gégout, J. Highlighting declines of cold‐demanding plant species in lowlands under climate warming. Ecography 42, 36–44 (2019).
Sanczuk, P. et al. Competition mediates understorey species range shifts under climate change. J. Ecol. 110, 1813–1825 (2022).
Loarie, S. R. et al. The velocity of climate change. Nature 462, 1052–1055 (2009).
Serra-Diaz, J. M. et al. Bioclimatic velocity: the pace of species exposure to climate change. Divers. Distrib. 20, 169–180 (2014).
Lenoir, J., Gégout, J. C., Marquet, P. A., de Ruffray, P. & Brisse, H. A significant upward shift in plant species optimum elevation during the 20th century. Science 320, 1768–1771 (2008).
Rolland, C. Spatial and seasonal variations of air temperature lapse rates in alpine regions. J. Clim. 16, 1032–1046 (2003).
Franks, S. J., Weber, J. J. & Aitken, S. N. Evolutionary and plastic responses to climate change in terrestrial plant populations. Evol. Appl. 7, 123–139 (2014).
Kubisch, A., Degen, T., Hovestadt, T. & Poethke, H. J. Predicting range shifts under global change: the balance between local adaptation and dispersal. Ecography 36, 873–882 (2013).
Lavergne, S., Mouquet, N., Thuiller, W. & Ronce, O. Biodiversity and climate change: integrating evolutionary and ecological responses of species and communities. Annu. Rev. Ecol. Evol. Syst. 41, 321–350 (2010).
De Frenne, P. et al. Global buffering of temperatures under forest canopies. Nat. Ecol. Evol. 3, 744–749 (2019).
De Lombaerde, E. et al. Maintaining forest cover to enhance temperature buffering under future climate change. Sci. Total Environ. https://doi.org/10.1016/j.scitotenv.2021.151338 (2021).
Suggitt, A. J. et al. Extinction risk from climate change is reduced by microclimatic buffering. Nat. Clim. Change https://doi.org/10.1038/s41558-018-0231-9 (2018).
Zellweger, F. et al. Forest microclimate dynamics drive plant responses to warming. Science 368, 772–775 (2020).
Bergès, L., Pellissier, V., Avon, C., Verheyen, K. & Dupouey, J.-L. Unexpected long-range edge-to-forest interior environmental gradients. Landsc. Ecol. 28, 439–453 (2013).
Bodin, J. et al. Shifts of forest species along an elevational gradient in Southeast France: climate change or stand maturation? J. Veg. Sci. 24, 269–283 (2013).
Vittoz, P. & Engler, R. Seed dispersal distances: a typology based on dispersal modes and plant traits. Bot. Helv. 117, 109–124 (2007).
Gasperini, C. et al. Edge effects on the realised soil seed bank along microclimatic gradients in temperate European forests. Sci. Total Environ. 798, 149373 (2021).
Leibold, M. A. & Chase, J. M. Metacommunity Ecology (Princeton Univ. Press, 2017).
Devictor, V. et al. Differences in the climatic debts of birds and butterflies at a continental scale. Nat. Clim. Change 2, 121–124 (2012).
Jackson, S. T. & Sax, D. F. Balancing biodiversity in a changing environment: extinction debt, immigration credit and species turnover. Trends Ecol. Evol. 25, 153–160 (2010).
Landuyt, D. et al. The functional role of temperate forest understorey vegetation in a changing world. Glob. Change Biol. 25, 3625–3641 (2019).
Mori, A. S., Isbell, F. & Seidl, R. β-diversity, community assembly and ecosystem functioning. Trends Ecol. Evol. 33, 549–564 (2018).
Tatsumi, S., Iritani, R. & Cadotte, M. W. Partitioning the temporal changes in abundance-based beta diversity into loss and gain components. Methods Ecol. Evol. 13, 2042–2048 (2022).
Danneyrolles, V. et al. Scale-dependent changes in tree diversity over more than a century in eastern Canada: landscape diversification and regional homogenization. J. Ecol. 109, 273–283 (2021).
Heinrichs, S. & Schmidt, W. Biotic homogenization of herb layer composition between two contrasting beech forest communities on limestone over 50 years. Appl. Veg. Sci. 20, 271–281 (2017).
Merle, H., Garmendia, A., Hernández, H. & Ferriol, M. Vegetation change over a period of 46 years in a Mediterranean mountain massif (Penyagolosa, Spain). Appl. Veg. Sci. 23, 495–507 (2020).
Fiches Descriptives des Grandes Régions Ecologiques (GRECO) et des Sylvoécorégions (SER) (IGN, 2013); https://inventaire-forestier.ign.fr/spip.php?article773
Le Mémento de l’Inventaire Forestier (IGN, 2019); www.ign.fr/institut/publications/memento-linventaire-forestier
Piedallu, C. et al. Soil and climate differently impact NDVI patterns according to the season and the stand type. Sci. Total Environ. 651, 2874–2885 (2019).
Chytrý, M., Tichý, L., Hennekens, S. M. & Schaminée, J. H. J. Assessing vegetation change using vegetation-plot databases: a risky business. Appl. Veg. Sci. 17, 32–41 (2014).
Gargominy, O. TAXREF v13.0, Référentiel Taxonomique pour la France [Dataset] (UMS PatriNat, 2022); https://doi.org/10.15468/VQUEAM
Vangansbeke, P. et al. ClimPlant: realized climatic niches of vascular plants in European forest understoreys. Glob. Ecol. Biogeogr. 30, 1183–1190 (2021).
Whittaker, R. H. Vegetation of the Siskiyou Mountains, Oregon and California. Ecol. Monogr. 30, 279–338 (1960).
Baselga, A. Partitioning the turnover and nestedness components of beta diversity: partitioning beta diversity. Glob. Ecol. Biogeogr. 19, 134–143 (2010).
Braun-Blanquet, J. Plant Sociology: The Study of Plant Communities 1st edn (CAB, 1932); www.cabdirect.org/cabdirect/abstract/19331600801
Rey, D. & Neuhäuser, M. in International Encyclopedia of Statistical Science (ed. Lovric, M.) 1658–1659 (Springer, 2011).
Zuur, A. F., Ieno, E. N. & Elphick, C. S. A protocol for data exploration to avoid common statistical problems. Methods Ecol. Evol. 1, 3–14 (2010).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2019).
Dowle, M. & Srinivasan, A. data.table: Extension of ‘data.frame‘. R package version 1.14 (2020).
Wickham, H. Ggplot2. WIREs Comput. Stat. 3, 180–185 (2011).
Pebesma, E. Simple features for R: standardized support for spatial vector data. R. J. 10, 439–446 (2018).
Kassambara, A. ggpubr: ‘ggplot2’ based publication ready plots. R package version 3.4 https://CRAN.R-project.org/package=ggpubr (2023).
Microsoft & Weston, S. foreach: Provides foreach looping construct. R package version 1.5 https://CRAN.R-project.org/package=foreach (2022).
Dunnington, D. & Thorne, B. ggspatial: Spatial data framework for ggplot2. R package version 1.1 (2020).
Planet Dump (OpenStreetMap, 2017); https://planet.osm.org
Corporation, M. & Weston, S. doParallel: Foreach parallel adaptor for the ‘parallel’ package. R package version 1.0 https://CRAN.R-project.org/package=doParallel (2022).
Acknowledgements
We are grateful to the French Institute for Geographic and Forest Information (IGN) and their field technicians for providing the NFI data and a precise description of the forest ecoregions. We acknowledge the funding from the Labex Arbre. J.B. was funded by a joint AgroParisTech and Région Grand-Est grant (grant no. 19_GE8_01020p05035) and J.M.S.-D. was funded by the ANR-JCJC (Agence Nationale de la Recherche, jeunes chercheuses et jeunes chercheurs) SEEDFOR (ANR-21-CE32-0003). J.M.S.-D. acknowledges the support from NASA for UConn’s Ecological Modeling Institute (no. 80NSSC 22K0883).
Author information
Authors and Affiliations
Contributions
J.B., J.-C.G. and J.M.S.-D. conceived this article. J.B. was responsible for analysis, visualization, methodology and software. J.-C.G. and J.M.S.-D. undertook supervision and funding acquisition. J.B. wrote the manuscript with contributions from all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Shinichi Tatsumi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Histogram of the randomized thermophilization values of the 80 ecoregions.
Results of the 200 iterations of the random thermal optimum model (thermal optima randomly assigned to the species). In this figure, the runs are not averaged: the 80 ecoregions randomized 200 times are displayed. The average values of thermophilization, Δβ-diversity and their contribution of the original dataset are displayed.
Extended Data Fig. 2 Thermophilization and Δβ-diversity change in lowland, mountain and Mediterranean ecoregions.
Thermophilization (a) and Δβ-diversity (b) in lowland, mountain and Mediterranean ecoregion clusters (Extended Data Fig. 1). Lowland (8,271 pairs, 45 ecoregions), mountain (4,116 pairs, 29 ecoregions), Mediterranean (377 pairs, 6 ecoregions). Each dot represents the values of one of the 80 ecoregions (ntot = 80).
Extended Data Fig. 3 Subsequent partitioning, into ‘rare’ and ‘common’ components, of the Thermophilization and Δβ-diversity change.
Partitioning of the data presented in Fig. 3. The contributions to a) thermophilization (°C decade−1) and b) Δβ-diversity (unitless) were partitioned on the basis of species declining or increasing in occurrences, of their thermal optimum relatively to their ecoregion and whether these species were rare (baseline occurrences < 10% of the plots) or common (baseline occurrences > 10% of the plots). Each dot represents the values of one of the 80 ecoregions (ntot = 80). The dashed grey line delineates the colonization and extinction components. The mean of each component is displayed. White dot; mean value of the thermophilization null model. The statistical difference between the null model value and the original dataset, obtained with a two-sided Wilcoxon test, is also displayed: p < 0.05 (*), p < 0.01 (**), p < 0.001 (***). Exact P values are available in Supplementary Table 1. Boxes; 25th centile, median and 75th centile; whiskers, 1.5 times the interquartile range.
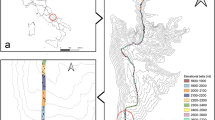
Extended Data Fig. 4 Map of the sampled ecoregions and display of the plots of one ecoregion.
(a) Map of the 86 forest ecoregions of France, with a coloured gradient representing the number of plot pairs. Three main biomes (lowland, Mediterranean, mountain) cluster different ecoregions delineated with bold black lines. The clusters without a label are mountain ecoregions. The zoomed ecoregion in (b) is outlined in red in (a). (b) Example of the plot pair sampling design, with NFI plot localization. Some plots may overlap. Green, forested areas. Basemap credits82.
Supplementary information
Supplementary Information
Supplementary Tables 1–4, Figs. 1 and 2 and equations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Borderieux, J., Gégout, JC. & Serra-Diaz, J.M. Extinction drives recent thermophilization but does not trigger homogenization in forest understorey. Nat Ecol Evol 8, 695–704 (2024). https://doi.org/10.1038/s41559-024-02362-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-024-02362-3