Abstract
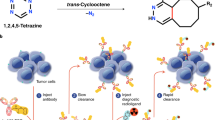
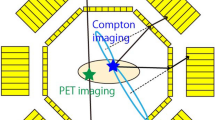
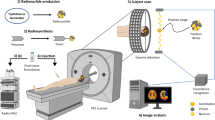
In conventional positron emission tomography (PET), only one radiotracer can be imaged at a time, because all PET isotopes produce the same two 511 keV annihilation photons. Here we describe an image reconstruction method for the simultaneous in vivo imaging of two PET tracers and thereby the independent quantification of two molecular signals. This method of multiplexed PET imaging leverages the 350–700 keV range to maximize the capture of 511 keV annihilation photons and prompt γ-ray emission in the same energy window, hence eliminating the need for energy discrimination during reconstruction or for signal separation beforehand. We used multiplexed PET to track, in mice with subcutaneous tumours, the biodistributions of intravenously injected [124I]I-trametinib and 2-deoxy-2-[18F]fluoro-d-glucose, [124I]I-trametinib and its nanoparticle carrier [89Zr]Zr-ferumoxytol, and the prostate-specific membrane antigen (PSMA) and infused PSMA-targeted chimaeric antigen receptor T cells after the systemic administration of [68Ga]Ga-PSMA-11 and [124I]I. Multiplexed PET provides more information depth, gives new uses to prompt γ-ray-emitting isotopes, reduces radiation burden by omitting the need for an additional computed-tomography scan and can be implemented on preclinical and clinical systems without any modifications in hardware or image acquisition software.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. Source Data and sinograms for the phantoms in Fig. 3 are available at https://doi.org/10.5281/zenodo.8034519 while figures are available from figshare with the identifier https://doi.org/10.6084/m9.figshare.21816069.
Code availability
The mPET code consists of several modules, requiring access to additional code considered proprietary to the PET-scanner manufacturers (although for PET/CT systems such as the Siemens Inveon, it can be made available in a compiled version). In addition, the software requires hands-on training for implementation, which can be made available on reasonable request. The Monte Carlo code, MCGPU-PET, can be accessed via Github at https://github.com/DIDSR/MCGPU-PET, whereas code for reconstructing doubles and triples can also be accessed via https://github.com/jlherraiz/GFIRST. Isotope separation is determined from phantom studies performed on each scanner and isotope pair used.
References
DeBerardinis, R. J. & Chandel, N. S. We need to talk about the Warburg effect. Nat. Metab. 2, 127–129 (2020).
Black, N. F., McJames, S. & Kadrmas, D. J. Rapid multi-tracer PET tumor imaging with F-FDG and secondary shorter-lived tracers. IEEE Trans. Nucl. Sci. 56, 2750–2758 (2009).
Marti-Climent, J. M. et al. Effective dose estimation for oncological and neurological PET/CT procedures. EJNMMI Res. 7, 37 (2017).
Farber, D. L. Tissues, not blood, are where immune cells function. Nature 593, 506–509 (2021).
Heinzmann, K., Carter, L. M., Lewis, J. S. & Aboagye, E. O. Multiplexed imaging for diagnosis and therapy. Nat. Biomed. Eng. 1, 697–713 (2017).
Andreou, C., Weissleder, R. & Kircher, M. F. Multiplexed imaging in oncology. Nat. Biomed. Eng. 6, 527–540 (2022).
Zanzonico, P. Principles of nuclear medicine imaging: planar, SPECT, PET, multi-modality, and autoradiography systems. Radiat. Res. 177, 349–364 (2012).
Ko, J. et al. Spatiotemporal multiplexed immunofluorescence imaging of living cells and tissues with bioorthogonal cycling of fluorescent probes. Nat. Biotechnol. 40, 1654–1662 (2022).
Moroz, M. A. et al. Introducing a new reporter gene, membrane-anchored Cypridina luciferase, for multiplex bioluminescence imaging. Mol. Ther. Oncolytics 21, 15–22 (2021).
Yagishita, A. et al. Simultaneous visualization of multiple radionuclides in vivo. Nat. Biomed. Eng. 6, 640–647 (2022).
Yang, M. et al. Multiplexed PET probes for imaging breast cancer early response to VEGF121/rGel treatment. Mol. Pharmaceutics 8, 621–628 (2011).
Bell, C. et al. Dual acquisition of18F-FMISO and18F-FDOPA. Phys. Med. Biol. 59, 3925–3949 (2014).
Andreyev, A. & Celler, A. Dual-isotope PET using positron-gamma emitters. Phys. Med. Biol. 56, 4539–4556 (2011).
Herraiz, J. L. et al. GPU-based fast iterative reconstruction of fully 3-D PET sinograms. IEEE Trans. Nucl. Sci. 58, 2257–2263 (2011).
Carter, L. M. et al. The impact of positron range on PET resolution, evaluated with phantoms and PHITS Monte Carlo simulations for conventional and non-conventional radionuclides. Mol. Imaging Biol. https://doi.org/10.1007/s11307-019-01337-2 (2019).
Cal-González, J. et al. Positron range effects in high resolution 3D PET imaging. In 2009 IEEE Nuclear Science Symposium Conference Record (NSS/MIC), 2788-2791 https://doi.org/10.1109/NSSMIC.2009.5401950 (2009).
Lubberink, M. & Herzog, H. Quantitative imaging of 124I and 86Y with PET. Eur. J. Nucl. Med. Mol. Imaging 38, S10–S18 (2011).
Kull, T., Ruckgaber, J., Weller, R., Reske, S. & Glatting, G. Quantitative imaging of yttrium-86 PET with the ECAT EXACT HR+ in 2D mode. Cancer Biother. Radiopharm. 19, 482–490 (2004).
Lubberink, M., Schneider, H., Bergström, M. & Lundqvist, H. Quantitative imaging and correction for cascade gamma radiation of 76Br with 2D and 3D PET. Phys. Med. Biol. 47, 3519–3534 (2002).
Beattie, B. J., Finn, R. D., Rowland, D. J. & Pentlow, K. S. Quantitative imaging of bromine-76 and yttrium-86 with PET: a method for the removal of spurious activity introduced by cascade gamma rays. Med. Phys. 30, 2410–2423 (2003).
Watson, C. C., Newport, D. & Casey, M. E. In Three-Dimensional Image Reconstruction in Radiology and Nuclear Medicine (eds Grangeat, P. & Amans, J.-L.) 255–268 (Springer, 1996).
Sanaat, A. et al. Deep learning-assisted ultra-fast/low-dose whole-body PET/CT imaging. Eur. J. Nucl. Med. Mol. Imaging 48, 2405–2415 (2021).
Jung, K. O. et al. Whole-body tracking of single cells via positron emission tomography. Nat. Biomed. Eng. 4, 835–844 (2020).
Cal-Gonzalez, J. et al. Simulation of triple coincidences in PET. Phys. Med. Biol. 60, 117–136 (2015).
Lage, E. et al. Recovery and normalization of triple coincidences in PET. Med. Phys. 42, 1398–1410 (2015).
Surti, S., Scheuermann, R. & Karp, J. S. Correction technique for cascade gammas in I-124 imaging on a fully-3D, time-of-flight PET scanner. IEEE Trans. Nucl. Sci. 56, 653–660 (2009).
Preylowski, V. et al. Is the image quality of I-124-PET impaired by an automatic correction of prompt gammas? PLoS ONE 8, e71729 (2013).
Surti, S. & Scheuermann, J. In Physics of PET and SPECT Imaging Ch. Data corrections and quantitative PET, 24 (CRC, 2017).
Ilisie, V. et al. High resolution and sensitivity gamma camera with active septa. A first Monte Carlo study. Sci. Rep. 9, 18431 (2019).
Andreyev, A., Sitek, A. & Celler, A. EM reconstruction of dual isotope PET using staggered injections and prompt gamma positron emitters. Med. Phys. 41, 022501 (2014).
Fukuchi, T. et al. Image reconstruction method for dual-isotope positron emission tomography. J. Instrum. 16, P01035–P01035 (2021).
Beekman, F. J., Kamphuis, C., Koustoulidou, S., Ramakers, R. M. & Goorden, M. C. Positron range-free and multi-isotope tomography of positron emitters. Phys. Med. Biol. 66, 065011 (2021).
Kaittanis, C. et al. Environment-responsive nanophores for therapy and treatment monitoring via molecular MRI quenching. Nat. Commun. 5, 3384 (2014).
Volpe, A. et al. Spatiotemporal PET imaging reveals differences in CAR-T tumor retention in triple-negative breast cancer models. Mol. Ther. 28, 2271–2285 (2020).
Pratt, E. C. et al. Synthesis of novel PET tracer 124I-trametinib for MAPK/ERK kinase distribution and resistance monitoring. J. Nucl. Med. https://doi.org/10.2967/jnumed.120.241901 (2020).
Parmenter, T. J. et al. Response of BRAF-mutant melanoma to BRAF inhibition is mediated by a network of transcriptional regulators of glycolysis. Cancer Discov. 4, 423–433 (2014).
Hrkach, J. & Langer, R. From micro to nano: evolution and impact of drug delivery in treating disease. Drug Deliv. Transl. Res. 10, 567–570 (2020).
Thorek, D. L. et al. Non-invasive mapping of deep-tissue lymph nodes in live animals using a multimodal PET/MRI nanoparticle. Nat. Commun. 5, 3097 (2014).
Yuan, H. et al. Heat-induced radiolabeling and fluorescence labeling of Feraheme nanoparticles for PET/SPECT imaging and flow cytometry. Nat. Protoc. 13, 392–412 (2018).
Boros, E., Bowen, A. M., Josephson, L., Vasdev, N. & Holland, J. P. Chelate-free metal ion binding and heat-induced radiolabeling of iron oxide nanoparticles. Chem. Sci. 6, 225–236 (2015).
MEKINIST (Trametinib) (Package Insert). (GlaxoSmithKline, 2019).
Sarrett, S. M. et al. Inverse electron demand Diels–Alder click chemistry for pretargeted PET imaging and radioimmunotherapy. Nat. Protoc. 16, 3348–3381 (2021).
Zeglis, B. M. et al. Modular strategy for the construction of radiometalated antibodies for positron emission tomography based on inverse electron demand Diels–Alder click chemistry. Bioconjug. Chem. 22, 2048–2059 (2011).
Tavare, R. et al. Engineered antibody fragments for immuno-PET imaging of endogenous CD8+ T cells in vivo. Proc. Natl Acad. Sci. USA 111, 1108–1113 (2014).
Tavare, R. et al. An effective immuno-PET imaging method to monitor CD8-dependent responses to immunotherapy. Cancer Res. 76, 73–82 (2016).
Miller, B. C. et al. Subsets of exhausted CD8(+) T cells differentially mediate tumor control and respond to checkpoint blockade. Nat. Immunol. 20, 326–336 (2019).
Kallies, A., Zehn, D. & Utzschneider, D. T. Precursor exhausted T cells: key to successful immunotherapy? Nat. Rev. Immunol. 20, 128–136 (2020).
Yu, A. R., Kim, H.-J., Lim, S. M. & Kim, J. S. Comparison of imaging characteristics of 124I PET for determination of optimal energy window on the Siemens Inveon PET. BioMed. Res. Int. 2016, 3067123 (2016).
Choi, H. et al. Transcriptome analysis of individual stromal cell populations identifies stroma–tumor crosstalk in mouse lung cancer model. Cell Rep. 10, 1187–1201 (2015).
Ebenhan, T. et al. Development of a single vial kit solution for radiolabeling of 68Ga-DKFZ-PSMA-11 and its performance in prostate cancer patients. Molecules 20, 14860–14878 (2015).
Man, F. et al. In vivo PET tracking of 89Zr-labeled Vγ9Vδ2 T cells to mouse xenograft breast tumors activated with liposomal alendronate. Mol. Ther. 27, 219–229 (2019).
Man, F., Khan, A. A., Carrascal-Minino, A., Blower, P. J. & de Rosales, R. T. M. A kit formulation for the preparation of [89Zr]Zr(oxinate)4 for PET cell tracking: white blood cell labelling and comparison with [111In]In(oxinate)3. Nucl. Med. Biol. 90-91, 31–40 (2020).
Acknowledgements
We thank D. Bauer of the Lewis Lab for their timely production of [68Ga]Ga-PSMA-11, and V. Longo and P. Zanzonico of the Small Animal Imaging Core (MSKCC) for their support maintaining the Inveon PET/CT. We also thank E. Fung and M. Du of the Citigroup Biomedical Imaging Center (Weill Cornell) for their availability and operation of the Biograph mCT clinical PET/CT and M. Conti from Siemens for their direction on how to acquire 64-bit data with the mCT. In addition, we thank E. Lage, V. Parot, S. R. Dave, S. C. Moore and the Madrid-MIT m+Vision Consortium for their help with the initial steps in the development of the mPET method. This work was supported in parts by the following grants: National Institutes of Health R01 CA215700 and R01 EB033000 (to J.G.), R01 CA220524-01A1 (to V.P.), R01 CA204924 (to V.P.), R21 CA250478 (to V.P.), S10 OD016207-01 (to P. Zanzonico, MSKCC), and P30 CA08748 (to S. M. Vickers MSKCC). A.V. is supported by The Center for Experimental Immuno-Oncology Fellowship Award (FP00001443, Memorial Sloan Kettering Cancer Center). E.C.P. is currently supported by the National Institutes of Health F32 CA268912-01. J.L.H. is currently supported by the Spanish Ministry of Science and Innovation (MCIN) (PID2021-126998OB-I00, PDC2022-133057-I00/AEI/10.13039/501100011033/ Unión Europea Next GenerationEU/PRTR). J.M.U. is currently supported by the Spanish Ministry of Science and Innovation (MCIN) (PID2021-126998OB-I00).
Author information
Authors and Affiliations
Contributions
E.C.P., A.L.M., A.V., M.J.C. and L.M.C. conducted and planned experiments, analysed data and wrote the manuscript. V.M., N.P., V.P, J.M.U., J.G. and J.L.H. designed experiments, analysed data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Huafeng Liu, Bertrand Tavitian and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Extended mPET imaging of nanoparticle biodistribution through 96 hours.
Including data from Fig. 5 and extending through 96 hours. No change in biodistribution seen by standard reconstruction or mPET between 24 and 96 hours. %IA/CC represents percent injected activity per cubic centimetre.
Extended Data Fig. 2 Characterization of the tricistronic CAR T-cell PET-reporter system.
Gene map of PET and reporter genes for CAR T cells containing the sodium Iodide symporter (hNIS), PSMA targeting scFv (P28z), and a BLI reporter system (exCLuc) to produce the CAR-T-cell hNIS.P28z.exCLuc. b, Flow plot of PC3 cells engineered lacking PSMA. c, PSMA-positive engineered cells. d, Flow plot of CAR T cells expressing the anti-PSMA-scFv. e, In vitro activation of the BLI reporter in tricistronic CAR T cells shows functional incorporation of the genes. f, Addition of 124I to wild-type or tricistronic CAR T cells shows an increase with only tricistronic CAR T cells, with mild blocking with sodium perchlorate, confirming the specific uptake of 124I through hNIS. g, Confocal imaging of wild-type and hNIS tricistronic CAR-T cells for nuclear staining, hNIS and WGA (wheat germ agglutinin). Scale bar, 5 µm. h, In vitro cytotoxicity of PSMA targeting CAR-T cells reduces cell population in PSMA-positive co-cultures, whereas PSMA-null cells were unaffected through 48 hours by the co culture with CAR-T cells. I, In vivo delivery of CAR T cells targeting PSMA-positive tumours led to reduced tumour growth in PSMA-positive tumours, whereas PSMA-negative tumours continued to grow. In e, f, h, I, the lines denote the mean with error bars representing the s.e.m. b-d, Representative flow-cytometry contour quadrant plot to show cell-intensity characteristics. The gating scheme can be found in Supplementary Fig. 11. e, Box plot representing min and max values, with the middle line as the mean. In f, h, n = 3 technical replicates per condition. In i, n = 5 mice per arm. In e,h, the P values are significant, using a multiple-comparison unpaired t-test and assuming the same s.d. in the population. %IA/CC represents percent injected activity per cubic centimetre.
Extended Data Fig. 3 Alternative ex vivo labelling of CAR T-cells with [89Zr]Zr-Oxine and PSMA imaging with [86Y]Y-DOTA-PSMA.
[86Y]Y-DOTA-PSMA / [89Zr]Zr-oxine CAR T-cell mPET was found to have uptake of both PSMA-11 and CAR T -cell tracers in the PSMA-positive tumour (left), whereas minor to no activity was observed in both tracers for the PSMA-null tumour (right). Distribution of the [86Y]Y-DOTA-PSMA tracer can be seen in the PSMA-positive tumour, in the ocular cavity where injected, and in the bladder during excretion. [89Zr]Zr-oxine CAR T-cells were observed in the PSMA-positive tumour as well as in the liver and bone. Images were calibrated to a maximal 2.5 %IA/CC for [86Y]Y-DOTA-PSMA and 7.5 %IA/CC for [89Zr]Zr-oxine CAR T-cells. We used one mouse. %IA/CC represents percent injected activity per cubic centimetre.
Extended Data Fig. 4 mPET for developing immunoPET detection of T-cell exhaustion.
Mice bearing HKP1 lung tumours were monitored for terminal and progenitor T-cell exhaustion using antibodies against CD39 or Ly108 during disease progression. BLI of lung tumour burden was measured prior to injection, with representative CT slice in thoracic cavity showing potential branch occlusions (bright spots off alveolar bifurcation). Ly108, a marker of effector T cells, decreased with disease burden whereas CD39, a marker of severely exhausted T cells, increased, according to flow-cytometry data. mPET imaging with [89Zr]Zr-DFO-CD39 and [124I]I-Ly108 at day 7 and day 14 with three levels of tumour burden. At day 7 no discernible difference between low, medium or high tumour burden in mice is seen with either [89Zr]Zr-DFO-CD39 or [124I]I-Ly108. By day 14, BLI shows an appreciable increase in tumour burden in all mice, with tumour occlusions seen on CT. By mPET there was a general increase in [89Zr]Zr-DFO-CD39 and [124I]I-Ly108 uptake in the lungs compared to the day-7 group. Less lung uptake was visible with [124I]I-Ly108, with increasing tumour burden in the day-14 imaged mice. mPET again could separate two radiotracers in vivo and could be used with further radiotracer engineering to define T-cell exhaustion. N = 3 mice imaged per week (n = 6 total cohort), with each mouse receiving simultaneously [89Zr]Zr-DFO-CD39 and [124I]I-Ly108 by intravenous injection 48 hours prior to mPET. %IA/CC represents percent injected activity per cubic centimetre.
Supplementary information
Supplementary Information
Supplementary Figures, Tables and References.
Supplementary Video 1
Rotating images with combined and separated PET/CT scans from a representative mouse (from Fig. 6), windowed at 0–5% injected dose per gram for both radiotracers, to identify PSMA-positive and CAR T-cell positive tumours.
Supplementary Video 2
Rotating images analogous to those in Supplementary Video 1, calibrated to 0–30% injected dose per gram, to show the majority of the radiotracer distribution and elimination.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pratt, E.C., Lopez-Montes, A., Volpe, A. et al. Simultaneous quantitative imaging of two PET radiotracers via the detection of positron–electron annihilation and prompt gamma emissions. Nat. Biomed. Eng 7, 1028–1039 (2023). https://doi.org/10.1038/s41551-023-01060-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-023-01060-y
This article is cited by
-
Extracting value from total-body PET/CT image data - the emerging role of artificial intelligence
Cancer Imaging (2024)
-
Signal separation of simultaneous dual-tracer PET imaging based on global spatial information and channel attention
EJNMMI Physics (2024)
-
Uncovering atherosclerotic cardiovascular disease by PET imaging
Nature Reviews Cardiology (2024)
-
Highlight selection of radiochemistry and radiopharmacy developments by editorial board
EJNMMI Radiopharmacy and Chemistry (2024)
-
Molecular imaging for better theranostics
European Journal of Nuclear Medicine and Molecular Imaging (2023)