Abstract
In the ATEMPT trial, adjuvant trastuzumab emtansine (T-DM1) compared to paclitaxel plus trastuzumab (TH) for stage I HER2-positive breast cancer improved patient-reported outcomes (PROs), while maintaining excellent disease outcomes. We report treatment discontinuation and use multivariable models to compare, patient-reported toxicity and quality-of-life (QOL) by age (≤50, >50) and treatment arm at 18 months post-enrollment among 366 eligible participants randomized in a 3:1 ratio to T-DM1 or TH. T-DM1 discontinuation was higher among women >50 vs. ≤50 (23% vs. 9%, p = 0.003, Fisher’s Exact test) with 4%, 8%, and 17% of older patients discontinuing treatment by 3, 6, and 9 months, respectively. Superior QOL with T-DM1 vs. TH was observed among women ≤50 with estimated mean difference of 6.48 (95% confidence interval (CI) 0.51–12.46) and driven by better social/family well-being and breast cancer-specific sub-scores. Among women >50, T-DM1 was associated with superior physical well-being and less activity impairment, with no differences in global QOL. Older women had decreased neuropathy with T-DM1 vs. TH. De-escalated treatment regimens for HER2 positive breast cancer may have age-varying impact on treatment tolerance, toxicities and subsequent QOL, which should be considered when selecting therapy options.
Clinical Trial Registration: ClinicalTrials.gov, NCT01853748
Similar content being viewed by others
Introduction
Trastuzumab is a well-established treatment for human epidermal growth factor receptor 2 (HER2)-positive early breast cancers. Given its high efficacy, recent efforts have concentrated on de-escalation of historic multi-agent chemotherapy protocols to safer and shorter regimens preserving previous achievements in long-term survival, while improving short and long-term quality of life (QOL)1. Progress is most evident for stage I HER2-positive breast cancers, with the single-arm APT trial showing excellent long-term outcomes with adjuvant paclitaxel plus trastuzumab (TH), omitting doxorubicin and cyclophosphamide2,3.
To further improve QOL outcomes in these patients, we conducted the ATEMPT trial, a multicenter, investigator-initiated randomized phase II study comparing a year of adjuvant T-DM1 (trastuzumab emtansine) to TH for toxicity and establishing the disease-free survival for one year of adjuvant T-DM14. While T-DM1 was associated with excellent 3-year invasive disease-free survival (iDFS, 97.8% [95% confidence interval (CI), 96.3–99.3]), the co-primary outcome, a prospectively defined composite outcome including clinically relevant toxicities seen with either treatment, was equivalent (46% T-DM1 vs. 47% TH, p = 0.83). T-DM1 was associated with a high rate of treatment discontinuation due to adverse events (17%); however, adverse event profiles, assessed by patient-reported outcomes (PROs), revealed better QOL, lower risk of neuropathy and superior work productivity with T-DM1 vs. TH.
While the ATEMPT trial supports the use of T-DM1 as a potential adjuvant systemic therapy in stage I HER2 + breast cancers, it remains unclear which patients stand to benefit from this de-escalation5. Patient age is an acknowledged factor in breast cancer therapy decision-making, in some instances driving over-treatment of younger patients and undertreatment of older patients6. Age may also be associated with development of negative physical and emotional sequelae following breast cancer. Young survivors are consistently found to be at higher risk for adverse physical and psychological effects which may impair their QOL for years following diagnosis7,8,9,10. When comparing age groups, several studies show worse QOL and increased symptom burden in younger survivors, primarily in early years post-diagnosis11,12,13,14. However, QOL deterioration is also observed in older populations, particularly those with comorbidities less common among younger women15.
Given these considerations, the aim of this unplanned post-hoc analysis is to compare rates of treatment discontinuation, and patient-reported QOL and toxicities between younger and older women in ATEMPT at 18 months post-enrollment. This timepoint, 6 months after completion of all protocol therapy, provides important information regarding women’s experience as they transition to breast cancer survivorship care following one year of adjuvant therapy.
Results
Patient characteristics
Of 512 participants recruited, 497 initiated study treatment. Following exclusion of participants without a baseline (n = 28) or 18-month assessment (n = 99), and male participants (n = 4), 366 patients were included in this analysis (Fig. 1). Among included patients, 34% (n = 124) were ≤50 years and 66% (n = 242) were >50 with an equal distribution observed for excluded patients (34 and 66%, respectively). Additional characteristics were similar for included and excluded patients (see Supplementary Table 1 in the Supplementary File).
In the analytic cohort (N = 366), overall median age was 56.69 (range 23.2–85.9), 45.37 (23.2–50.9) in women ≤50 and 61.13 (51.2–85.9) in women >50 (Table 1). Treatment distribution was similar and consistent with the 3:1 allocation, with 75% of women ≤50 and 79% women >50 randomized to T-DM1 (p = 0.428, Fisher’s Exact test). Younger women were more commonly premenopausal at enrollment (86% vs. 11%, p < 0.001, Fisher’s Exact test). Tumor characteristics were similar; however, younger women more frequently underwent mastectomy (53% vs. 28%, p < 0.001, Fisher’s Exact test) and accordingly received less adjuvant radiotherapy (50% vs. 73%, p < 0.001, Fisher’s Exact test). Among patients with hormone receptor-positive (≥1% estrogen receptor expression) disease, endocrine therapy utilization at 18 months was similar between age groups (≤50: 87% vs. >50: 83%, p = 0.385, Fisher’s Exact test) and between arms (T-DM1: 85% vs. TH: 83%, p = 0.840, Fisher’s Exact test).
Treatment discontinuation and dose reduction
Discontinuations of all protocol therapy were 6 and 18% for TH and T-DM1, respectively. T-DM1 discontinuation was significantly higher among women >50 vs. ≤50 (23% vs. 9%, p = 0.003, Fisher’s Exact test) with 4%, 8%, and 17% of older patients discontinuing treatment by 3, 6, and 9 months, respectively (Table 2). Similarly, different rates were observed in extreme age groups: ≤40 years (5%, 1/19), ≥70 years (23%, 6/26). Time to discontinuation was significantly shorter for older women vs. younger women (p = 0.002, Log-rank test, Fig. 2a) and for T-DM1 vs. TH (p = 0.007, Log-rank test, Fig. 2b). Older women receiving T-DM1 were at particular risk for discontinuation, while discontinuation for younger women receiving T-DM1 and both age groups receiving TH, was comparable (p < 0.001, Log-rank test, Fig. 2c). Following T-DM1 discontinuation, 25% (2/8) of younger and 45% (20/44) of older women switched to trastuzumab to complete a year of treatment.
Toxicity was the primary reason for T-DM1 discontinuation and was higher among older women (18% vs. 8%). In both age groups approximately half of discontinuations were protocol-mandated and half based on the treating physician’s decision (Table 2). Among older women, the most common toxicities for T-DM1 discontinuation were elevated liver enzymes or bilirubin (29%), neuropathy (17%), and thrombocytopenia (17%). Discontinuations due to cardiotoxicity were infrequent (n = 2) and limited to the older subgroup.
T-DM1 dose reductions occurred in 18% of women included in this analysis and were more common in older compared to younger women, (20%, 38/128 vs. 14%, 13/93, respectively, p = 0.011, Fisher’s Exact test).
Patient-reported outcomes
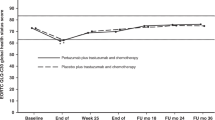
PRO scores at baseline and 18-months are summarized by arm and age group (Table 3). In multivariable analysis, better 18-month Functional Assessment of Cancer Therapy-Breast Cancer (FACT-B)16 total score was associated with better baseline FACT-B total score (estimated mean difference 0.73, p < 0.001, linear regression), but not with age or treatment arm (Table 4). Independent associations between 18-month FACT-B and age or arm were not found; however, an interaction between the two was observed (p = 0.037, linear regression): among women ≤50, treatment with T-DM1 (vs. TH) was associated with better 18-month FACT-B total score with an estimated mean difference of 6.48 (95% CI 0.51–12.46), approaching the minimally important difference (MID) threshold of 7–8 points17. Additionally, within the T-DM1 group, younger age was associated with better adjusted 18-month FACT-B total scores than older age (estimated mean difference in score between age ≤50 and age >50, 4.12; 95% CI 0.32–7.92). We performed a sensitivity analysis replacing the dichotomized age variable with baseline menopausal status (premenopausal or postmenopausal). In contrast to the primary model which showed a significant interaction between age group and treatment arm, in the sensitivity model, the interaction between baseline menopausal status and arm was not significant (p = 0.993, linear regression).
Table 5 and Supplementary Fig. 1 (in the Supplementary File) list adjusted mean differences in 18-month PROs for age groups and treatment arms (interaction). When controlling for baseline values of the outcome measure and other covariates, higher 18-month FACT-B total scores among younger women treated with T-DM1 vs. TH were driven by differences in social/family well-being (SWB) (estimated mean difference in score between T-DM1 and TH, 2.61; 95% CI 0.64–4.58) and breast cancer subscale (BCS) (estimated mean difference in score between T-DM1 and TH, 1.92; 95% CI 0.05–3.79) sub-scores. T-DM1 was significantly associated with better physical well-being (PWB) scores vs. TH in women >50 (estimated mean difference in score between T-DM1 and TH, 1.43; 95% CI 0.26–2.60). A similar non-significant point estimate was found for women ≤50 (estimated mean difference in score between T-DM1 and TH, 1.35; 95% CI −0.15–2.86). All significant inter-group differences met or approached MID threshold (2–3 points for BCS, 1–3 points for PWB)17,18.
Adjusted 18-month Rotterdam Symptom Checklist (RSCL)19,20 scores were comparable between age groups and arms; only activity level was significantly worse in younger vs. older women treated with T-DM1 (estimated mean difference in score between age ≤50 and age >50, 1.10; 0.28–1.93), although a similar point estimate was seen after TH (1.06, −0.42–2.53).
Using the 18-month Work Productivity and Activity Impairment Questionnaire: Specific Health Problem (WPAI:SHP)21, among women >50 years, T-DM1 vs. TH was associated with less activity impairment due to breast cancer (estimated mean difference in score between T-DM1 and TH, −6.53; 95% CI −12.79 to −0.28). No additional differences in mean WPAI:SHP scores were observed.
Adjusted odds of alopecia at 18 months, as reported on the Alopecia Patient Assessment (APA)22, did not significantly differ by age or arm. Using the Patient Neurotoxicity Questionnaire (PNQ)23, adjusted odds of 18-month residual moderate, moderate-severe or severe neuropathy were significantly lower with T-DM1 vs. TH among women >50 (odds ratio [OR] 0.33, 95% CI 0.16–0.68) with a trend for reduction among women ≤50 (OR 0.32, 95% CI 0.10–1.03).
Discussion
In light of favorable disease-related outcomes seen in contemporary trials for HER2-positive early breast cancer, ongoing efforts increasingly emphasize treatment de-escalation as a means of optimizing health-related quality of life (HRQOL) while sustaining treatment efficacy. In ATEMPT, adjuvant T-DM1 was associated with superior overall HRQOL, lower risk of neuropathy and superior work productivity, while maintaining excellent 3-year iDFS in patients with stage I HER2-positive breast cancer4. Analogous findings were reported in the KAITLIN trial, comparing similar regimens (plus pertuzumab) for stage II-III disease though following anthracycline-based chemotherapy24. Our current analysis shows that younger women, while opting for more aggressive surgery, and more often completing protocol therapy than older women, report larger HRQOL gains at 18 months with T-DM1 vs. TH, with differences within or approaching the range of clinical relevance17.
Multiple studies have identified a distinct and often more severe impact of breast cancer on younger survivors’ HRQOL and emotional well-being7,8,9,13. In a systematic review comparing younger (≤50) to older women (>50), HRQOL was more severely compromised in younger women, with greater deterioration noted for mental health as opposed to physical functioning domains9. In a recent longitudinal report, a steeper drop in HRQOL was observed among younger (≤50) vs. older (>50) survivors during the first three years post-diagnosis, and although HRQOL improved thereafter, at 10 years it remained below the general population level14. Our findings, showing better 18-month HRQOL particularly in young women treated with T-DM1 vs. TH, suggest that a modern, de-escalated chemotherapy approach may temper these effects on young women’s HRQOL. Additionally, complementary to prior studies, this improvement was driven by better SWB and BCS sub-scores, (including items focused on body image and sexuality) rather than PWB.
It is uncertain why T-DM1 led to superior 18-month HRQOL in young women. We previously showed that during the first 12 weeks of treatment, T-DM1 vs. TH was associated with less missed work time and work/activity impairment, and lower rates of alopecia and neuropathy4. By 18 months, these differences attenuated, although a lower risk of neuropathy following T-DM1 persists, regardless of age. Specific to younger women, treatment-related menopause is a toxicity with more long-term effects, which may in part explain our findings. In a preplanned sub-study, among premenopausal women enrolled to ATEMPT, 18-month chemotherapy-related amenorrhea was significantly lower with T-DM1 vs. TH (24% vs. 50%)25. Treatment-related menopause is associated with physiologic symptoms such as night sweats, hot flashes, vaginal dryness, and weight gain, which can adversely affect patients’ psychosocial QOL26,27. In younger premenopausal women, preserved ovarian function also contributes to fertility preservation, an important issue for many young patients28. In a sensitivity model however, after replacing age with baseline menopausal status, we did not replicate the significant interaction observed between age group and treatment arm.
Over half of women ≤50 treated for stage I breast cancers in ATEMPT underwent mastectomy, nearly twice the rate observed for women >50. Additionally, half of mastectomies in younger women were bilateral. These observations conform with national trends showing increasing rates of mastectomy, and particularly bilateral mastectomy, with steeper increases in younger patients and those with node-negative tumors ≤2 cm29. Compared to breast-conserving surgery, mastectomy with implant reconstruction is associated with inferior breast satisfaction, psychosocial well-being scores, and sexual well-being scores, even when restricting to stage I cancers30. Given the extremely low rates of locoregional recurrence associated with HER2-positive disease following adequate anti-HER2 therapy, less aggressive surgery may serve as an additional means to retain QOL31.
Among older women, we did not observe a significant difference in 18-month global HRQOL between arms, although T-DM1 was associated with better physical well-being, less activity impairment and lower odds of neuropathy. This may be partially related to increased toxicity and higher rates of T-DM1 discontinuation in older women, although we corrected for these in multivariable analyses. Additionally, our study was underpowered to examine differential treatment effects on HRQOL in women at extremes of age (≥65–70) and at higher risk of developing chemotherapy toxicity6,32. The ATEMPT 2.0 trial (NCT04893109) is evaluating whether six cycles of T-DM1 followed by trastuzumab can decrease toxicity while maintaining efficacy and will compare toxicities of this regimen to TH in patients with stage I HER2-positive breast cancer.
The current study’s strengths include its prospective nature and high-quality data captured within the setting of a multicenter clinical trial. We applied an age stratification (≤50, >50) commonly used in the study of breast cancer, facilitating comparisons to prior studies, but limiting our ability to comment on women at age extremes, primarily the elderly. However, the care of elderly patients can be complicated by geriatric factors and comorbidities (not captured within our data), and thus a clinical trial population may not be representative6. Safety and efficacy of adjuvant T-DM1 in older patients (≥60 years) with stage I-III HER2-positive breast cancer is being evaluated in the ATOP trial (NCT03587740). Generalizability of our findings may also be limited by the small number of minority participants. Racial/ethnic variations in HRQOL after breast cancer have been described, although they may be less evident in younger women due to their overall worse HRQOL33. Lastly, although baseline characteristics, including distribution of age groups, were similar for patients with missing surveys (n = 127) and the study population, we cannot exclude divergent PROs.
Our findings suggest that younger breast cancer patients, a population at times overtreated and at particular risk for QOL impairment, may benefit more than older women from use of T-DM1 rather than TH with regard to HRQOL. This is notable as it was observed with a full year of T-DM1 and compared to an already “de-escalated” regimen. Although younger patients and their providers may hesitate to accept de-escalated regimens, recent data suggest against an association between age and prognosis in adequately treated HER2-positive early breast cancer1,34,35,36,37. The potential for greater improvement in QOL further supports the prudent application of de-escalation strategies in the treatment of young and older breast cancer patients alike. PRO analyses, upcoming reports of longer-term outcomes from ATEMPT, and future data regarding the efficacy of a shorter course of T-DM1 from ATEMPT 2.0, will continue to shape recommendations for T-DM1 in the adjuvant setting.
Methods
Study population and procedures
ATEMPT (TBCRC033) was a randomized phase II trial that enrolled 512 participants within 90 days of their most recent surgery for stage I HER2-positive breast cancer at 24 institutions throughout the United States between 17 May 2013 and 13 December 20164. Patients were stratified by age (<55/≥55 years), planned radiotherapy (yes/no), and planned endocrine therapy (yes/no) and randomized in a 3:1 ratio to receive T-DM1 or TH, respectively. T-DM1 (3.6 mg/kg) was administered intravenously on day 1 of each 21-day cycle and continued for 17 cycles or 1 year. TH entailed intravenous weekly administration of paclitaxel (80 mg/m2) with concurrent weekly trastuzumab (4 mg/kg loading dose followed by 2 mg/kg(for 12 weeks, with trastuzumab (6 mg/kg) subsequently continued intravenously every 21 days for 13 cycles. Adjuvant radiotherapy and hormonal therapy could be initiated after 12 weeks of T-DM1 or completing paclitaxel. Female participants completing both baseline and 18-month survey assessments were included. The study (NCT01853748) was conducted in accordance with the International Conference on Harmonization Good Clinical Practice Standards and the Declaration of Helsinki. Institutional review board (IRB) approval was obtained at Dana-Farber/Harvard Cancer Center and participating sites (see the list of participating IRBs in the Supplementary Note, included in the Supplementary File). Written informed consent was obtained from each patient. The full trial protocol is available as a supplement.
Measures
Following randomized treatment allocation, English-speaking participants were surveyed at baseline (day 1 of treatment), 3 and 12 weeks, and 6, 12, and 18 months (24 months for QOL and symptom distress). To focus on posttreatment outcomes, the current analysis includes data collected at baseline and 18 months, the last timepoint at which all PRO instruments were administered. PRO surveys included FACT-B16, RSCL20, APA22, PNQ23, and WPAI:SHP21.
The FACT-B (Version 4) is designed to measure multidimensional health-related QOL (HRQOL) in breast cancer patients providing a total score (37 items, range 0–148) and 5 subscale scores: (PWB, 7 items, range 0–28), SWB, 7 items, range 0–28), emotional well-being (EWB, 6 items, range 0–24), functional well-being (FWB, 7 items, range 0–28), and (BCS, 10 items, range 0–40)16. Higher scores indicate better HRQOL. Differences of 7–8 points on the FACT-B score and 1–3 points for subscale scores are considered a minimally important difference (MID)17,18.
The RSCL measures HRQOL and symptoms in cancer patients, non-specific to breast cancer. It is comprised of four scales: physical distress (23 items, range 23–92), psychological distress (7 items, range 7–28), activity level (8 items, range 8–32), and a single item measuring overall HRQOL (range 1–7)19. Higher scores correlate with poor HRQOL, except for activity scale, for which higher scores correlate with better HRQOL.
The WPAI:SHP is a 6-item instrument designed to quantitatively assess the amount of absenteeism (percent work time missed), presenteeism (percent reduced on-the-job effectiveness), productivity loss and overall activity impairment attributable to a specific health problem20. Scores are transformed into percentages (range 0–100) with higher percentages indicating greater impairment. Missing data were handled similarly for all instruments – scores were calculated only when at least 50% of items were available and prorated for missing items. FACT-B total score was calculated only if all component subscales were valid.
Peripheral neuropathy symptoms and alopecia were assessed using specific instruments. PNQ asks participants to grade each, sensory and motor neuropathy symptoms on a 5-point Likert scale. Results were categorized as no/mild neuropathy or moderate/moderate-severe/severe neuropathy. For alopecia, we used a single item from the APA, “Have you had any hair loss during the past week?” for which participants responded “yes” or “no”.
Socio-demographic and disease characteristics were collected at enrollment. Age was dichotomized as ≤50 and >50 years, a cut-off used in other studies and approximating menopausal status9,11,14. Race and ethnicity were extracted from the medical record. Race was categorized as American Indian or Alaskan Native, Asian, Black or African American, Native Hawaiian or Other Pacific Islander, White, more than one, or other. Ethnicity was categorized as Hispanic or Latino, Non-Hispanic or unknown. Women reporting at least one menstrual period within 12 months prior to registration were considered premenopausal and otherwise postmenopausal. Primary breast surgery (lumpectomy/mastectomy) was defined at enrollment. Receipt of radiotherapy and endocrine therapy use were collected through follow-up.
Statistical analysis
Patient characteristics were compared between age groups using Fisher’s exact test. Discontinuation rates, as defined in the main study protocol, were compared between arms and age groups using Kaplan–Meier curves and a log-rank test was used to compare discontinuation rates at 12 months, the duration of protocol treatment. FACT-B, RSCL and WPAI:SHP scores were expressed as means ± standard deviations. PNQ and APA results were categorized as described and reported as percentages. Linear (RSCL, WPAI:SHP, FACT-B) and logistic (APA, PNQ) multivariable regression models were used to compare PROs within age groups and arms at 18 months post-enrollment. Multivariate regression models included age group, treatment arm, and their interaction term (arm*age group) and were adjusted for covariates: race, early discontinuation, surgery type, receipt of radiotherapy and endocrine therapy use at 18 months and the respective PRO score at baseline. Regression models were implemented on non-missing data for variables entered. Models were not adjusted for multiple comparisons. All p-values were 2-sided and considered statistically significant if < 0.05. Analyses were conducted using SAS Software, Version 9.4 (SAS Institute, Cary, NC).
Data availability
Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
References
Piccart, M. J. et al. Road map to safe and well-designed de-escalation trials of systemic adjuvant therapy for solid tumors. J. Clin. Oncol. 38, 4120–4129 (2020).
Tolaney, S. M. et al. Adjuvant paclitaxel and trastuzumab for node-negative, HER2-positive breast cancer. N. Engl. J. Med. 372, 134–141 (2015).
Tolaney, S. M. et al. Seven-year follow-up analysis of adjuvant paclitaxel and trastuzumab trial for node-negative, human epidermal growth factor receptor 2-positive breast cancer. J. Clin. Oncol. 37, 1868–1875 (2019).
Tolaney, S. M. et al. Adjuvant trastuzumab emtansine versus paclitaxel in combination with trastuzumab for stage I HER2-positive breast cancer (ATEMPT): a randomized clinical trial. J. Clin. Oncol. 39, 2375–2385 (2021).
Thomssen, C., Balic, M., Harbeck, N. & Gnant, M. St Gallen/Vienna 2021: a brief summary of the consensus discussion on customizing therapies for women with early breast cancer. Breast Care 16, 135–143 (2021).
Freedman, R. A. & Partridge, A. H. Emerging data and current challenges for young, old, obese, or male patients with breast cancer. Clin. Cancer Res. 23, 2647–2654 (2017).
Ganz, P. A., Greendale, G. A., Petersen, L., Kahn, B. & Bower, J. E. Breast cancer in younger women: reproductive and late health effects of treatment. J. Clin. Oncol. 21, 4184–4193 (2003).
Avis, N. E., Crawford, S. & Manuel, J. Quality of life among younger women with breast cancer. J. Clin. Oncol. 23, 3322–3330 (2005).
Howard-Anderson, J., Ganz, P. A., Bower, J. E. & Stanton, A. L. Quality of life, fertility concerns, and behavioral health outcomes in younger breast cancer survivors: a systematic review. J. Natl. Cancer Inst. 104, 386–405 (2012).
Mokhtari-Hessari, P. & Montazeri, A. Health-related quality of life in breast cancer patients: review of reviews from 2008 to 2018. Health Qual. Life Outcomes 18, 338 (2020).
Bantema-Joppe, E. J. et al. The impact of age on changes in quality of life among breast cancer survivors treated with breast-conserving surgery and radiotherapy. Br. J. Cancer 112, 636–643 (2015).
Maurer, T. et al. Health-related quality of life in a cohort of breast cancer survivors over more than 10 years post-diagnosis and in comparison to a control cohort. Cancers (Basel) 13, 1854 (2021).
Champion, V. L. et al. Comparison of younger and older breast cancer survivors and age-matched controls on specific and overall quality of life domains. Cancer 120, 2237–2246 (2014).
Roine, E. et al. Long-term health-related quality of life of breast cancer survivors remains impaired compared to the age-matched general population especially in young women. Results from the prospective controlled BREX exercise study. Breast 59, 110–116 (2021).
Ganz, P. A. et al. Breast cancer in older women: quality of life and psychosocial adjustment in the 15 months after diagnosis. J. Clin. Oncol. 21, 4027–4033 (2003).
Brady, M. J. et al. Reliability and validity of the Functional Assessment of Cancer Therapy-Breast quality-of-life instrument. J. Clin. Oncol. 15, 974–986 (1997).
Eton, D. T. et al. A combination of distribution- and anchor-based approaches determined minimally important differences (MIDs) for four endpoints in a breast cancer scale. J. Clin. Epidemiol. 57, 898–910 (2004).
King, M. T. et al. Meta-analysis provides evidence-based interpretation guidelines for the clinical significance of mean differences for the FACT-G, a cancer-specific quality of life questionnaire. Patient Relat. Outcome Meas. 1, 119–126 (2010).
De Haes, J., et al. The Rotterdam Symptom Checklist (RSCL): A Manual. https://meetinstrumentenzorg.nl/wp-content/uploads/instrumenten/194_2_N.pdf (2012).
de Haes, J. C., van Knippenberg, F. C. & Neijt, J. P. Measuring psychological and physical distress in cancer patients: structure and application of the Rotterdam Symptom Checklist. Br. J. Cancer 62, 1034–1038 (1990).
Reilly, M. C., Zbrozek, A. S. & Dukes, E. M. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics 4, 353–365 (1993).
Mathias, S. D., Colwell, H. H., Lalla, D. & Masaquel, A. S. Evaluation of the alopecia patient assessment (APA) questionnaire in women with breast cancer and alopecia. Value Health 16, A39 (2013).
Hausheer, F. H., Schilsky, R. L., Bain, S., Berghorn, E. J. & Lieberman, F. Diagnosis, management, and evaluation of chemotherapy-induced peripheral neuropathy. Semin. Oncol. 33, 15–49 (2006).
Krop, I. E. et al. Trastuzumab emtansine plus pertuzumab versus taxane plus trastuzumab plus pertuzumab after anthracycline for high-risk human epidermal growth factor receptor 2-positive early breast cancer: The phase III KAITLIN Study. J. Clin. Oncol. 40, 438–448 (2022).
Ruddy, K. J. et al. Chemotherapy-related amenorrhea (CRA) after adjuvant ado-trastuzumab emtansine (T-DM1) compared to paclitaxel in combination with trastuzumab (TH) (TBCRC033: ATEMPT Trial). Breast Cancer Res Treat. 189, 103–110 (2021).
Rosenberg, S. M. et al. Treatment-related amenorrhea and sexual functioning in young breast cancer survivors. Cancer 120, 2264–2271 (2014).
Rosenberg, S. M. & Partridge, A. H. Premature menopause in young breast cancer: effects on quality of life and treatment interventions. J. Thorac. Dis. 5(Suppl 1), S55–S61 (2013).
Ruddy, K. J. et al. Prospective study of fertility concerns and preservation strategies in young women with breast cancer. J. Clin. Oncol. 32, 1151–1156 (2014).
Kummerow, K. L., Du, L., Penson, D. F., Shyr, Y. & Hooks, M. A. Nationwide trends in mastectomy for early-stage breast cancer. JAMA Surg. 150, 9–16 (2015).
Flanagan, M. R. et al. A comparison of patient-reported outcomes after breast-conserving surgery and mastectomy with implant breast reconstruction. Ann. Surg. Oncol. 26, 3133–3140 (2019).
Bellon, J. R. et al. Local-regional recurrence in women with small node-negative, HER2-positive breast cancer: results from a prospective multi-institutional study (the APT trial). Breast Cancer Res Treat. 176, 303–310 (2019).
Magnuson, A. et al. Development and validation of a risk tool for predicting severe toxicity in older adults receiving chemotherapy for early-stage breast cancer. J. Clin. Oncol. 39, 608–618 (2021).
Janz, N. K. et al. Racial/ethnic differences in quality of life after diagnosis of breast cancer. J. Cancer Surviv. 3, 212–222 (2009).
Rocque, G. B. et al. Patient perspectives on chemotherapy de-escalation in breast cancer. Cancer Med. 10, 3288–3298 (2021).
Partridge, A. H. et al. Subtype-dependent relationship between young age at diagnosis and breast cancer survival. J. Clin. Oncol. 34, 3308–3314 (2016).
Partridge, A. H. et al. Effect of age on breast cancer outcomes in women with human epidermal growth factor receptor 2-positive breast cancer: results from a herceptin adjuvant trial. J. Clin. Oncol. 31, 2692–2698 (2013).
Lambertini, M. et al. Abstract P2-13-42: effect of young age at diagnosis on clinical outcomes and efficacy of anti-HER2 targeted therapy in patients with HER2-positive early breast cancer: Results from the APHINITY trial. Cancer Res. 82, P2-13-42-P12-13-42 (2022).
Acknowledgements
This study was funded by Genentech and the Gloria Spivak Faculty Advancement Fund (Tolaney). We would like to thank Valerie Hope Goldstein, a full-time employee of Dana-Farber Cancer Institute, for editing and submission assistance. We are grateful for the funding support to the Translational Breast Cancer Research Consortium (TBCRC) from The Breast Cancer Research Foundation and Susan G. Komen.
Author information
Authors and Affiliations
Contributions
Conceptualization: T.S., Y.Z., N.T., K.J.R., R.A.F., A.H.P. and S.M.T. Data curation: T.S., Y.Z., M.D., N.T. and S.M.T. Formal analysis: Y.Z. Funding acquisition: S.M.T. Investigation: C.D., D.Y., S.J.I., V.V., H.J.B., E.P.W., A.C.W., I.K., A.H.P. and S.M.T. Methodology: N.T. Supervision: S.M.T. Writing - original draft: T.S. and S.M.T. Writing - review and editing: all authors.
Corresponding author
Ethics declarations
Competing interests
T.S. reports support from the Pinchas Borenstein Talpiot Medical Leadership Program, Sheba Medical Center Israel, and The American Physicians Fellowship for Medicine in Israel. TS reports no competing non-financial interests. C.D. receives research support (to institution) from Roche/Genentech, PUMA, and Daichii Sankyo and consulting fees/honoraria from Daichii Sankyo, Pfizer, Novartis, Gilead, and Seagen. CD reports no competing non-financial interests. D.Y. reports consulting or advisory roles at Novartis, Biotheranostics, Bristol Myers Squibb, G1 Therapeutics, Athenex, Immunomedics, Sanofi/Aventis, R-Pharm, and Lilly; serves on the Speakers’ Bureaus of Novartis, and Genentech/Roche; receives research funding from Genentech/Roche, Novartis, MedImmune, Lilly, Medivation, Pfizer, Tesaro, Macrogenics, AbbVie, Merck, Clovis Oncology, Amgen, Biomarin, Biothera, Dana-Farber Cancer Institute, Incyte, Innocrin Pharma, Nektar, NSABP Foundation, Odonate Therapeutics and Polyphor; and receives travel/accommodations/other expenses from Novartis and Genentech/Roche. DY reports no competing non-financial interests. S.J.I. reports consulting or advisory roles at Seattle Genetics and Novartis, and receives research funding from Genentech, AbbVie, OncoPep, Merck, AstraZeneca/MedImmune, and Outcomes4Me. SJI reports no competing non-financial interests. V.V. receives honoraria from and serves in consulting or advisory roles for Genentech/Roche, Merck, and Novartis; and receives travel/accommodations/other expenses from Genentech/Roche. VV reports no competing non-financial interests. E.P.W. served over the past three years as a consultant to and received honoraria from Carrick Therapeutics, Genentech/Roche, and Jounce Therapeutics. He is an uncompensated member of the Scientific Advisory Boards for Leap Therapeutics. EPW reports no other competing financial or non-financial interests. A.C.W. receives research funding from Roche and Pfizer awarded to Johns Hopkins University; has been named as inventor on one or more issued patents or pending patent applications related to methylation in breast cancer, and has assigned his rights to JHU and participates in a royalty sharing agreement with JHU. Open Payments Link: https://openpaymentsdata.cms.gov/physician/357301/. A.C.W. reports no competing non-financial interests. I.K. receives institutional research support from Genentech/Roche, Macrogenics, and Pfizer; receives fees from Novartis and Merck for Data Monitoring Board participation and receives consulting fees from Bristol Meyers Squibb, Daiichi/Sankyo, Macrogenics, Taiho Oncology, Genentech/Roche, SeaGen, and AstraZeneca. IK reports no competing non-financial interests. S.M.T. serves in a consulting or advisory role for Novartis, Pfizer, Merck, Eli Lilly, AstraZeneca, Genentech/Roche, Eisai, Sanofi, Bristol Myers Squibb, Seattle Genetics, Odonate Therapeutics, CytomX Therapeutics, Daiichi Sankyo, Athenex, Gilead, Mersana, Certara, Ellipses Pharma, 4D Pharma, OncoSec Medical Inc., BeyondSpring Pharmaceuticals, OncXerna, Zymeworks, Zentalis, Blueprint Medicines, Reveal Genomics, ARC Therapeutics, Infinity Therapeutics, Chugai Pharmaceuticals, Myovant, Zetagen, Umoja Biopharma, Menarini/Stemline, Aadi Bio and Bayer. SMT receives research funding from Genentech/Roche, Merck, Exelixis, Pfizer, Lilly, Novartis, Bristol Myers Squibb, Eisai, AstraZeneca, NanoString Technologies, Cyclacel, Nektar, Gilead, Sanofi, Seattle Genetics. SMT declares no competing non-financial interests. The remaining authors declare no relevant conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sella, T., Zheng, Y., Tayob, N. et al. Treatment discontinuation, patient-reported toxicities and quality-of-life by age following trastuzumab emtansine or paclitaxel/trastuzumab (ATEMPT). npj Breast Cancer 8, 127 (2022). https://doi.org/10.1038/s41523-022-00495-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-022-00495-x