Abstract
Study design
Retrospective chart audit.
Objectives
Describing the respiratory complications and their predictive factors in patients with acute traumatic spinal cord injuries at C5–T5 level during the initial hospitalization.
Setting
Hospital Vall d’Hebron, Barcelona.
Methods
Data from patients admitted in a reference unit with acute traumatic injuries involving levels C5–T5. Respiratory complications were defined as: acute respiratory failure, respiratory infection, atelectasis, non-hemothorax pleural effusion, pulmonary embolism or haemoptysis. Candidate predictors of these complications were demographic data, comorbidity, smoking, history of respiratory disease, the spinal cord injury characteristics (level and ASIA Impairment Scale) and thoracic trauma. A logistic regression model was created to determine associations between potential predictors and respiratory complications.
Results
We studied 174 patients with an age of 47.9 (19.7) years, mostly men (87%), with low comorbidity. Coexistent thoracic trauma was found in 24 (19%) patients with cervical and 35 (75%) with thoracic injuries (p < 0.001). Respiratory complications were frequent (53%) and were associated to longer hospital stay: 83.1 (61.3) and 45.3 (28.1) days in patients with and without respiratory complications (p < 0.001). The strongest predictors of respiratory complications were: previous respiratory disease (OR 5.4, 95% CI: 1.5–19.2), complete motor function impairment (AIS A–B) (OR 4.7, 95% CI: 2.4–9.5) and concurrent chest trauma (OR 3.73, 95% CI: 1.8–7.9).
Conclusions
Respiratory complications are common in traumatic spinal cord injuries between C5–T5. We identified previous respiratory disease, complete motor function impairment and the coexistence of thoracic trauma as predictors of respiratory complications. Identification of patients at risk might help clinicians to implement preventive strategies.
Similar content being viewed by others
Introduction
Traumatic spinal cord injuries are medical emergencies associated with substantial morbidity and mortality [1]. Respiratory complications are common in patients with spinal cord injuries and their presence predicts worse prognosis and increases health care costs [2, 3]. The level of the injury and its degree, complete or incomplete, are key factors for predicting the severity of respiratory events in these patients. It is known that injuries above C5 seriously compromise the respiratory pump and frequently cause long-term full or partial dependence on mechanical ventilation or diaphragm pacing [4,5,6,7].
Acute respiratory complications associated to spinal cord injuries below C4 are less well known. A reduction in the number of respiratory events has been reported as the level of the lesion descends: while respiratory complications are still frequent in patients with injuries above T1 [2], respiratory function is usually normal with lesions at T12 level or lower [4, 5]. Historical series have shown that these complications are frequent in patients with high thoracic spinal cord injuries [8, 9], who experience mortality rates of 10%, nearly 27 times higher than in patients without respiratory complications [10].
Consequently, it is of great interest to investigate the factors that may predict the development of respiratory complications in these patients during their initial hospitalization. Previous research has found AIS impairment scales A and B are predictors of respiratory complications [9]. However, to our knowledge, at least two other potential predictive factors have not been evaluated. First, the coexistence of previous comorbidities, especially respiratory diseases. Second, the frequent presence of thoracic trauma in patients with spinal cord lesions between C5–T5, which may itself lead to respiratory complications. In recent years significant advances in the respiratory care of spinal cord injuries, especially non-invasive ventilation and assisted coughing techniques, have been made. The identification of predictive factors of respiratory complications might provide clinicians with useful information in order to identify those patients at risk of developing respiratory complications and lead to a better prevention and management of these complications.
Based on these considerations, the aims of our study were: (1) To describe the respiratory complications in patients with acute traumatic spinal cord injuries at C5–T5 level during the initial hospitalization and (2) to analyze the predictors of these respiratory complications.
Methods
The research protocol was approved by the Ethics and Clinical Research Committee of our institution.
The Vall d’Hebron University Hospital Spinal Cord Injury Unit is a reference service in our region’s public health system and provides care to the population of Catalonia, Balearic Islands and Andorra.
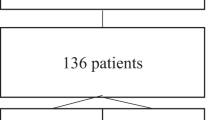
We retrospectively reviewed all patients with an acute THSCI and a level of injury between C5 and T5 admitted during the period 2010–2015. Patient information is systematically recorded at our unit during admission. The only exclusion criteria was the coexistence of cranioencephalic trauma with significant head injury (Glasgow Coma Scale of <8 at first assessment).
For this study the following variables were assessed:
-
(1)
Demographic data (age, gender), etiology of the traumatic spinal cord injury, and duration of hospital stay from initial admission to discharge.
-
(2)
Smoking: yes (current or previous) or not.
-
(3)
Presence of a history of respiratory disease. Patients were asked for a previous diagnosis of asthma, COPD, sleep apnea or other chronic respiratory disease.
-
(4)
Comorbidity was assessed by the Charlson Index [11] that takes into account the number and the severity of comorbid diseases and results in a single score, with lower values indicating less comorbidity: no comorbidity, with score 0; mild, with scores of 1–2; moderate, with scores of 3–4; and severe, with scores ≥5.
-
(5)
Characteristics of the spinal cord injury: its level and ASIA Impairment Scale (AIS) classification according to the American Society of Spinal Cord Injury [12].
-
(6)
Presence of thoracic trauma. Both open and closed injuries were considered. Closed trauma was classified as: rib, sternal, clavicle or scapula fractures; hemothorax or pneumothorax; pulmonary contusion or presence of lesion in intrathoracic organs (vascular structures, trachea and bronchi, esophagus or diaphragm).
-
(7)
Presence of respiratory complications during the hospital stay, defined as:
(A) Acute respiratory failure, defined as:
(1) An arterial oxygen saturation of <90% or a partial pressure of arterial oxygen of <60 mm while breathing room air, and/or
(2) Respiratory acidosis (defined as an arterial pH below 7.35 with a partial pressure of arterial carbon dioxide >45 mm Hg).
(B) Respiratory infection according to Centre for Disease Control criteria [13].
(1) Non-pneumonia lower respiratory infection was diagnosed if the patient had no clinical or radiographic evidence of pneumonia and had two of the following: fever (>38 °C), cough, new or increased sputum production, rhonchus, wheezing and either of the following:
a. Organism isolated from culture obtained by deep tracheal aspirate or bronchoscopy,
b. Positive antigen test on respiratory secretions.
(2) Pneumonia was diagnosed if the patient met one of the following criteria:
1. Rales or dullness to percussion on physical examination of chest, and any of the following:
a. New onset of purulent sputum or change in character of sputum,
b. Organism isolated from blood culture,
c. Isolation of pathogen from specimen obtained by transtracheal aspirate, bronchial brushing, or biopsy;
2. Chest radiographic examination with new or progressive infiltrate, consolidation, cavitation, or pleural effusion, and any of the following:
a. New onset of purulent sputum or change in character of sputum;
b. Organism isolated from blood culture;
c. Isolation of pathogen from specimen obtained by transtracheal aspirate, bronchial brushing or biopsy;
d. Isolation of virus or detection of viral antigen in respiratory secretions;
e. Diagnostic single antibody titre (IgM) or fourfold increase in paired serum samples (IgG) for pathogen;
f. Histopathologic evidence of pneumonia.
(C) Atelectasis confirmed by chest radiography or CT scan.
(D) Non-hemothorax pleural effusion.
(E) Pulmonary embolism.
(F) Haemoptysis.
Statistical analysis
Descriptive data are presented as percentages, means (SD) or medians (range). Comparisons between patients with and without respiratory complications were performed with unpaired t test for quantitative variables or Pearson chi-squared test for categorical data. Subsequently, a logistic regression model was created to determine the predictors of respiratory complications. In this model, the presence of respiratory complications was the dependent variable and age, gender, smoking and the variables that were significant in the univariate study were included as predictors of the model. Analyses were conducted using Stata IC 14 (StataCorp. 2015. Stata Statistical Software:Release 14. College Station, TX: StataCorp LP).
Results
The characteristics of the 174 patients studied are shown in Table 1. The majority were young men with low comorbidity. The hospital stay (median, p25–75) was 56 days (34–82). Fifty-nine patients (34%) had thoracic trauma associated with the spinal cord injury, most of them with some kind of fracture as detailed in Table 1. Thoracic trauma was more frequent in patients with a lesion level of T1–T5: 24 patients (19%) with cervical lesions and 35 patients (75%) with thoracic spine injuries (p < 0.001).
Ninety-two patients (53%) had some respiratory complication during the admission (Table 2). Regarding ventilatory assistance, 52 patients (30%) required mechanical ventilatory support during admission: invasive in 45 cases and non- invasive in seven. At the time of discharge, two patients required home ventilatory support, one invasive and the other non-invasive. Two patients died during their stay, both due to respiratory failure.
Patients’ characteristics according to the presence or absence of respiratory complications during admission are displayed in Table 3. Patients who had respiratory complications during their admission required a longer hospital and intensive care unit stay. Respiratory complications were more frequent in patients with thoracic injury, complete motor impairment (AIS A–B), chest trauma or a history of previous respiratory disease. These variables were introduced in the multivariate analysis along with age, gender and smoking. The best model for predicting the onset of respiratory complications in our patients included the presence of previous respiratory disease, motor impairment and the concurrence of chest trauma (Hosmer–Lesmeshow goodnes of fit test = 0.877). Table 4 shows the OR values for this model.
Discussion
The results of our study in a relatively large series of patients with traumatic low cervical or high thoracic spinal cord injuries showed a high prevalence of respiratory complications during the initial hospitalization just after the injury. As predictors of these complications we identified the presence of previous respiratory disease, complete motor impairment (AIS A–B) and coexistent thoracic trauma. Hospital stay was almost twice as long in patients with respiratory complications than in those without.
Only two patients died due to respiratory complications during admission, and two more required home ventilatory support at discharge. These data contrast with the 11% mortality and 6.5% ventilator-dependance rates previously reported in patients with high spinal cord injuries [14]. In our patients, the presence of a complete motor lesion was independently associated with a fourfold risk of respiratory complications. In contrast in the multivariate analysis the level of injury was not identified as an independent predictive factor. These findings are consistent with a predictive model previously described, that only found AIS impairment scales A and B as predictors of respiratory complications [9]. We know that the lesions in the segments considered in the present study affect intercostal musculature, as well as accessory inspiratory muscles and abdominal muscles. A decrease in the respiratory activity of the inspiratory motor neurons in the caudal direction has been demonstrated [15,16,17], and the presence of interneurons modulating the activity of these motoneurons has been found in segments of the thoracic medulla [18]. However, we do not have precise knowledge of the interactions between the different muscle groups. The integration of information from respiratory centres into each level of the lower cervical and upper thoracic spinal cord and its transmission to respiratory muscles is not yet clearly understood either. The non-explanatory design of our study do not allow us a causal interpretation of our findings and studies addressing these causal mechanisms and their relationship with the alterations of respiratory mechanics described in the first phase of spinal shock [4, 19] and their posterior improvement linked to the progressive increase in muscle tone [20] and potencial plasticity of the involved neural network [21, 22] are needed.
As in previous series of traumatic spinal cord injuries [23, 24], our patients were predominantly men with little associated comorbidity and Charlson comorbidity index was not predictive of respiratory complications. However, the presence of previous respiratory disease was associated with a very high risk of respiratory complications. This finding has been previously found in chronic spinal cord injuries [25] but has not been analysed until now in acute injuries. We think that it is clinically relevant since it could identify a group of patients in need of specific preventive protocols. Early introduction of intensive physiotherapy and assisted coughing techniques together with the early use of mucolytics and bronchodilators should be considered [26].
The presence of thoracic trauma was also an independent risk factor for respiratory complications. As expected, it was more frequent in patients with thoracic injury but it was also detected in 18.9% of cervical injuries. In previous series focused on high cervical injuries its presence was not considered. Our data underline the need to assess the coexistence of thoracic trauma when considering the study of respiratory complications in these patients and to be aware of the likelihood of respiratory complications.
Our study has several strengths. It tests a model to predict respiratory complications in a wide series of patients seen at a reference unit of spinal cord injuries and focuses on levels of spinal cord injuries on which little previous information is available. However, its retrospective nature is a limitation. As spinal cord injuries are rare events, this limitation is a common characteristic in the literature; we believe that it is counteracted in our study by the systematic approach applied to spinal cord injury and the information collected during the admission at our unit, which is a specialised reference centre. However, even though assisted coughing techniques have been progressively introduced during the years of study, precise data on its use or other physiotherapy techniques were not available. It would also be desirable to have information on lung function data and breathing during sleep in our patients to be able to assess periods of hypoventilation or the coexistence of sleep apnoeas, which are frequent in spinal cord injuries [27,28,29]. For purposes of the predictive model analysis, we clumped several respiratory complications all together. The predictor factors for each respiratory complications might be different. Lastly, although in this study we focused on C5–T5 spinal cord injuries, it would have been interesting to compare the rates of respiratory complications in people with high and low cervical injuries. In summary, spinal cord injuries are acknowledged as catastrophic clinical events requiring complex clinical management at specialised units. Our study draws attention to the frequent presence of respiratory complications during the hospital stay in patients admitted after and acute traumatic spinal cord injury between C5–T5. We have identified former respiratory disease, motor involvement according to AIS grades A and B and the coexistence of thoracic trauma as predictive factors of respiratory complications in these patients. Early identification of patients at risk of respiratory complications might help clinicians to implement preventive strategies to decrease these complications.
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Middleton JW, Dayton A, Walsh J, Rutkowski SB, Leong G, Duong S. Life expectancy after spinal cord injury: a 50-year study. Spinal Cord. 2012;50:803–11.
Winslow C, Bode RK, Felton D, Chen D, Meyer PR. Impact of respiratory complications on length of stay and hospital costs in acute cervical spine injury. Chest. 2002;121:1548–54.
Van den Berg MEL, Castellote JM, de Pedro-Cuesta J, Mahillo-Fernandez I. Survival after spinal cord injury: a systematic review. J Neurotrauma. 2010;27:1517–28.
Brown R, DiMarco AF, Hoit JD, Garshick E. Respiratory dysfunction and management in spinal cord injury. Respir Care. 2006;51:853–68.
Linn WS, Spungen AM, Gong H, Adkins RH, Bauman WA, Waters RL. Forced vital capacity in two large outpatient populations with chronic spinal cord injury. Spinal Cord. 2001;39:263–8.
Chiodo AE, Scelza W, Forchheimer M. Predictors of ventilator weaning in individuals with high cervical spinal cord injury. J Spinal Cord Med. 2008;31:72–7.
Onders RP, Khansarinia S, Weiser T, Chin C, Hungness E, Soper N, et al. Multicenter analysis of diaphragm pacing in tetraplegics with cardiac pacemakers: positive implications for ventilator weaning in intensive care units. Surgery. 2010;148:893–8.
Jackson AB, Groomes TE. Incidence of respiratory complications following spinal cord injury. Arch Phys Med Rehabil. 1994;75:270–5.
Aarabi B, Harrop JS, Tator CH, Alexander M, Dettori JR, Grossman RG, et al. Predictors of pulmonary complications in blunt traumatic spinal cord injury. J Neurosurg Spine. 2012;17:38–45.
Cotton BA, Pryor JP, Chinwalla I, Wiebe DJ, Reilly PM, Schwab CW, et al. Respiratory complications and mortality risk associated with thoracic spine injury. J Trauma. 2005;59:1400–9.
Yurkovich M, Avina-Zubieta JA, Thomas J, Gorenchtein M, Lacaille D. A systematic review identifies valid comorbidity indices derived from administrative health data. J Clin Epidemiol. 2015;68:3–14.
Maynard FM, Bracken MB, Creasey G, Ditunno JF, Donovan WH, Ducker TB, et al. International Standards for Neurological and Functional Classification of Spinal Cord Injury. American Spinal Injury Association. Spinal Cord. 1997;35:266–74.
Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM. CDC definitions for nosocomial infections, 1988. Am J Infect Control. 1988;16:128–40.
Quesnel A, Veber B, Proust F, Agasse E, Beuret Blanquart F, Verin E. What are the perspectives for ventilated tetraplegics? A French retrospective study of 108 patients with cervical spinal cord injury. Ann Phys Rehabil Med. 2015;58:74–7.
De Troyer A, Gorman RB, Gandevia SC. Distribution of inspiratory drive to the external intercostal muscle in humans. J Physiol. 2003;546:943–54.
De Troyer A, Kirkwood PA, Wilson TA. Respiratory action of the intercostal muscles. Physiol Rev. 2005;85:717–56.
Gandevia SC, Hudson AL, Gorman RB, Butler JE, De Troyer A. Spatial distribution of inspiratory drive to the parasternal intercostal muscles in humans. J Physiol. 2006;573:263–75.
Ikeda K, Kawakami K, Onimaru H, Okada Y, Yokota S, Koshiya N, et al. The respiratory control mechanisms in the brainstem and spinal cord: integrative views of the neuroanatomy and neurophysiology. J Physiol Sci. 2017;67:45–62.
Ditunno JF, Little JW, Tessler A, Burns AS. Spinal shock revisited: a four-phase model. Spinal Cord. 2004;42:383–95.
Bluechardt MH, Wiens M, Thomas SG, Plyley MJ. Repeated measurements of pulmonary function following spinal cord injury. Paraplegia. 1992;30:768–74.
Alilain WJ, Li X, Horn KP, Dhingra R, Dick TE, Herlitze S, et al. Light-induced rescue of breathing after spinal cord injury. J Neurosci. 2008;28:11862–70.
Alilain WJ, Horn KP, Hu H, Dick TE, Silver J. Functional regeneration of respiratory pathways after spinal cord injury. Nature. 2011;475:196–200.
Cao Y, Selassie AW, Krause JS. Risk of death after hospital discharge with traumatic spinal cord injury: a population-based analysis, 1998-2009. Arch Phys Med Rehabil. 2013;94:1054–61.
Wyndaele M, Wyndaele JJ. Incidence, prevalence and epidemiology of spinal cord injury: what learns a worldwide literature survey? Spinal Cord. 2006;44:523–9.
Stolzmann KL, Gagnon DR, Brown R, Tun CG, Garshick E. Risk factors for chest illness in chronic spinal cord injury. Am J Phys Med Rehabil. 2010;89:576–83.
Chatwin M, Ross E, Hart N, Nickol AH, Polkey MI, Simonds AK. Cough augmentation with mechanical insufflation/exsufflation in patients with neuromuscular weakness. Eur Respir J. 2003;21:502–8.
Sankari A, Martin JL, Bascom AT, Mitchell MN, Badr MS. Identification and treatment of sleep-disordered breathing in chronic spinal cord injury. Spinal Cord. 2015;53:145–9.
Tran K, Hukins C, Geraghty T, Eckert B, Fraser L. Sleep-disordered breathing in spinal cord-injured patients: a short term longitudinal study. Respirology. 2010;15:272–6.
Sankari A, Martin JL, Safwan Badr M. Sleep-disordered breathing and spinal cord injury: challenges and opportunities. Curr Sleep Med Rep. 2017;3:272–8.
Acknowledgements
We gratefully acknowledge the Spinal Cord Injury Unit and the Respiratory Department in our centre, Hospital Universitari Vall d’Hebron, for their generous support during the development of this study.
Author information
Authors and Affiliations
Contributions
JS: responsible for the conceptualization of the study, data curation, methodology and formal analysis and paper development. MAGV: assisted with conceiving and designing the study and major revisions. AG, SM and MP: contributed with data curation, writing and overall editing. ER and PL: assisted with writing of results and overall editing. GS: responsible for conceiving and designing the study, writing introduction and editing. Also provided input on methodology, analysis, results and discussion section. JF: contributed with the conceptualization of the study and assisted with writing of introduction, results and discussion sections and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sampol, J., González-Viejo, M.Á., Gómez, A. et al. Predictors of respiratory complications in patients with C5–T5 spinal cord injuries. Spinal Cord 58, 1249–1254 (2020). https://doi.org/10.1038/s41393-020-0506-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-020-0506-7