Abstract
Multiscale neuroscience conceptualizes mental illness as arising from aberrant interactions across and within multiple biopsychosocial scales. We leverage this framework to propose a multiscale disease progression model of psychosis, in which hippocampal-cortical dysconnectivity precedes impairments in episodic memory and social cognition, which lead to more severe negative symptoms and lower functional outcome. As psychosis represents a heterogeneous collection of biological and behavioral alterations that evolve over time, we further predict this disease progression for a subtype of the patient sample, with other patients showing normal-range performance on all variables. We sampled data from two cross-sectional datasets of first- and multi-episode psychosis, resulting in a sample of 163 patients and 119 non-clinical controls. To address our proposed disease progression model and evaluate potential heterogeneity, we applied a machine-learning algorithm, SuStaIn, to the patient data. SuStaIn uniquely integrates clustering and disease progression modeling and identified three patient subtypes. Subtype 0 showed normal-range performance on all variables. In comparison, Subtype 1 showed lower episodic memory, social cognition, functional outcome, and higher negative symptoms, while Subtype 2 showed lower hippocampal-cortical connectivity and episodic memory. Subtype 1 deteriorated from episodic memory to social cognition, negative symptoms, functional outcome to bilateral hippocampal-cortical dysconnectivity, while Subtype 2 deteriorated from bilateral hippocampal-cortical dysconnectivity to episodic memory and social cognition, functional outcome to negative symptoms. This first application of SuStaIn in a multiscale psychiatric model provides distinct disease trajectories of hippocampal-cortical connectivity, which might underlie the heterogeneous behavioral manifestations of psychosis.
Similar content being viewed by others
Introduction
Psychotic disorders can be characterized along the dimensions of positive, negative, and cognitive symptoms [1, 2]. Positive symptoms describe abnormal perceptions, thoughts, and behaviors (e.g., hallucinations) while negative symptoms describe a significant reduction of typical behaviors and motivation (i.e., asociality) [1]. Cognitive symptoms tend to arise before the onset of positive and negative symptoms [3, 4] and include neurocognitive deficits, particularly in episodic memory [2]. Due to its broad range of symptoms, psychotic disorders are among the most disabling mental disorders and a leading cause of disability worldwide [5]. The most prominent measure of disability caused by psychotic disorders is functional outcome, which assesses the influence of disease on social and occupational functioning [6, 7]. Within these two domains, affected individuals have fewer stable friendships, difficulties with social interactions, higher unemployment rates, and increased occupational stress [7]. Beyond the considerable impact on the affected individual and their social surroundings, psychosis also carries an immense economic burden caused by the often-persisting difficulties of maintaining employment following a first episode of psychosis (FEP) [8, 9]. Thus, identifying disease trajectories up to and including poor functional outcome of psychosis is crucial.
Multiscale neuroscience is a framework within which poor functional outcome in psychosis may be understood through its neurocognitive mechanisms, for example, from neurobiological changes (meso scale) that result in poor functional outcomes (social scale) through cognitive and clinical symptoms (manifest or behavior scale). Within this framework, psychiatric disorders arise as the result of aberrant interactions within and across those scales [10, 11], implying that dysfunction in one scale influences and reflects functionality of higher- and lower-order scales [10]. On the meso scale, graph-theoretical measures of connectivity identify the hippocampus as a key convergence zone for cortical input [12,13,14]. By grouping hippocampal regions into a hippocampal module, Makowski et al. [15] show that longitudinal structural covariance-based connectivity between the hippocampus and large-scale brain networks [16] is significantly reduced in FEP compared to controls. These findings further provide a link between the hippocampus and the manifest- or behavior-scale, as reductions in hippocampal connectivity are associated with heightened negative symptoms, as mediated by impaired episodic memory [15]. Using longitudinal data, Makowski et al. [15] were, thus, the first to characterize a multiscale disease progression from hippocampal-cortical dysconnectivity to episodic memory and negative symptoms. Notably, the association between episodic memory and psychosis is robustly established cross-sectionally and longitudinally across the psychosis spectrum, spanning individuals at clinical high risk [17], FEP [18], and chronic psychosis [2]. Within the manifest-scale, the relationship between episodic memory and negative symptoms is mediated through social cognition (i.e., emotion recognition and theory of mind) [19], linking cognitive impairments to negative symptoms. On the social-scale, sex differences in episodic memory impairments [20], and impaired social cognitive abilities [21, 22] predict functional outcome as mediated by negative symptoms, while negative symptoms are also directly associated with impaired functioning [23, 24]. We thus propose a multiscale model in which hippocampal-cortical dysconnectivity leads to impaired episodic memory and social cognition, resulting in higher negative symptoms and, ultimately, poor functional outcome.
To assess this model, we implemented an unsupervised machine-learning algorithm called Subtype and Stage Inference (SuStaIn) [25]. SuStaIn uniquely combines disease progression modeling and clustering to infer longitudinal disease progressions from cross-sectional data. By clustering patient groups with shared common disease progression patterns, SuStaIn is particularly well-suited to evaluate a multiscale model of psychosis, as heterogeneous impairments are seen across patients [26,27,28,29,30,31], indicating that our hypothesized disease progression pattern may apply to only a sub-population of patients. While SuStaIn has primarily been implemented in neurodegenerative disorders [25, 32,33,34,35,36,37], two studies to date have applied SuStaIn to neuroimaging markers of psychosis [38, 39]. Both identified distinct subtypes of volume atrophy in patients, with one subtype beginning to deteriorate in the hippocampus. Jiang et al. [38] further related these subtypes to distinct treatment responses to antipsychotic medication. Building on this important work, our study will, for the first time, implement SuStaIn in a multiscale psychiatric model.
Further, SuStaIn’s staging approach provides a translational component to our data-driven model by directly allowing for patient stratification and disease prediction. This feature is of particular importance in relation to clinical staging models, such as McGorry’s clinical stages [40, 41]. McGorry et al. [40] hypothesize a unidirectional clinical development of severe mental disorders, in which new stages represent a significant change in clinical status [40, 41]. In this work, we sampled across clinical stages by combining data on first- and multi-episode psychosis (MEP). Based on the qualitative differences between clinical stages regarding symptoms, cognition, and functioning [40, 41], it was of further interest to explore how SuStaIn’s data-driven disease stages would stratify individuals from distinct clinical stages. To this end, we employed z-score SuStaIn [25], which assesses monotonous disease progression through z-score deviations from the mean, resembling the assumed unidirectional progression of McGorry’s clinical stages.
Applying z-score SuStaIn thus allowed us to investigate our novel multiscale model across subtypes of psychosis while addressing whether our data-driven disease stages would map onto clinical staging models [40]. Taking into consideration the heterogeneous nature of psychotic disorders, we hypothesize (i) that there may exist at least two patient subtypes, one subtype showing normal-range performance on the components of the model and one subtype showing impairments. In the impaired subtype, we hypothesize (ii) a disease progression pattern from hippocampal-cortical dysconnectivity to impaired episodic memory and social cognition, higher negative symptoms, and poorer functional outcome. This work is the first to outline a relationship between hippocampal-cortical connectivity and functional outcomes through cognition, social cognition, and symptoms. Given this multiscale nature, our data-driven findings can thus contribute to precision diagnostic and treatment efforts across scales.
Methods
Participants
We sampled patient and control data from two independent datasets. Study 1 collected data in a FEP cohort (recruitment: 2015–2021) and Study 2 from a MEP cohort (recruitment: 2011–2016; [42]); (see Supplementary F1 for pre-established inclusion and exclusion criteria of both studies). Patient samples were recruited from the Douglas Research Centre, Montréal, Canada. FEP patients were recruited after being admitted to the prevention and early intervention program for psychosis (PEPP-Montréal) [43], while MEP patients were patients of the outpatient and inpatient units. Both control samples were recruited from the same catchment area. After excluding participants (see Supplementary F2 for the exclusion process), Study 1 consisted of 55 patients and 53 controls, and Study 2 consisted of 108 patients and 66 controls, rendering our total sample size 163 patients and 119 non-clinical controls. Ethical approval was granted by the Douglas Institute Research Ethics Board. Both studies were conducted in accordance with the Declaration of Helsinki, and written informed consent was obtained prior to the study. Participants were compensated with monetary rewards. See Supplementary S1 for a power calculation of the sample size.
Data collection
In both studies, data including sex, age, duration of illness in years (first onset of frank psychotic symptoms), and medication were assessed through an interview with trained research staff. Diagnoses were established through the Structured Clinical Interview for DSM-IV and V diagnoses [44, 45]. Episodic memory and social cognition were measured via the Cogstate Schizophrenia Battery [46], namely, the International Shopping List and Social-Emotional Cognition Tests. Depressive symptoms were assessed through the Calgary Depression Scale [47]. Negative and positive symptoms were assessed using the Scale for the Assessment of Negative/Positive Symptoms (SANS/SAPS) [48, 49], respectively. Functional outcome was measured through the Social and Occupational Functional Assessment Scale (SOFAS, [6]), which assesses social and occupational functioning on a scale from 0 (grossly impaired functioning) to 100 (excellent functioning). The SOFAS was not measured in MEP patients, and we thus reconstructed SOFAS scores on the basis of the Personal and Social Performance Scale (PSP, [50]). The PSP assesses occupational functioning, interpersonal functioning, self-care, and aggressive behavior and is scored on a scale from 1 (no autonomy) to 100 (excellent functioning in all four domains). To score the PSP, we consulted the SANS for occupational functioning (item 15), interpersonal functioning (items 20 and 21), self-care (item 14), and the SAPS for aggressive behavior (item 23). PSP scores were then transferred onto the SOFAS. The scoring of both scales was completed by two independent raters, and a consensus was reached. Validity of this procedure was justified through the scoring of an independent dataset with pre-existing SOFAS ratings. See Supplementary S2 for scale and scoring descriptions, and Supplementary S3 and F6 for details on the reconstruction procedure.
Hippocampal-cortical connectivity
All MRI data were acquired with the same 3T Siemens Magnetom Trio scanner located at the Cerebral Imaging Centre of the Douglas Research Centre. See Supplementary S4 for the scan parameters of both studies. We used the high-performance computing resources available via the Digital Research Alliance of Canada to extract cortical thickness using CIVET [51], and hippocampal volumes with MAGeT [52, 53]. See Fig. 1 and Supplementary S5 for a detailed explanation of the processing steps. We then computed structural covariance between the 62 DKT and 18 hippocampal regions by performing full-sample correlations for the patient and the control samples separately [13] and calculated subject-specific structural covariance matrices through the jackknife bias estimation procedure [54, 55]. Hippocampal-cortical connectivity for each hippocampal region was derived through the graph-theoretical participation coefficient [56], with higher values indicating more diverse nodal connectivity to large-scale cortical networks [16] rather than hippocampal regions. See Supplementary S5 for a detailed explanation of these calculations. The participation coefficients of the hippocampal regions were averaged for each hemisphere respectively.
SuStaIn analyses
We first regressed out the effects of sex, age, and total brain volume on the six input markers and then z-scored the data in comparison to control values. As no control values were sampled for the SANS and SOFAS, we z-scored these values in relation to previously reported control means [57, 58] and applied z-score SuStaIn [25] to the patient data only. Z-score SuStaIn places subjects at specific disease stages depending on their z-score deviation from a non-clinical control sample. Through maximum likelihood estimation, SuStaIn assesses the temporal sequence of marker deviations and employs purely cross-sectional data to infer common patterns of disease progression via a stage progression model. Under the assumption of monotonous marker deterioration, z-score SuStaIn thus places markers with the highest likelihood of impairment into early disease stages, while markers with less likelihood are placed into later disease stages. This type of modeling implies that patients in early disease stages share impairments on a few markers, whereas the number of markers that show impairments grows over the course of the disease progression. SuStaIn’s disease stages thus provide an overview of shared patterns of marker impairment at a given stage while also allowing for the inference of disease progression following this stage. In addition, distinct subtypes with common impairments are identified through a clustering component, the split-cluster expectation maximization, with likelihood testing determining the optimal number of clusters.
Z-score SuStaIn was performed in Python (pySuStaIn [59]). We set the threshold of z-scores to one, two, and three deviations from the control mean for each biomarker while reaching a maximum threshold of five z-scores at the end of the disease progression, as in previous work [25]. Considering the absence of negative symptom and functional outcome data in controls, we ran SuStaIn on the patient data only. We then validated the resulting subtypes using 10-fold Markov Chain Monte Carlo iteration cross validation. Log-likelihood tests and the Cross-Validation Information Criterion (CVIC, [25]) were used to obtain an indication of the optimal number of subtypes. Following SuStaIn, follow-up analyses were performed and corrected for multiple comparisons. See Supplementary S6 for a detailed explanation of these analyses.
Results
Demographic data
A summary of demographic characteristics can be seen in Table 1. We further examined the role of diagnosis and depressive symptoms in our sample. Study 2 had significantly more individuals with non-affective psychosis than Study 1 (X2(1, N = 163) = 9.865; p = 0.002), but none of the input markers of our model differed significantly between diagnoses (ps > 0.582). When looking at depressive symptoms, no significant difference between FEP and MEP was found (p > 0.244). We did, however, find a significant correlation between negative symptoms and depressive symptoms in FEP (r(52) = 0.372, p = 0.006), but not MEP. See Supplementary F3 for further visualization of age and illness duration in both datasets.
Subtype and stage inference
SuStaIn considers individuals who are sorted into disease stage 0 as normal-range performing on all markers and assigns them to a separate Subtype 0. In our case, Subtype 0 thus consists of patients with no impairment on the markers. To differentiate Subtype 0 from other subtypes with impairment, we therefore refer to all subtypes except Subtype 0 as disease subtypes. We ran SuStaIn on a maximum of two disease subtypes, which resulted in a CVIC of 3329.44 (log-likelihood of—168.32) for two disease subtypes and a CVIC of 3326.73 (log-likelihood of—168.08) for one disease subtype. Lower CVIC scores suggest a better model fit [25], yet differences <6 are not considered to be meaningful [60]. As our results, therefore, suggest a suitable fit of both models, we report the results for a model with two disease subtypes below (see Supplementary S7 for the model with one disease subtype). In this model, 58 participants (25 FEP; 33 MEP) were sorted into Subtype 0, 83 participants (24 FEP; 59 MEP) were sorted into Subtype 1, and 22 participants were sorted into Subtype 2 (6 FEP; 16 MEP).
The Kruskal–Wallis test showed a significant main effect of Subtype on left (H(2) = 52.203, p < .001, η2H = 0.31) and right hippocampal-cortical connectivity (H(2) = 35.592, p < .001, η2H = 0.21). Post-hoc Mann–Whitney U tests revealed that Subtype 2 exhibited significantly lower bilateral hippocampal-cortical connectivity than Subtype 0 and Subtype 1, while these two did not differ significantly. Thus, only patients in Subtype 2 were characterized by impaired hippocampal-cortical connectivity when collapsing across stages. The significant main effect of Subtype on episodic memory (H(2) = 87.867, p < 0.001, η2H = 0.54) and follow-up analyses showed that Subtype 1 showed significantly lower episodic memory than Subtype 0 and Subtype 2, while Subtype 2 also showed lower episodic memory than Subtype 0. A different pattern was observed for social cognition (H(2) = 21.816, p < 0.001, η2H = 0.12) and negative symptoms (H(2) = 26.319, p < 0.001, η2H = 0.15), where follow-up analyses revealed significantly poorer social cognition and higher negative symptoms in Subtype 1 in comparison to Subtype 0 and Subtype 2, while Subtype 0 and Subtype 2 did not differ significantly. Regarding functional outcome (H(2) = 10.860, p = 0.004, η2H = 0.06), Subtype 1 was significantly more impaired than Subtype 0, yet did not differ from Subtype 2, indicating that Subtype 1 scored lowest on functional outcome. See Fig. 2a for a visualization of the follow-up Mann–Whitney U tests and Table 2 for demographic characteristics and statistical comparisons of the subtypes. With regard to subtype differences on diagnosis and depressive symptoms, no significant effects were observed after correcting for multiple comparisons (ps > 0.233).
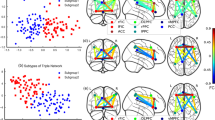
a Shows the mean comparisons between subtypes on the six markers of our model. **p < 0.001, *p < 0.05. b, c Shows the disease progression of the markers in both disease subtypes, respectively. The color coding indicates a deviation from normality by one z-score in blue, by two z-scores in magenta, and by three z-scores or more (up to five) in red. The stronger the color, the higher the probability of individuals at that stage to deviate by one z-score, two z-scores, three z-scores. Subtype 1 progresses from episodic memory to social cognition, negative symptoms, functional outcome, left hippocampal-cortical connectivity, and right hippocampal-cortical connectivity. Subtype 2 progresses from left hippocampal-cortical connectivity to right hippocampal-cortical connectivity, episodic memory and social cognition, functional outcome, and negative symptoms.
As Subtype 0 per definition implies no disease progression, SuStaIn exclusively infers disease progressions for the disease subtypes. For Subtypes 1 and 2, data are provided up to disease stages 9 and 7, respectively, with SuStaIn’s longitudinal inference inferring biomarker progression until disease stage 18. As seen in Fig. 2b, c, disease progression is shown in z-score deviations from the control mean, with blue squares indicating one z-score deviation, magenta indicating two z-score deviations, and red indicating three or more (up to five) z-score deviations. In case no change occurs at a specific stage, the square is left white. The chosen model only allows for up to five z-score deviations from the mean, meaning that i.e., episodic memory and social cognition reach the highest possible deviation at disease stage 6. Higher z-score deviations might still occur in later disease stages yet will not be modeled due to the chosen cut-off score of five in line with prior implementations of SuStaIn [25].
Based on the z-score means per stage (Fig. 3), Subtype 1 shows an early deterioration of episodic memory, which deviates one z-score from the mean at disease stage 1, followed by social cognition, reaching one z-score deviation from the mean at disease stage 4. Negative symptoms and functional outcome then both reach one z-score deviation from the mean at disease stage 8. Left hippocampal-cortical connectivity reaches one z-score deviation from the mean at disease stage 9, while right hippocampal-cortical connectivity never substantially deviates from the mean. The longitudinal inference of SuStaIn shows that it most likely starts deteriorating from disease stage 12 onwards, which is beyond the stages represented in our sample. Subtype 2 shows a deterioration of left hippocampal-cortical connectivity by two z-score deviations from the mean at disease stage 1, while right hippocampal-cortical connectivity follows with one z-score deviation from the mean at disease stage 2. Social cognition reaches one z-score deviation from the mean at disease stage 3, while episodic memory reaches two z-score deviations from the mean at disease stage 4. Given that social cognition and episodic memory, therefore, start deteriorating at around the same stage, yet episodic memory continues to deteriorate while social cognition scores improve again (Fig. 3), we mention episodic memory before social cognition in our disease progression. Negative symptoms and functional outcome do not deviate substantially from the mean in our sample, yet the longitudinal inference of SuStaIn (Fig. 2c) shows that functional outcome deviates slightly earlier. We, therefore, identify a disease progression from episodic memory to social cognition, negative symptoms, functional outcome, left and right hippocampal-cortical connectivity in Subtype 1, and a disease progression from left and right hippocampal-cortical connectivity to episodic memory, social cognition, functional outcome and negative symptoms in Subtype 2 (Fig. 2).
This figure shows how patients are first subtyped into Subtype 0 (white), disease Subtype 1 (yellow), or disease Subtype 2 (blue) and how the disease progression of the respective disease subtypes is then inferred by placing the patients onto a disease stage of this subtype. The numbers on top of the bars reflect the raw number of (FEP/MEP) patients in each stage. The table on the right indicates the percentage of affective and non-affective FEP and MEP patients in each stage.
First- and multi-episode psychosis
FEP and MEP patients are distributed equally across stages (Fig. 3). To identify further similarities, we split both samples into low versus high negative symptoms based on the respective mean value for negative symptoms. Results revealed a significant main effect of negative symptom group (FEPlow (n = 28), FEPhigh (n = 27), MEPlow (n = 53), MEPhigh (n = 55)) on negative symptoms (H(3) = 121.174, p < 0.001, η2H = 0.74) and functional outcome (H(3) = 20.119, p < 0.001, η2H = 0.11). Follow-up results for those variables are visualized in Fig. 4a. The findings of functional outcome suggest that MEP with high negative symptoms are particularly impaired, yet FEP and MEP overall score comparably on most variables, irrespective of negative symptoms. Figure 4b, c further visualize that individuals with low negative symptoms tend to be grouped into earlier disease stages while individuals with high negative symptoms are distributed across all stages and dominate the later disease stages.
Difference to one-subtype model
In a model with one disease subtype, Subtype 1 is characterized by significantly lower episodic memory, social cognition, higher negative symptoms, and poorer functional outcome than Subtype 0, and progresses from episodic memory to social cognition, negative symptoms, functional outcome, left and right hippocampal-cortical connectivity (see Supplementary S7 for the subtype description, T1 for subtype demographics, F4 for a circle diagram showing the patient distribution in a model with one vs. two disease subtypes, and F5 for figures of SuStaIn). While both models show a statistically similar fit for our data, the two-subtypes model seems to uncover the importance of hippocampal-cortical connectivity in leading the disease progression in part of the patient sample.
Discussion
We proposed a multiscale model of psychosis to advance our understanding of functional outcomes of psychosis and explored heterogeneous disease trajectories across patient subtypes through machine-learning. This first implementation of SuStaIn in a multiscale model of psychiatry allowed us to identify three distinct patient subtypes with heterogeneous disease progression patterns. Subtype 0 included patients without impaired connectivity, (social) cognition, symptoms, or functioning. Subtype 1 and Subtype 2 mostly supported our proposed progression model in terms of episodic memory, social cognition, negative symptoms, and functional outcome. Notably, hippocampal-cortical dysconnectivity preceded these markers in Subtype 2, while Subtype 1 showed that hippocampal-cortical dysconnectivity emerged following cognitive and functional impairments and higher symptoms. This suggests that hippocampal-cortical dysconnectivity can but does not necessarily precede impairments at the manifest- and social-scale. These results demonstrate how multiscale frameworks can offer important insights into the complex temporal nature of cross-scale interactions while underlining the need to differentiate between patient subtypes in disorders as heterogeneous as psychosis.
We identified two distinct disease trajectories between our subtypes, with Subtype 1 progressing from episodic memory to social cognition, negative symptoms, functional outcome, and hippocampal centrality, while hippocampal centrality led the disease progression in Subtype 2. These disease progression patterns do not seem to be driven by differences in demographic (i.e., age) or clinical (i.e., diagnoses, depressive symptoms) variables, pointing towards distinct methodological and neurobiological mechanisms underlying these trajectories. Notably, we performed cluster analyses, which could explain why the impairments in hippocampal centrality, memory, and symptoms, as identified by Makowski et al. [15], were spread across our two distinct subtypes. More precisely, we identified Subtype 1 with impaired episodic memory and high negative symptoms and Subtype 2 with impaired hippocampal centrality and episodic memory. While the findings by Makowski et al. [15] might thus indicate a pooling of impairments across the sample, their use of longitudinal data may also point toward impairments at later time points, not captured in the cross-sectional implementation of SuStaIn. This would be in line with the longitudinal inference feature of SuStaIn for Subtype 2, showing an increase in negative symptoms at disease stage 9. These methodological aspects are of particular importance when considering the partial overlap of Makowski’s sample and the sample of Study 1.
That hippocampal dysconnectivity does not lead the disease progression in Subtype 1 might further be explained by other neural markers. Makowski et al. [15] used a quantitative MRI, which is an index of intracortical myelination. Considering other MRI modalities, such as quantitative, diffusion-weighted, or functional MRI, might thus provide a more comprehensive overview of the hippocampal dysfunction in our neurocognitive model and might underlie the disease progression in Subtype 1. In addition, neural correlates of social cognition might drive this progression, such as the amygdala [61], for which dorsomedial shape development has been shown to predict conversion to psychosis [62] while further being associated with poorer emotion recognition [62]. Beyond the association with social cognition, the amygdala and hippocampus are distinctly associated with negative symptoms [63]. Future work aiming to disentangle multiscale contributions to impaired social cognition and episodic memory may wish to consider distinct MRI modalities while also looking at the independent contributions of the amygdala and hippocampus.
We further identified an almost identical temporal development of negative symptoms and functional outcome in both disease subtypes. One explanation might lie in the pooling of SOFAS social and occupational functioning scores [6], which are used to estimate overall functional outcome. With regard to the social functioning component, premorbid social functioning, in particular, has been shown to be predictive of negative symptom severity in later disease stages [64, 65]. While premorbid social functioning and social outcomes of psychosis are two distinct constructs, considering premorbid functioning as a potential confounder may help to disentangle the relationship between symptoms and functioning. We would further like to point out that our key objective was to propose a multiscale perspective on functional outcomes in psychosis, providing a link from the brain to functional outcomes through socio-cognitive deficits and symptoms. We did not intend to provide an exhaustive account of factors influencing functional outcomes of psychosis and recommend that future research considers these aspects further.
Our findings further need to be discussed in the light of clinical staging models. McGorry et al. [40] hypothesize a unidirectional disease progression in which new clinical stages represent a significant change in clinical status [40, 41]. By including patients from two distinct clinical stages and applying a machine-learning algorithm which models unidirectional disease progression, we were in the unique position to address whether McGorry’s clinical stages could be mapped onto the disease stages identified by SuStaIn. As McGorry suggests, MEP patients showed lower episodic memory and functioning and higher negative symptoms, yet FEP and MEP participants were spread equally across SuStaIn subtypes and, most importantly, disease stages. While this seems contradictory to McGorry’s clinical stages at first sight, it is important to differentiate between clinical stages and disease states. Clinical stages carry qualitative value about patient history and inform treatment decisions. Disease states, however, are the patient’s scores on symptom severity, functioning, and cognition at a specific point in time. SuStaIn, therefore, infers longitudinal disease stages from patient states and places individuals with good state performance (i.e., low negative symptoms, high functioning at assessment) in early disease stages and individuals with poor state performance (high symptoms, low functioning) in later disease stages. The assumption underlying this modeling is the one of unidirectionality, implying that individuals who currently have good state performance will later develop poorer state performance, with the degree of impairment and the order of deterioration differing between disease subtypes. By showing that MEP and FEP are placed in the same SuStaIn stages, we show that states across MEP and FEP are comparable, indicating that states can vary within clinical stages. In this sense, our results can be considered a natural extension of McGorry’s clinical stages, contributing to a refining of stage-based frameworks by allowing for a fusion between clinical staging models and biomarker-informed disease trajectories and potentially explaining clinical variation seen within clinical stages.
An additional point to consider is the cyclical nature of psychotic symptomatology. Z-score SuStaIn is well-suited to mimic the unidirectionality of clinical staging models; however, progression patterns might deviate across the scales of our model. When looking at markers such as negative symptoms, symptom severity might not increase but fluctuate over the course of the disease progression [66]. In line with this hypothesis, low and high negative symptoms are associated with distinct degrees of functional impairment, suggesting that the markers might fluctuate depending on symptomatic vs. remitted illness. SuStaIn thus likely sorted remitted MEP patients into early disease stages, as these patient scores had similar mean scores to patients in early clinical stages (FEP). To better capture intra-individual fluctuations within clinical stages, future work should evaluate other disease progression models which do not assume unidirectional disease progression but rather model bidirectional marker fluctuations. Such an approach might allow for an even more reliable modeling of McGorry’s clinical stages while acknowledging the variety of disease states within each clinical stage.
Our findings have important implications for multiscale perspectives on psychosis. To our knowledge, our work addresses a multiscale model of psychiatry using SuStaIn for the first time. Even within multiscale frameworks, the components of clustering in combination with disease progression modeling are essential to capture the complex nature of psychosis. By implementing a mechanistic approach towards disease progression that integrates multiple levels of measurement, we provide a data-driven approach which could complement traditional symptom-based diagnostics [1]. As z-score SuStaIn assigns individuals to distinct disease subtypes and stages, our results also provide clinical utility and translational opportunities for patient stratification and diagnostics based on machine-learning. A good example of clinical utility beyond the implications for McGorry’s clinical staging model [40] is the application of SuStaIn in psychosis by Jiang et al. [38], who found that one of their disease subtypes was related to higher antipsychotic medication efficacy. While we did not find significant differences in medication, future studies in larger samples are warranted. Our findings might further allow for transdiagnostic applications predicting disease progression, considering that hippocampal volume and episodic memory are also highly valuable in major depressive disorder [67, 68]. Lastly, it should be noted that negative and depressive symptoms correlated significantly in FEP, underlining the importance of mood symptoms in early clinical stage. While this might point to potential confounding of negative symptoms and depressive symptoms in FEP, there is also a rich literature on the presence of depressive symptoms early on in psychosis, which diminish following the initiation of treatment [69]. With regard to the role of persistent negative symptoms (PNS) and secondary persistent symptoms (including mood symptoms) in predicting clinical and functional outcomes [70], we encourage future research to investigate the potential of SuStaIn in identifying distinct trajectories of PNS.
Our study has several limitations that need to be considered. We used a novel procedure to reconstruct the SOFAS. While we have statistical support for this method, we nevertheless suggest the replication of this methodology in a larger sample. Further, the SOFAS [6] and SANS [48] were not sampled for the control groups of both studies, and the SOFAS and the SANS patient data had to be z-scored relative to previously reported control data [57, 58]. Our SuStaIn analysis thus had to include patient data only, and our reference for follow-up analyses (Subtype 0) also consisted of patients. Through the future inclusion of a control group, which would most likely be almost entirely subtyped as Subtype 0 [25], it would be possible to compare the deterioration of markers to a more reliable Subtype 0. Additionally, we need to acknowledge the relatively small sample of our study, which consists of 163 patients. According to SuStaIn power estimates, our results are, therefore, slightly underpowered, and we suggest a replication of our findings in a larger sample.
To conclude, as the first investigation of multiscale disease progression in psychosis using SuStaIn, we identified two data-driven disease subtypes with distinct longitudinal disease trajectories. These findings are of particular importance for elucidating the complex and heterogeneous nature of psychosis even within multiscale models and further elucidate the role of hippocampal connectivity and episodic memory in predicting poor functional outcomes. In addition, these are the first data-driven findings to complement traditional clinical staging models by inferring longitudinal disease trajectories across clinical stages. Further research should target broader measures of hippocampal-cortical connectivity through combining distinct neuroimaging modalities and should consider implementing machine-learning models which allow for bidirectional modeling.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to containing sensitive patient information but are available from the corresponding author on reasonable request.
Code availability
The code for the steps of data preparation and SuStaIn has been made publicly available on GitHub (https://github.com/janatotzek/2023-multiscale-markers-psychosis).
References
American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. 5th ed. American Psychiatric Association; 2013.
Heinrichs RW, Zakzanis KK. Neurocognitive deficit in schizophrenia: a quantitative review of the evidence. Neuropsychology. 1998;12:426.
Sommer IE, Bearden CE, Van Dellen E, Breetvelt EJ, Duijff SN, Maijer K, et al. Early interventions in risk groups for schizophrenia: what are we waiting for? NPJ Schizophr. 2016;2:1–9.
Bora E, Lin A, Wood S, Yung A, McGorry P, Pantelis C. Cognitive deficits in youth with familial and clinical high risk to psychosis: a systematic review and meta‐analysis. Acta Psychiatr Scand. 2014;130:1–15.
Vos T, Abajobir AA, Abate KH, Abbafati C, Abbas KM, Abd-Allah F, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390:1211–59.
Goldman HH, Skodol AE, Lave TR. Revising axis V for DSM-IV: a review of measures of social functioning. Am J Psychiatry. 1992;149:1148–56.
Lepage M, Bodnar M, Bowie CR. Neurocognition: clinical and functional outcomes in schizophrenia. Can J Psychiatry. 2014;59:5–12.
Chong HY, Teoh SL, Wu DB-C, Kotirum S, Chiou C-F, Chaiyakunapruk N. Global economic burden of schizophrenia: a systematic review. Neuropsychiatr Dis Treat. 2016;12:357.
Wu EQ, Birnbaum HG, Shi L, Ball DE, Kessler RC, Moulis M, et al. The economic burden of schizophrenia in the United States in 2002. J Clin Psychiatry. 2005;66:1122–9.
van den Heuvel MP, Scholtens LH, Kahn RS. Multiscale neuroscience of psychiatric disorders. Biol Psychiatry. 2019;86:512–22.
Lavigne KM, Kanagasabai K, Palaniyappan L. Ultra-high field neuroimaging in psychosis: a narrative review. Front Psychiatry. 2022;13:994372.
Mišić B, Goñi J, Betzel RF, Sporns O, McIntosh AR. A network convergence zone in the hippocampus. PLoS Comput Biol. 2014;10:e1003982.
Evans AC. Networks of anatomical covariance. Neuroimage. 2013;80:489–504.
Bullmore E, Sporns O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci. 2009;10:186–98.
Makowski C, Lewis JD, Khundrakpam B, Tardif CL, Palaniyappan L, Joober R, et al. Altered hippocampal centrality and dynamic anatomical covariance of intracortical microstructure in first episode psychosis. Hippocampus. 2020;30:1058–72.
Yeo BT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011;106:1125–65.
Valli I, Tognin S, Fusar-Poli P, Mechelli A. Episodic memory dysfunction in individuals at high-risk of psychosis: a systematic review of neuropsychological and neurofunctional studies. Curr Pharm Des. 2012;18:443–58.
Mesholam-Gately RI, Giuliano AJ, Goff KP, Faraone SV, Seidman LJ. Neurocognition in first-episode schizophrenia: a meta-analytic review. Neuropsychology. 2009;23:315.
Raucher-Chéné D, Thibaudeau E, Sauvé G, Lavigne KM, Lepage M. Understanding others as a mediator between verbal memory and negative symptoms in schizophrenia-spectrum disorder. J Psychiatr Res. 2021;143:429–35.
Buck G, Lavigne KM, Makowski C, Joober R, Malla A, Lepage M. Sex differences in verbal memory predict functioning through negative symptoms in early psychosis. Schizophr Bull. 2020;46:1587–95.
Lin CH, Huang CL, Chang YC, Chen PW, Lin CY, Tsai GE. Clinical symptoms, mainly negative symptoms, mediate the influence of neurocognition and social cognition on functional outcome of schizophrenia. Schizophr Res. 2013;146:231–7.
Kharawala S, Hastedt C, Podhorna J, Shukla H, Kappelhoff B, Harvey PD. The relationship between cognition and functioning in schizophrenia: a semi-systematic review. Schizophr Res Cognit. 2022;27:100217.
Devoe DJ, Braun A, Seredynski T, Addington J. Negative symptoms and functioning in youth at risk of psychosis: a systematic review and meta-analysis. Harv Rev Psychiatry. 2020;28:341.
Milev P, Ho B-C, Arndt S, Andreasen NC. Predictive values of neurocognition and negative symptoms on functional outcome in schizophrenia: a longitudinal first-episode study with 7-year follow-up. Am J Psychiatry. 2005;162:495–506.
Young AL, Marinescu RV, Oxtoby NP, Bocchetta M, Yong K, Firth NC, et al. Uncovering the heterogeneity and temporal complexity of neurodegenerative diseases with Subtype and Stage Inference. Nat Commun. 2018;9:1–16.
Green MJ, Girshkin L, Kremerskothen K, Watkeys O, Quidé Y. A systematic review of studies reporting data-driven cognitive subtypes across the psychosis spectrum. Neuropsychol Rev. 2020;30:446–60.
Rocca P, Galderisi S, Rossi A, Bertolino A, Rucci P, Gibertoni D, et al. Social cognition in people with schizophrenia: a cluster-analytic approach. Psychol Med. 2016;46:2717–29.
Strauss GP, Horan WP, Kirkpatrick B, Fischer BA, Keller WR, Miski P, et al. Deconstructing negative symptoms of schizophrenia: avolition–apathy and diminished expression clusters predict clinical presentation and functional outcome. J Psychiatr Res. 2013;47:783–90.
Sauvé G, Malla A, Joober R, Brodeur MB, Lepage M. Comparing cognitive clusters across first-and multiple-episode of psychosis. Psychiatry Res. 2018;269:707–18.
Uren J, Cotton SM, Killackey E, Saling MM, Allott K. Cognitive clusters in first-episode psychosis: overlap with healthy controls and relationship to concurrent and prospective symptoms and functioning. Neuropsychology. 2017;31:787.
Lewandowski K, Sperry S, Cohen B, Öngür D. Cognitive variability in psychotic disorders: a cross-diagnostic cluster analysis. Psychol Med. 2014;44:3239–48.
Young AL, Bocchetta M, Russell LL, Convery RS, Peakman G, Todd E, et al. Characterizing the clinical features and atrophy patterns of MAPT-related frontotemporal dementia with disease progression modeling. Neurology. 2021;97:e941–e52.
Young AL, Vogel JW, Aksman LM, Wijeratne PA, Eshaghi A, Oxtoby NP, et al. Ordinal SuStaIn: subtype and stage inference for clinical scores, visual ratings, and other ordinal data. Front Artif Intell. 2021;4:613261.
Vogel JW, Young AL, Oxtoby NP, Smith R, Ossenkoppele R, Strandberg OT, et al. Four distinct trajectories of tau deposition identified in Alzheimer’s disease. Nat Med. 2021;27:871–81.
Oxtoby NP, Leyland L-A, Aksman LM, Thomas GE, Bunting EL, Wijeratne PA, et al. Sequence of clinical and neurodegeneration events in Parkinson’s disease progression. Brain. 2021;144:975–88.
Fiford CM, Sudre CH, Young AL, Macdougall A, Nicholas J, Manning EN, et al. Presumed small vessel disease, imaging and cognition markers in the Alzheimer’s disease neuroimaging initiative. Brain Commun. 2021;3:fcab226.
Firth NC, Primativo S, Brotherhood E, Young AL, Yong KX, Crutch SJ, et al. Sequences of cognitive decline in typical Alzheimer’s disease and posterior cortical atrophy estimated using a novel event‐based model of disease progression. Alzheimer’s Dement. 2020;16:965–73.
Jiang Y, Wang J, Zhou E, Palaniyappan L, Luo C, Ji G, et al. Neuroimaging biomarkers define neurophysiological subtypes with distinct trajectories in schizophrenia. Nat Mental Health. 2023;1:186–99.
Sone D, Young A, Shinagawa S, Tsugawa S, Iwata Y, Tarumi R, et al. Disease progression patterns of brain morphology in schizophrenia: More progressed stages in treatment-resistance. Schizophr Bull. 2024;50:393–402.
McGorry PD, Purcell R, Hickie IB, Yung AR, Pantelis C, Jackson HJ. Clinical staging: a heuristic model for psychiatry and youth mental health. Med J Aust. 2007;187:S40–S2.
Shah JL, Scott J, McGorry PD, Cross SP, Keshavan MS, Nelson B, et al. Transdiagnostic clinical staging in youth mental health: a first international consensus statement. World Psychiatry. 2020;19:233–42.
Béland S, Lepage M. The relative contributions of social cognition and self-reflectiveness to clinical insight in enduring schizophrenia. Psychiatry Res. 2017;258:116–23.
Iyer S, Jordan G, MacDonald K, Joober R, Malla A. Early intervention for psychosis: a Canadian perspective. J Nerv Mental Dis. 2015;203:356–64.
First MB, Spitzer RL, Gibbon M, Williams JBW. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Patient Edition (SCID-I/P). New York, NY: Biometrics Research; 2010.
First MB, Williams JBW, Karg RS, Spitzer RL. Structured clinical interview for DSM-5—Research version (SCID-5-RV). Arlington, VA: American Psychiatric Association; 2015.
Pietrzak RH, Olver J, Norman T, Piskulic D, Maruff P, Snyder PJ. A comparison of the CogState schizophrenia battery and the measurement and treatment research to improve cognition in schizophrenia (MATRICS) battery in assessing cognitive impairment in chronic schizophrenia. J Clin Exp Neuropsychol. 2009;31:848–59.
Addington D, Addington J, Maticka-Tyndale E. Assessing depression in schizophrenia: the Calgary Depression Scale. Br J Psychiatry. 1993;163:39–44.
Andreasen NC. The scale for the assessment of negative symptoms (SANS): conceptual and theoretical foundations. Br J Psychiatry. 1983;155:49–52.
Andreasen NC. The scale for the assessment of positive symptoms (SAPS). Br J Psychiatry. 1984;155:49–52.
Morosini PL, Magliano L, Brambilla L, Ugolini S, Pioli R. Development, reliability and acceptability of a new version of the DSM‐IV Social and Occupational Functioning Assessment Scale (SOFAS) to assess routine social funtioning. Acta Psychiatr Scand. 2000;101:323–9.
Ad-Dab’bagh Y, Lyttelton O, Muehlboeck J, Lepage C, Einarson D, et al. The CIVET image-processing environment: a fully automated comprehensive pipeline for anatomical neuroimaging research. In Proc: of the 12th annual meeting of the organization for human brain mapping; Florence, Italy: 2006.
Chakravarty MM, Steadman P, Van Eede MC, Calcott RD, Gu V, Shaw P, et al. Performing label‐fusion‐based segmentation using multiple automatically generated templates. Hum Brain Mapp. 2013;34:2635–54.
Pipitone J, Park MTM, Winterburn J, Lett TA, Lerch JP, Pruessner JC, et al. Multi-atlas segmentation of the whole hippocampus and subfields using multiple automatically generated templates. Neuroimage. 2014;101:494–512.
Ajnakina O, Das T, Lally J, Di Forti M, Pariante CM, Marques TR, et al. Structural covariance of cortical gyrification at illness onset in treatment resistance: a longitudinal study of first-episode psychoses. Schizophr Bull. 2021;47:1729–39.
Das T, Borgwardt S, Hauke DJ, Harrisberger F, Lang UE, Riecher-Rössler A, et al. Disorganized gyrification network properties during the transition to psychosis. JAMA Psychiatry. 2018;75:613–22.
Rubinov M, Sporns O. Complex network measures of brain connectivity: uses and interpretations. Neuroimage. 2010;52:1059–69.
Agid O, McDonald K, Siu C, Tsoutsoulas C, Wass C, Zipursky RB, et al. Happiness in first-episode schizophrenia. Schizophr Res. 2012;141:98–103.
Oruç S, Gülseren G, Kusbec O, Özbulut O. An evaluation of neuropsychiatric symptoms in Parkinson’s disease patients. Niger J Clin Pract. 2017;20:900–4.
Aksman LM, Wijeratne PA, Oxtoby NP, Eshaghi A, Shand C, Altmann A, et al. pySuStaIn: a Python implementation of the subtype and stage inference algorithm. SoftwareX. 2021;16:100811.
UCL-Pond. SuStaIn tutorial using simulated data GitHub. 2021. https://github.com/ucl-pond/pySuStaIn/blob/master/notebooks/SuStaIn%20tutorial%20using%20simulated%20data.ipynb.
Jáni M, Kašpárek T. Emotion recognition and theory of mind in schizophrenia: a meta-analysis of neuroimaging studies. World J Biol Psychiatry. 2018;19:S86–S96.
Guimond S, Mothi SS, Makowski C, Chakravarty MM, Keshavan MS. Altered amygdala shape trajectories and emotion recognition in youth at familial high risk of schizophrenia who develop psychosis. Transl Psychiatry. 2022;12:1–8.
Makowski C, Bodnar M, Shenker J, Malla A, Joober R, Chakravarty M, et al. Linking persistent negative symptoms to amygdala–hippocampus structure in first-episode psychosis. Transl Psychiatry. 2017;7:e1195.
Addington J, Addington D. Patterns of premorbid functioning in first episode psychosis: relationship to 2‐year outcome. Acta Psychiatr Scand. 2005;112:40–6.
Bucci P, Galderisi S, Mucci A, Rossi A, Rocca P, Bertolino A, et al. Premorbid academic and social functioning in patients with schizophrenia and its associations with negative symptoms and cognition. Acta Psychiatr Scand. 2018;138:253–66.
Ventura J, Subotnik KL, Gitlin MJ, Gretchen-Doorly D, Ered A, Villa KF, et al. Negative symptoms and functioning during the first year after a recent onset of schizophrenia and 8 years later. Schizophr Res. 2015;161:407–13.
Campbell S, MacQueen G. The role of the hippocampus in the pathophysiology of major depression. J Psychiatry Neurosci. 2004;29:417–26.
Jayaweera H, Hickie I, Duffy S, Mowszowski L, Norrie L, Lagopoulos J, et al. Episodic memory in depression: the unique contribution of the anterior caudate and hippocampus. Psychol Med. 2016;46:2189–99.
Pelizza L, Leuci E, Quattrone E, Azzali S, Paulillo G, Pupo S, et al. Depressive features in individuals with first episode psychosis: psychopathological and treatment considerations from a 2-year follow-up study. Clin Neuropsychiatry. 2023;20:39.
Lepage M, Bodnar M, Raucher-Chéné D, Lavigne KM, Makowski C, Joober R, et al. Neurocognitive functions in persistent negative symptoms following a first episode of psychosis. Eur Neuropsychopharmacol. 2021;47:86–97.
Acknowledgements
We would like to thank all participants who took part in the two studies which contributed to our project. We would also like to thank PEPP-Montréal and the CRISP Research Lab of the Douglas Research Centre for participant recruitment and data collection. We would also like to thank Caroline Dakoure and Joshua Unrau for their support with data preparation and Karyne Anselmo for her help with reconstructing the SOFAS. A special thank you also goes to Alan C. Evans for early methodological guidance. We used the high-performance computing resources available via the Digital Research Alliance of Canada (https://alliancecan.ca/en).
Funding
Funding Study 1 was funded by the Canadian Institutes of Health Research in collaboration with the Otsuka Lundbeck Alliance (#141636), while Study 2 was funded by the Canadian Institutes of Health Research (#106434) and the Otsuka/Lundbeck Alliance (#20135257). This research was undertaken thanks in part to funding from the Canada First Research Excellence Fund and Fonds de recherche du Québec, awarded to the Healthy Brains, Healthy Lives initiative at McGill University. The funding agencies did not influence the study design, data collection or writing of the manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization: ML, KML, JFT. Supervision: ML, KML, DH. Writing—Original Draft: JFT. Formal analysis: JFT. Writing—Review & Editing: JFT, KML, RJ, AM, JLS, ML, DH, ALY, DR. Software: JFT, KML, ALY, MMC. Methodology: JFT, KML, ALY. Visualization: JFT. Funding Acquisition: ML, MMC, RJ, AM, JLS.
Corresponding author
Ethics declarations
Competing interests
ML holds salary awards through the James McGill Professorship and reports grants from Otsuka Lundbeck Alliance, Hoffman-La Roche, personal fees from Lundbeck Canada, personal fees from Otsuka Canada, personal fees from Boehringer-Ingelheim, grants and personal fees from Janssen outside the submitted work. KML reports personal fees from Otsuka Canada, Lundbeck Canada, and Boehringer Ingelheim. DH has received financial compensation as a consultant for P1vitalProducts Ltd. These activities were unrelated to the work presented in this manuscript. JLS holds a salary award from the Fonds de recherche du Québec—Santé. AM reports receipt of grants, fees or honoraria from Lundbeck and Otsuka and salary awards by the Canada Research Chairs program. MMC holds salary awards from the Fonds de Recherche du Québec—Santé and reports funding from the Canadian Institute of Health Research, the Natural Sciences and Engineering Research Council of Canada, the Weston Brain Institute, Healthy Brains Healthy Lives, and the Fonds de Recherche du Québec—Santé. RJ served as member of advisory board committees and speaker for Bristol Myers Squibb, Pfizer, Sunovian, Janssen, Myelin and Associates, Lundbeck, Otsuka, Shire, and Perdue, and received grants from Janssen, Otsuka, Lundbeck, Bristol Myers Squibb, Astra Zeneca, and HLS Therapeutics Inc. ALY was supported by a Skills Development Fellowship (MR/T027800/1) from the Medical Research Council and a Career Development Award from the Wellcome Trust [227341/Z/23/Z]. This research was funded in whole, or in part, by the Wellcome Trust [227341/Z/23/Z]. For the purpose of open access, the author has applied a CC BY public copyright license to any Author Accepted Manuscript version arising from this submission. JFT reports receipt of the Healthy Brains Healthy Lives Graduate Student Fellowship. All of these disclosures are unrelated to the present study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Totzek, J.F., Chakravarty, M.M., Joober, R. et al. Longitudinal inference of multiscale markers in psychosis: from hippocampal centrality to functional outcome. Mol Psychiatry (2024). https://doi.org/10.1038/s41380-024-02549-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41380-024-02549-x