Abstract
Family history of hypertension is associated with early autonomic dysfunction and increased oxidative stress. These alterations have been found to be reinforced by the overweight factor. Conversely, an active lifestyle is effective in improving the mechanisms regulating blood pressure control. Hence, we ought to investigate the effects of an active lifestyle on the hemodynamic, autonomic and oxidative stress parameters in individuals carrying both family history of hypertension and overweight risk factors. Fifty-six normotensive males were divided into four groups: eutrophic offspring of normotensive parents (EN, n = 12), eutrophic and inactive with hypertensive parents (EH, n = 14), overweight and inactive with hypertensive parents (OH, n = 13), and overweight and physically active with hypertensive parents (OAH, n = 17). Cardiovascular autonomic modulation was assessed by heart rate (HRV) and blood pressure (BPV) variability indexes. Oxidative stress included pro/antioxidant markers and nitrite concentration. Inactive offspring of hypertensive parents (EH and OH) showed higher LFSBP (vs EN), an indicator of sympathetic outflow to the vasculature and reduced anti-oxidant activity (vs EN), while higher pro-oxidant markers were found exclusively in OH (vs EN and EH). Conversely, the OAH group showed bradycardia, higher vagally-mediated HFabs index (vs OH and EN), lower sympathovagal balance (vs OH) and preserved LFSBP. Yet, the OAH showed preserved pro/antioxidant markers and nitrite levels. Our findings indicates that overweight offspring of hypertensive parents with an active lifestyle have improved hemodynamic, cardiac autonomic modulation and oxidative stress parameters compared to their inactive peers.
Similar content being viewed by others
Introduction
Individuals with a positive family history of hypertension are at genetic risk to develop systemic arterial hypertension [1, 2]. However, it is well established that genetic heritability is not the only factor responsible for the onset of hypertension, as environmental factors account for approximately 50% of the increase in blood pressure (BP) [2]. Hence, modifiable risk factors, such as overweight and sedentary behavior have been shown to play a crucial role in modulating the determinants of BP [3]. In this sense, it is well established that weight gain is strongly associated with an increase in BP and the establishment of hypertension [4]. This correlation is particularly critical to individuals with a family history of hypertension [1, 3]. As such, a 5-year follow-up cohort study indicated that offspring of hypertensive parents were not only genetically prone to develop hypertension but also had the propensity to accumulate central body fat, which over time, was accompanied by an exaggerated BP response to exercise even prior any significant rise in resting parameters [3].
Augmented cardiac and vascular sympathetic activity and reduced cardiac vagal activity seem to play a crucial role in the progressive increase in BP in the offspring of hypertensive parents [5, 6]. In addition, results from pre-clinical [7] and clinical models [8] indicate that increased oxidative stress resulting from the imbalance between prooxidants and antioxidants might be a key mediator of endothelial injury and vascular remodeling in the pathology of hypertension. These early disarrangements are also present in individuals carrying the ‘overweight risk factor’ [5]. In this sense, our group previously demonstrated that males with overweight and a family history of hypertension (as overlapping or in isolation) presented with early overall cardiac autonomic dysfunction, assessed by indexes of the heart rate variability (HRV) and increased pro-oxidant mediators [5]. Although the overlapping risk factors did not result, necessarily, in an additive effect for cardiovascular outcomes in normotensive adults [5], it is possible that, over time, the gain of weight may reinforce or even amplify the long-term alterations already carried by the genetic component, leading to the establishment of hypertension in this population [1, 3].
Sedentary behavior is another modifiable risk factor known to have multiple detrimental cardiovascular [9] and metabolic effects [10], which are correlated to the development of cardiovascular diseases and all-cause of mortality [11]. Conversely, having an active lifestyle improves the mechanisms underpinning BP control in both healthy [12] and clinical populations [13]. As such, in overweight normotensive subjects, increased physical activity levels are associated with lower BP, which is likely due to improvement in autonomic branches and endothelial function [13]. Specifically, in offspring of hypertensive parents, our group demonstrated that while inactive individuals showed higher diastolic BP (DBP), cardiac sympathetic modulation and systemic reactive oxygen species (ROS) compared to offspring of normotensive parents, these early alterations were partially normalized/prevented in the group of physically active peers [9].
However, it is yet to be determined whether an active lifestyle would overcome the early systemic alterations established by overlapping risk factors, i.e, the combination of positive family history of hypertension and overweight. Thus, we hypothesized that overweight offspring of hypertensive parents with an active lifestyle would have improved hemodynamic, cardiac autonomic modulation and oxidative stress parameters compared to their inactive peers.
Methods
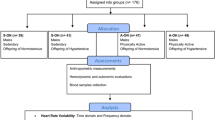
Subjects and study design
This was a cross-sectional, observational study conducted with male students from a University Center (Universidade Nove de Julho, Sao Paulo, Brazil; UNINOVE ethical board approval: CAE 49446315.5.0000.5511). Participants were selected through a preliminary questionnaire evaluating the family history of hypertension (one or both parents) and established diseases, body mass and height, as well as willingness to participate in the study. Eligibility criteria were as follows: (a) age between 18 and 35 y.o; (b) body mass index (BMI) between 18.5 and 29.9 Kg/m2 [14]; not engaged in professional exercise training. Exclusion criteria included: history of chronic diseases, BMI equivalent to obesity type I or above, smokers /significant smoking history, deceased or unknown parents, and acute or chronic use of medications which could interfere with BP levels.
Following the preliminary screening and subjects’ informed consent obtention, 56 male subjects were divided into the following 4 groups: a eutrophic and inactive group with normotensive parents (EN, n = 12), a eutrophic and inactive group with hypertensive parents (EH, n = 14), overweight and inactive group with hypertensive parents (OH, n = 13), and overweight and physically active group with hypertensive parents (OAH, n = 17). Sample size was chosen based on the autonomic/oxidative stress data (mean, standard deviation and effect size) of a previous study using a similar population [5].
Experimental protocol
The participants underwent two visits to the laboratory over a week period, in accordance with the following order: [1] assessment of physical activity level and resting autonomic function and [2] assessment of the body mass composition followed by blood sampling collection. For all visits, subjects were asked to abstain from caffeinated and alcoholic beverages and vigorous exercise for 24 h before testing.
Physical activity level
Physical activity level was established by the International Physical Activity Questionnaire (IPAQ, short form) [15]. The IPAQ-SF measures the number and duration of any walking and other moderate-to-vigorous physical activities that lasted more than 10 min in the previous 7 days and can be used to calculate the metabolic equivalent minutes per week (METs/min/week). Based on the answers, the overall total physical activity score was computed as the sum of Total (Walking + Moderate + Vigorous) MET- minutes/week scores and subjects were classified as active/moderately active (≥600 METs/min/week) or inactive (<600 METs/min/week) [15].
Body composition assessment
Anthropometric measurements
Weight, height and BMI were obtained by established methods [16] and subjects were classified as eutrophic (18.5–24.9 kg/m2) or overweight (25–29.9 kg/m2). Waist/hip circumferences ratio (WC/HC) was obtained from the midpoint of the iliac crest to the last rib, and in the greater trochanter, respectively [5]. Body fat percentage was established by skinfold thickness measurements from the triceps and subscapular region in non-consecutive triplicate (Cescorf ®) [16].
Bioelectrical impedance analysis
Whole-body bioelectrical impedance was obtained in accordance with standard procedures (BIA 450, Biodynamics, USA) [5]. Individuals were instructed to abstain from water for four hours and to empty their bladder 30 min prior to the procedure. Emitter electrodes were placed close to the metacarpal phalangeal joint of the right hand and distally from the transverse arch of the right foot. The detector electrodes were placed between the distal prominences of the radius and ulna of the right wrist and between the medial and lateral malleoli of the right ankle [5].
Hemodynamic and autonomic measurements
Acquisition
Prior to data collection, subjects sat on a chair with a backrest, with their feet flat on the floor and their thighs parallel to the ground. Then, subjects were evaluated at rest for 20 minutes. Throughout the protocol, beat-by-beat BP was measured at the middle finger of the right hand using photoplethysmography (Finapres Medical Systems, The Netherlands) and recorded at 1000 Hz. BP peaks, nadirs and pulse interval (PI) were automatically identified via Beatscope Software to quantify systolic BP (SBP), DBP and heart rate (HR) [17].
Autonomic data analyses
PI and SBP time series were transferred to CardioSeries Software [9]. Resting HRV and BP variability (BPV) data were analyzed in the time and frequency domain according to guidelines [18]. For the time-domain HRV index, the square root of mean squared differences of successive PI (RMSSD) was calculated [18]. For the frequency-domain indexes, PI and SBP time series were re-sampled by a 4 Hz-cubic spline interpolation and a Hanning window was applied. Segments of 256 beats with 50% overlap were then analyzed using the Fast Fourier Transform algorithm. Following the calculation of PI and SBP variances (ms2 and mmHg2, respectively), low-frequency (LF; 0.04–0.15 Hz) and high-frequency (HF; 0.15–0.4 Hz) bands were expressed as absolute (ms2) and normalized (nu) units, as well as LF/HF ratio. The PI variance, RMSSD and HFPI indexes were considered surrogates of the parasympathetic modulation [19], whereas LFPI were considered to be jointly mediated by both sympathetic and vagal influences, with sympathetic predominance [19]. The LF/HF were considered an estimation of the interaction between vagal and sympathetic influences on the cardiac pacemaker [19, 20] and the LFSBP (mmHg2), an index of sympathetic vasomotor modulation [20].
Blood sampling and oxidative stress analyses
Blood samples were collected after a 12-h overnight to measure hematological and biochemical parameters. Glucose, triglycerides, total cholesterol, HDL, LDL, urea, creatinine and glycated hemoglobin (HbA1C) were obtained. Withal, plasma samples were collected for the determination of oxidative stress damage, and anti and pro-oxidant profiles. All participants were advised to avoid foods rich in nitrates (beet, cabbage, spinach) throughout the day prior to blood collection.
Protein concentration was determined according the method described by Lowry et al. [21]. Oxidative damage markers included lipoperoxidation by thiobarbituric acid reactive substances (TBARS), as well as protein oxidation via carbonyls assay. Pro-oxidant markers included the NADPH oxidase and hydrogen peroxide. Anti-oxidant activity included ferric reducing/antioxidant power (FRAP), catalase (CAT) and superoxide dismutase (SOD). In addition, nitrite concentration was measured as a surrogate of nitric oxide (NO) metabolization. The analyses of the aforementioned markers were conducted in accordance with Santa-Rosa et al. [9] and Viana et al. [5].
Statistical analyses
Variables are presented as mean ± standard deviation (SD). Data normality and the variance homogeneity were checked using Shapiro-Wilk’s and Levene’s tests, respectively. The main outcomes did not present normal distribution, hence all autonomic and oxidative stress parameters were log-transformed (ln). The difference between groups was analyzed via Welch ANOVA followed by Games-Howell post hoc (unequal variances) or classic one-way ANOVA followed by Tukey’s post hoc test (equal variances).
To evaluate the main effects of parental history of hypertension, overweight and physical inactivity to the cardiovascular parameters, these predictors were entered into a multiple linear regression model. Additionally, a second model was created for the following interactions: history of hypertension x overweight and history of hypertension x overweight x physical inactivity. The level of significance was set at P ≤ 0.05. Analyses were performed on JAMOVI software (version 2.3.26).
Results
Subjects characteristics
The groups had similar ages, as displayed in Table 1. As expected, overweight groups (OH and OAH) had higher BMI, WC/HC and percentage of fat mass compared to eutrophic groups (EN and EH). The IPAQ total score confirmed the ‘physically active’ status of the OAH and the ‘inactive’ status of the remaining groups. Note that OAH showed lower fat mass compared to the OH group, as demonstrated by the skinfold thickness.
Effect of an active lifestyle on biochemical parameters
All groups showed blood biochemical parameters within the limits of normality (Table 2). However, EH showed a tendency for higher glucose compared to EN (P = 0.06) and the OH group showed lower HDL compared to both eutrophic groups (EH and EN). Conversely, the physically active group (OAH) showed higher HDL compared to the OH and similar values to the EN group. The LDL levels did not differ between OH and OAH groups; however, they remained higher in the OAH group compared to inactive eutrophic groups (EH and EN). The remaining biochemical outcomes did not change among groups, regardless of the physical activity level.
Effect of an active lifestyle on hemodynamic and autonomic function
All groups showed similar SBP and DBP (Table 1). The HR did not differ between inactive groups (EN, EH, OH). However, the physically active group (OAH) showed lower HR compared to both EH and OH (Table 1). As determined by the multiple linear regression (Table 3), the relationship between positive parental history of hypertension and SBP reached borderline significance. Furthermore, being physically inactive was independently associated with an increase in HR.
The HRV and BPV indexes are detailed in Fig. 1. All inactive groups (EN, EH and OH) showed similar values for HRV indexes (Fig. 1A–H). However, the physically active overweight group (OAH) showed higher HFPI-abs compared to OH and EN, as well as lower LF/HF compared to EH and OH groups (Fig. 1E, H, respectively). Inactive offspring of hypertensive parents, regardless of the weight (EH and OH), had higher LFSBP compared to EN; however, the OAH showed lower LFSBP compared to EH and similar values to the EN group (Fig. 1I).
A PI variance, pulse interval variance; (B): SBP variance, systolic blood pressure variance; (C): RMSSD, root mean square of successive differences between RR intervals; (D): LF, low frequency power of HRV expressed as absolute units; (E): HF, high frequency power of HRV expressed as absolute units; (F): LF, low frequency power of HRV expressed as normalized units; (G): HF, high frequency power of HRV expressed as normalized units; (H): LF/HF ratio obtained from absolute values; (I): LF-SBP, low frequency power of systolic blood pressure. EN, eutrophic offspring of normotensive parents; EH, eutrophic offspring of hypertensive parents; OH, overweight offspring of hypertensive parents; OAH, overweight and physically active offspring of hypertensive parents. Data presented as mean ± SD. Analysis: Natural log transformation (Ln) followed by a Welch’s ANOVA with Games-Howell post hoc (unequal variances) or classic one-way ANOVA with Tukey’s post hoc test (equal variances). *P ≤ 0.05 vs EN group; †P < 0.05 vs EH group; #P ≤ 0.05 vs OH group.
A positive parental history of hypertension was associated with increased LFPI-abs and LFSBP. Overweight and physical inactivity status were independently associated with a reduction in HFPI-abs. Being physically inactive was additionally associated with an increase in LF/HF and in LFSBP.
Effect of an active lifestyle on oxidative stress parameters
Pro-oxidant/antioxidant markers are detailed in Fig. 2(A–C and D–F, respectively). The OH had higher pro-oxidant activity compared to eutrophic groups (EH and/or EN), as demonstrated by hydrogen peroxide (Fig. 2A), nitrite (Fig. 2B) and NADPH oxidase (Fig. 2C). In addition, both OH and EH had lower SOD, an antioxidant activity marker, compared to EN (Fig. 2E). However, the physically active group (OAH) did not present the aforementioned alterations, as demonstrated by the lower hydrogen peroxide, nitrite and NADPH, as well as higher SOD compared to OH group. In parallel, all these variables in OAH presented similar values to NE (Fig. 2A, B, C, E, respectively). Finally, oxidative damage markers did not differ between groups, as demonstrated by TBARS (EN: 0.5 ± 0.2; EH: 0.5 ± 0.3; OH: 0.5 ± 0.2; OAH: 0.4 ± 0.1 nmol/mg protein; P = .69) and carbonyls (EN: 1.3 ± 0.2; EH: 1.8 ± 0.6; OH: 1.7 ± 0.5; OAH: 1.5 ± 0.6 nmol/mg protein; P = 0.19).
A Hydrogen peroxide concentration; (B) nitrite concentration; (C) NADPH oxidase activity; (D) catalase concentration; (E): superoxide dismutase activity; (F) FRAP activity. EN, eutrophic offspring of normotensive parents; EH, eutrophic offspring of hypertensive parents; OH, overweight offspring of hypertensive parents; OAH, overweight and physically active offspring of hypertensive parents. Data presented as mean ± SD. Analysis: Natural log transformation (Ln) followed by one-way ANOVA with Tukey’s post hoc test. *P ≤ 0.05 vs EN group; †P ≤ 0.05 vs EH group; #P ≤ 0.05 vs OH group.
A positive parental history of hypertension was inversely associated to the antioxidant SOD. The overweight and physical inactivity predictors were positively associated with pro-oxidant markers nitrite and NADPH oxidase and inversely associated with SOD. Being physically inactive was also associated with a reduction in FRAP. The predictor interactions were not significant for any of oxidative stress parameters.
Discussion
We observed that inactive offspring of hypertensive parents, regardless of the weight status (EH and OH), showed higher LFSBP (vs EN), an indicator of increased sympathetic outflow to the vasculature, as well as reduced antioxidant activity (vs EN), while higher pro-oxidant markers were found exclusively in OH (vs EN and EH). Conversely, the OAH group showed bradycardia and higher vagally-mediated HFabs index (vs OH and EN), lower sympathovagal balance (vs OH) and preserved LFSBP. In addition, these individuals showed preserved pro and antioxidant markers and nitrite concentration, presenting similar values to NE. Taking together, our findings corroborate the hypothesis that overweight offspring of hypertensive parents with an active lifestyle would have improved hemodynamic, cardiac autonomic modulation and oxidative stress parameters compared to their inactive peers.
Early alterations in autonomic modulation and oxidative stress in overweight offspring of hypertensive parents
Studies conducted by our group [5, 9] and others [6] have shown that early alterations of the sympathetic and vagal branches to the heart are one of the mechanisms underpinning the long-term increase in BP in offspring of hypertensive parents. In addition, studies suggest that overlapping risk factors, such as overweight, might act reinforcing these alterations already carried by the heritable component [3, 5]. Indeed, we found that a positive history of hypertension was independently associated with increased LFPI - a marker of overall cardiac dysfunction with sympathetic predominance [19]- and LFSBP - a marker of sympathetic efferent activity to the peripheral vasculature [20]. The overweight risk factor, alone, showed impact on the vagal branch, as demonstrated by its negative correlation with the HFPI.
Aligned with these observations,we found an increased LFSBP in offspring of hypertensive parents, regardless of the weight. As LFSBP are greatly modified by sympathetic pharmacological blockade [20], our data support the enhanced sympathetic vasomotor outflow in these individuals and adds the information that the sympathoexcitation in overweight offspring of hypertensive parents might not be limited to the heart. Given that increased BPV has been described in established essential hypertension [20] and associated with end-organ damage [22], our findings carry clinical value. Interestingly, we showed that the overweight factor did not have an amplifier effect over the genetic component, as the autonomic dysfunction happened regardless of the weight. Similar findings were previously observed by our group [5] and might show that among normotensive young adults, the overweight might reinforce rather than amplify these early autonomic disarrangements.
Oxidative stress is another feature of hypertension [7] that has been early identified in offspring of hypertensive parents [9] and might be aggravated by the increase in body fat [5, 23]. Indeed, we found that a positive history of hypertension was inversely associated with antioxidant activity (SOD) and overweight was independently associated with higher pro-oxidant markers. When comparing all groups, we found that the inactive OH group had increased nitrite and NADPH oxidase compared to EH and EN, suggesting an additive effect of the overweight factor over these variables in offspring of hypertensive parents (OH vs EH).
Our data might be partially mediated by the lower levels of circulating HDL (as observed in OH group), as modification in lipid profile has been shown to promote ROS in the endothelium [24]. In addition, excessive adipose tissue has been found to stimulate the production of ROS and to reduce the activity of antioxidant enzymes (such as SOD) in the adipose cells [25]. In parallel, increased nitrite, a surrogate of NO metabolization, is positively correlated to body fat [23] and to SBP [26]. Obesity‐induced inflammatory cytokines have been found to increase the expression of inducible NO synthase (iNOS), which in excess, damages normal tissues [27]. Thus, higher levels of nitrite could reflect the iNOS overexpression, the NO production in the adipose tissue [23] or a compensatory role of NO to an excessive vasoconstrictor response mediated by increased ROS in OH group [28]. It should be noted that the excessive NO actively reacts with superoxide to produce peroxynitrate, a highly cytotoxic reactive nitrogen species implicated in vascular dysfunction [27].
Evidence in experimental model suggests that oxidative stress precedes and likely mediates the sympathetic overactivity in arterial hypertension [29]. In this sense, depletion of scavengers of ROS, such as SOD, might have particular relevance in the autonomic dysfunction. For example, Tempol (a SOD mimetic) infused intracerebroventricularly reduced renal sympathetic nerve activity (RSNA) and SBP in rats [29]. Knowing that SOD was reduced in offspring of hypertensive parents (regardless of the weight), we suggest that the increase in LFSBP might be partially mediated by the early endothelial dysfunction. The autonomic dysfunction and oxidative stress preceded any detectable alteration in BP values. Although this study did not intend to evaluate the time course of these alterations, it is plausible that overtime, the overweight factor might act amplifying the dysfunction in the mechanisms underpinning the control of BP, contributing to the establishment of hypertension.
Impact of an active lifestyle on cardiovascular and oxidative stress in overweight offspring of hypertensive parents
We found that being physically inactive was associated with several markers of autonomic dysfunction and oxidative stress. Conversely, there is consistent evidence from our group [9] and others [30] indicating that in subjects with parental hypertension, an active lifestyle or chronic exercise is associated with improvement in cardiac autonomic modulation and oxidative stress biomarkers.
To the best of our knowledge, however, this is the first study investigating the effect of an active lifestyle on hemodynamic, autonomic modulation and oxidative stress in individuals carrying overlapping parental hypertension and overweight risk factors. Specifically, physically active OH showed lower HR which was followed by higher in HFabs and lower LF/HF compared to inactive OH. The continuous control of HR is influenced by the vagal and sympathetic autonomic branches. In this sense, the HF index is greatly reduced by muscarinic receptors blockade [31], and consequently, has been considered an indicator of cardiac vagal control [18]. Thus, our data suggest that the bradycardia achieved by the active lifestyle might be partially driven by the increase in cardiac vagal modulation.
Physiological interpretation of LF/HF ratio—also referred to as sympathovagal balance—remains controversial; however, evidence with pharmacological blockade suggests that this index could reflect both vagal and sympathetic influences on the cardiac pacemaker and its reduction might reflect an overall improvement in cardiac autonomic modulation in OAH [18, 20]. Yet, the preserved LFSBP in OAH suggests that maintaining an active lifestyle prevented the increase in sympathetic vasomotor drive previously observed in inactive OH [20]. Indeed, consistent evidence shows that healthy adults with an active lifestyle demonstrate improved autonomic function than their sedentary peers [12, 32]. As resting HR and vagal modulation indexes independently predict mortality in cardiovascular a healthy cohort [33] and increased sympathetic drive is a well-known mechanism underlying the onset of hypertension and target organ damage [22], our findings might hold clinical relevance.
Overweight offspring of hypertensive parents that were physically actives did not present any of the alterations in oxidative stress markers previously observed in their inactive peers (OH). Indeed, it has been shown that modifiable lifestyle behaviors play an important role in modulating oxidative stress and endothelial function [7]. As such, being physically active or lowering sedentary time have been shown to improve the blood flow and laminar shear stress in conduit arteries [34] and microcirculation, which in turn, upregulates the antioxidant defences (such as SOD) and reduces the ROS (such as NADPH oxidase) in the endothelium [7]. In parallel, as alterations in lipid profile have been found to stimulate the production of ROS, our findings might also be due to the HDL amelioration in OAH [24]. Finally, the marked improvement in oxidative stress might be directly linked to the lower body fat mass in OAH, as NADPH oxidase, often increased by adipocytes and implicated in the generation of ROS [25], was reduced in OAH compared to the inactive peers. Indeed, Allemann et al. [1, 3] previously showed that as long as the offspring of hypertensive parents preserved their leanness, only minor BP elevation within normal limits was observed, which highlights the role of fat mass on the mechanisms underpinning the control of BP.
Limitations
This study was designed to follow up on the findings of autonomic dysfunction/oxidative stress in males previously reported by our group [5]. However, it is crucial to account for sex as a biological variable, as men and women differ in the prevalence and pathophysiology of hypertension [35]. Hence, the main limitation of our study lies in the lack of female participants. While we explored the benefits of an active lifestyle on mechanisms controlling BP, these findings are limited to only a portion of those carrying the hypertension risk factors.
Secondly, we could not exclude the possibility of underreporting evaluation of the parental history of hypertension as we relied solely on questionnaires to assess the status of parental hypertension. A questionnaire designed to be responded by the parents rather than by the offspring would have been desirable. Thirdly, the onset of hypertension is multifactorial and thus, we could not rule out that other non-modifiable and environmental factors, such as dietary preferences, could play a role in our findings [36].
Finally, the level of physical activity was measured by IPAQ rather than pedometers and accelerometers. However, consistent evidence supports the IPQA-short version as a reproducible and valid tool for monitoring population levels of physical activity among 18- to 65-yr-old adults in diverse settings [15].
Conclusion and implications
Our findings indicate that inactive offspring of hypertensive parents, regardless of the weight, had increased sympathetic vasomotor drive compared to EN. In addition, although presenting BP within normal values, this population showed early markers of oxidative stress compared to eutrophic groups. Such mechanisms are tightly implicated in the progressive increase in BP among those carrying the heritable component [1, 5, 6]. Thus, it is plausible that over time, the overweight factor might amplify these alterations, contributing to the establishment of hypertension [1, 3, 4]. Conversely, overweight offspring of hypertensive parents with an active lifestyle showed improved hemodynamic, cardiac autonomic modulation and oxidative stress parameters compared to their inactive peers. Altogether, our findings highlight the impact of modifiable risk factors in the mechanisms underlying cardiovascular regulation and provide support for future studies focused on lifestyle interventions for those carrying the parental hypertension and overweight risk factors.
Summary
What is known about topic
-
Positive family history of hypertension is associated with early autonomic dysfunction and increased oxidative stress.
-
Cardiovascular alterations and oxidative stress in the offspring of hypertensive parents are complemented and even amplified by the overweight factor.
-
An active lifestyle is a well-known approach to improving the mechanisms regulating blood pressure control in different populations.
What this study adds
-
Inactive offspring of hypertensive parents, regardless of the weight, present with increased sympathetic outflow to the vasculature.
-
Inactive males with an overlapping overweight and family history of hypertension show early markers of oxidative stress compared to eutrophic males.
-
An active lifestyle improves and partially normalizes the hemodynamic, autonomic and oxidative stress parameters among males with overweight and positive history of hypertension.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Allemann Y, Weidmann P. Cardiovascular, metabolic and hormonal dysregulations in normotensive offspring of essential hypertensive parents. J Hypertens. 1995;13:163–73.
Butler MG. Genetics of hypertension. Curr status J Med Liban. 2010;58:175–78.
Allemann Y, Hutter D, Aeschbacher BC, Fuhrer J, Delacretaz E, Weidmann P. Increased central body fat deposition precedes a significant rise in resting blood pressure in male offspring of essential hypertensive parents: a 5 year follow-up study. J Hypertens. 2001;19:2143–48.
Cassano PA, Segal MR, Vokonas PS, Weiss ST. Body fat distribution, blood pressure, and hypertension. A prospective cohort study of men in the normative aging study. Ann Epidemiol. 1990;1:33–48.
Viana A, Dias DDS, Nascimento MC, Dos Santos F, Lanza FC, Irigoyen MC, et al. Impact of overweight in mens with family history of hypertension: early heart rate variability and oxidative stress disarrangements. Oxid Med Cell Longev. 2020;2020:3049831.
Piccirillo G, Viola E, Nocco M, Durante M, Tarantini S, Marigliano V. Autonomic modulation of heart rate and blood pressure in normotensive offspring of hypertensive subjects. J Lab Clin Med. 2000;135:145–52.
Man AWC, Li H, Xia N. Impact of lifestyles (diet and exercise) on vascular health: oxidative stress and endothelial function. Oxid Med Cell Longev. 2020;2020:1496462.
Campbell R, Fisher JP, Sharman JE, McDonnell BJ, Frenneaux MP. Contribution of nitric oxide to the blood pressure and arterial responses to exercise in humans. J Hum Hypertens. 2011;25:262–70.
Santa-Rosa FA, Shimojo GL, Dias DS, Viana A, Lanza FC, Irigoyen MC, et al. Impact of an active lifestyle on heart rate variability and oxidative stress markers in offspring of hypertensives. Sci Rep. 2020;10:12439.
Brocklebank LA, Falconer CL, Page AS, Perry R, Cooper AR. Accelerometer-measured sedentary time and cardiometabolic biomarkers: A systematic review. Prev Med. 2015;76:92–102.
Ekelund U, Steene-Johannessen J, Brown WJ, Fagerland MW, Owen N, Powell KE, et al. Does physical activity attenuate, or even eliminate, the detrimental association of sitting time with mortality? A harmonised meta-analysis of data from more than 1 million men and women. Lancet (Lond, Engl). 2016;388:1302–10.
Jennings GL, Nelson L, Esler MD, Leonard P, Korner PI. Effects of changes in physical activity on blood pressure and sympathetic tone. J Hypertens Suppl. 1984;2:S139–41.
Machado EA, Farinatti P, Sicuro FL, Rodrigues F Jr, Bouskela E, Collett-Solberg PF. Daily physical activity, cardiorespiratory fitness, nutritional status, endothelial function, and autonomic modulation in school-age adolescents: a principal component analysis. Obes Res Clin Pr. 2021;15:205–11.
Godoy-Matos A, Oliveira J, Guedes E, Carraro L, Lopes A, Mancini M, et al. Diretrizes brasileiras de obesidade 2009/2010. 2009.
Craig CL, Marshall AL, Sjostrom M, Bauman AE, Booth ML, Ainsworth BE, et al. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc. 2003;35:1381–95.
Jackson AS, Pollock ML. Practical assessment of body composition. Phys Sportsmed. 1985;13:76–90.
Rodrigues B, Barboza CA, Moura EG, Ministro G, Ferreira-Melo SE, Castano JB, et al. Transcranial direct current stimulation modulates autonomic nervous system and reduces ambulatory blood pressure in hypertensives. Clin Exp Hypertens. 2021;43:320–27.
Force T. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation. 1996;93:1043–65.
Ahmed MW, Kadish AH, Parker MA, Goldberger JJ. Effect of physiologic and pharmacologic adrenergic stimulation on heart rate variability. J Am Coll Cardiol. 1994;24:1082–90.
Diedrich A, Jordan J, Tank J, Shannon JR, Robertson R, Luft FC, et al. The sympathetic nervous system in hypertension: assessment by blood pressure variability and ganglionic blockade. J Hypertens. 2003;21:1677–86.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193:265–75.
Irigoyen MC, De Angelis K, Dos Santos F, Dartora DR, Rodrigues B, Consolim-Colombo FM. Hypertension, blood pressure variability, and target organ lesion. Curr Hypertens Rep. 2016;18:31.
Choi JW, Pai SH, Kim SK, Ito M, Park CS, Cha YN. Increases in nitric oxide concentrations correlate strongly with body fat in obese humans. Clin Chem. 2001;47:1106–09.
Terasaka N, Yu S, Yvan-Charvet L, Wang N, Mzhavia N, Langlois R, et al. ABCG1 and HDL protect against endothelial dysfunction in mice fed a high-cholesterol diet. J Clin Invest. 2008;118:3701–13.
Vincent HK, Taylor AG. Biomarkers and potential mechanisms of obesity-induced oxidant stress in humans. Int J Obes (Lond). 2006;30:400–18.
Li R, Lyn D, Lapu-Bula R, Oduwole A, Igho-Pemu P, Lankford B, et al. Relation of endothelial nitric oxide synthase gene to plasma nitric oxide level, endothelial function, and blood pressure in African Americans. Am J Hypertens. 2004;17:560–67.
Noronha BT, Li J-M, Wheatcroft SB, Shah AM, Kearney MTJD. Inducible nitric oxide synthase has divergent effects on vascular and metabolic function in obesity. Diabetes. 2005;54:1082–89.
Fitzgerald SM, Kemp-Harper BK, Parkington HC, Head GA, Evans RG. Endothelial dysfunction and arterial pressure regulation during early diabetes in mice: roles for nitric oxide and endothelium-derived hyperpolarizing factor. Am J Physiol Regul Integr Comp Physiol. 2007;293:R707–13.
Campese VM, Ye S, Zhong H, Yanamadala V, Ye Z, Chiu J. Reactive oxygen species stimulate central and peripheral sympathetic nervous system activity. Am J Physiol Heart Circ Physiol. 2004;287:H695–703.
Lénárd Z, Studinger P, Mersich B, Pavlik G, Kollai M. Cardiovagal autonomic function in sedentary and trained offspring of hypertensive parents. J Physiol. 2005;565:1031–38.
Pomeranz B, Macaulay RJ, Caudill MA, Kutz I, Adam D, Gordon D, et al. Assessment of autonomic function in humans by heart rate spectral analysis. Am J Physiol. 1985;248:H151–53.
Melanson EL. Resting heart rate variability in men varying in habitual physical activity. Med Sci Sports Exerc. 2000;32:1894–1901.
Wulsin LR, Horn PS, Perry JL, Massaro JM, D'Agostino RB. Autonomic imbalance as a predictor of metabolic risks, cardiovascular disease, diabetes, and mortality. J Clin Endocrinol Metab. 2015;100:2443–48.
Franzoni F, Ghiadoni L, Galetta F, Plantinga Y, Lubrano V, Huang Y, et al. Physical activity, plasma antioxidant capacity, and endothelium-dependent vasodilation in young and older men. Am J Hypertens. 2005;18:510–16.
Gohar EY, Pollock DM. Sex-specific contributions of endothelin to hypertension. Curr Hypertens Rep. 2018;20:58.
Unger T, Borghi C, Charchar F, Khan NA, Poulter NR, Prabhakaran D, et al. International society of hypertension global hypertension practice guidelines. Hypertension. 2020;75:1334–1357.0
Acknowledgements
We would like to acknowledge all of the volunteers for their willingness to participate in the study.
Funding
This study was supported by CNPq (457200/2014-6; 407398/2021-0; 406792/2022-4), CAPES (88881.062178/2014-01) and FAPESP (2019/27782-5). De Angelis, K. and Irigoyen, MC. are recipients of CNPq Fellowship (CNPq-BPQ).
Author information
Authors and Affiliations
Contributions
Authors have contributed to either (i) conception and design of the study (MCN, AV, SDC, MPR, KDA), (ii) acquisition, analysis, or interpretation of data (MCN, AV, DSD, FS, SDC, MCI, MPR, KDA), or (iii) drafting the work or revising it critically for important intellectual content (MCN, AV, DSD, FS, SDC, MCI, MPR, KDA). All authors have qualified for authorship and approved the final version of the manuscript. Finally, all authors agree being accountable with regards to ensuring accuracy and integrity for the work currently investigated.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Ethical approval was granted for this study from the UNINOVE Research Ethics Committee (No 1.672.599). All subjects provided written informed consent to participate. This study was conducted in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nascimento, M.C., Viana, A., Dias, D.d.S. et al. Impact of an active lifestyle on cardiovascular autonomic modulation and oxidative stress in males with overweight and parental history of hypertension. J Hum Hypertens 38, 404–412 (2024). https://doi.org/10.1038/s41371-024-00895-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41371-024-00895-5