Abstract
Introduction
Obesity is associated with alterations in cardiac structure and haemodynamics leading to cardiovascular mortality and morbidity. Culminating evidence suggests improvement of cardiac structure and function following bariatric surgery.
Objective
To evaluate the effect of bariatric surgery on cardiac structure and function in patients before and after bariatric surgery.
Methods
Systematic review and meta-analysis of studies reporting pre- and postoperative cardiac structure and function parameters on cardiac imaging in patients undergoing bariatric surgery.
Results
Eighty studies of 3332 patients were included. Bariatric surgery is associated with a statistically significant improvement in cardiac geometry and function including a decrease of 12.2% (95% CI 0.096–0.149; p < 0.001) in left ventricular (LV) mass index, an increase of 0.155 (95% CI 0.106–0.205; p < 0.001) in E/A ratio, a decrease of 2.012 mm (95% CI 1.356–2.699; p < 0.001) in left atrial diameter, a decrease of 1.16 mm (95% CI 0.62–1.69; p < 0.001) in LV diastolic dimension, and an increase of 1.636% (95% CI 0.706–2.566; p < 0.001) in LV ejection fraction after surgery.
Conclusion
Bariatric surgery led to reverse remodelling and improvement in cardiac geometry and function driven by metabolic and haemodynamic factors.
Similar content being viewed by others
Introduction
Obesity is a global health issue with a plethora of medical and socioeconomic implications that continues to increase in prevalence. It is predicted that one in five women and one in seven men will be living with obesity by 2030 [1]. Obesity leads to the development of cardiovascular risk factors and cardiovascular disease mortality [2]. The adverse effects of obesity on the cardiovascular system can lead to intrinsic cardiac changes including an increase in left ventricular mass (LVM), left ventricular hypertrophy, and ventricular and atrial dilatations. These obesity-related structural changes combined with metabolic syndrome can cause impaired cardiac function as a result of maladaptive cardiac remodelling [3,4,5,6].
Current literature suggests that weight loss may lead to reverse cardiac remodelling and improve cardiac structure and function [7]. Bariatric surgery has been recognised as the most effective and sustainable long-term intervention to achieve weight loss in patients with severe obesity. In addition to the reduction in metabolic syndrome, the beneficial effects of bariatric surgery on cardiac structure and function have been demonstrated in various studies [8,9,10,11,12]. The aim of this systematic review and meta-analysis is to quantify the impact of bariatric surgery on changes to cardiac structure and function by synthesising extensive data available in the literature.
Methods
This systematic review was performed in line with a registered protocol and reported according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [13]. The review was also registered on PROSPERO Centre for Reviews and Dissemination (registration number CRD42023396430).
A literature search was performed in August 2022 using MEDLINE (via PubMed), EMBASE (via OVID) and Cochrane database using MeSH terms in all combinations: ‘bariatric surgery’ or ‘metabolic surgery’ or ‘weight loss’ or ‘obesity surgery’ and ‘echocardiography’ or ‘magnetic resonance imaging’ or ‘cardiac imaging’ or ‘cardiac dimensions’ or ‘ventricular dimensions’ or ‘cardiac function’ or ‘cardiac structure’. Studies identified from the search strategy were entered into Covidence (Victoria, Australia) for duplication removal and bibliographic management. Authors NS, LA and JYC independently identified relevant studies and discrepancies were resolved by consensus between all authors. The exact search strategy is outlined in Supplementary material 1.
Inclusion and exclusion criteria
The inclusion and exclusion criteria were defined before commencement of the literature search. The following criteria were required for inclusion in the study:
-
i.
Randomised controlled trials (RCT), prospective or retrospective cohort studies, case (control) studies, cross-sectional studies.
-
ii.
Patients who had undergone bariatric surgery and had an echocardiogram pre- and postoperatively.
-
iii.
Reported outcomes of interest including cardiac geometry, systolic and diastolic function.
-
iv.
Original, full publications published in the English language.
All studies reporting echocardiographic or magnetic resonance cardiac structural or functional parameters were included. Studies were excluded from analysis if extraction of data was not possible due to inconsistencies, defined as the lack of reported pre- and/or postoperative data.
Data extraction and quality assessment
A standardised data extraction form was developed on Covidence and two authors (NS and JYC) independently extracted all relevant data. All discrepancy was resolved by group discussion. Quality scoring of studies was performed using the Newcastle–Ottawa assessment tool.
Statistical analysis
Data analysis was performed using Stata Software, Version 15.1. StataCorp LCC, TX. Random effects analysis was used to calculate weighted mean difference and mass effect. All studies were included in the analysis if relevant data were available. Data were analysed using a random effects model and statistical heterogeneity was calculated using I2. An I2 of <30 was considered as low, 30–60 as moderate and >60 as high heterogeneity. Results were computed and represented on forest plots (see supplemented section).
Results
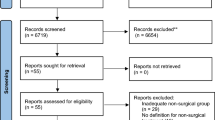
Eighty studies were found to fulfil the inclusion criteria and were included in this systematic review, producing a pooled patient population of 3332 patients. Fifty-four were prospective studies and 26 were retrospective studies. The average quality of studies according to the Newcastle–Ottawa scale was good (6.4). The mean age was 42 years old with a mean starting BMI of 45.9 kg/m2. The mean follow-up period was 12.2 [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58] months. The study characteristics are summarised in Table 1.
Cardiac geometry
Left ventricular mass (LVM)
Absolute LVM was reported in 35 studies, which included 2369 patients. Pooled analysis demonstrated a mean decrease of 31.27 g in LVM after surgery (95% CI 26.53–36.12; p < 0.001). There was a moderate heterogeneity between studies of I2 = 54.7% (Fig. 1).
Left ventricular mass index (LVMI)
LVMI is calculated to standardised measurements of LVM. It is often recommended that height is raised to the power of 2.7 (m2.7) as described by deSimone et al. [59]. This method accounts for close to the equivalent of the effect of lean body mass and excludes the effect of obesity and blood pressure elevation on LVM [60]. Twenty-two studies including 1142 patients reported on LVMI relative to body surface area (m2), analysis showed a decrease after surgery of 10.13% (95% CI 4.270–15.996; p < 0.001); 7 studies including 198 patients, indexed to height (m) showed a decrease of 19.26% (95% CI 1.815–40.336; p < 0.001) and 16 studies including 1021 patients indexed to height (m2.7) showed a decrease of 8.66% (95% CI 6.347–10.977; p < 0.001). Proportional analysis of findings in 45 studies with 2361 patients demonstrated a weighted mean decrease of 12.2% (95% CI 0.096–0.149; p < 0.001) in LVMI after surgery with heterogeneity of I2 = 74.2% (Fig. 2).
Left ventricular end-systolic volume (LVESV)
Sixteen studies including 1024 patients reported on LVESV demonstrating a weighted mean decrease of 7.29 mL (95% CI 2.31–12.27; p = 0.004) after surgery, with a heterogeneity score of I2 = 93.3% (Fig. 3).
Left ventricular end-diastolic volume (LVEDV)
Nineteen studies including 1111 patients reported on LVEDV demonstrating a weighted mean decrease of 14.49 mL (95% CI 6.42–22.56; p < 0.001) after surgery with heterogeneity of I2 = 90.5% between studies (Fig. 4).
Left ventricular global longitudinal strain (LVGLS)
Thirteen studies including 539 patients reported LVGLS demonstrating a weighted mean increase of 3.43% (95% CI 2.41–4.45; p < 0.001), with a heterogeneity of I2 = 91.1% between studies (Fig. 5).
Left ventricular systolic dimension (LVSD)
Twenty-four studies including 993 patients reported on LVSD pooled analysis demonstrated a decrease of 0.51 mm (95% CI 0.08–1.11; p < 0.001) after surgery, with a heterogeneity of I2 = 74.4% between studies.
Left ventricular diastolic dimension (LVDD)
Thirty-eight studies including 2071 patients reported on LVDD with analysis demonstrating a decrease of 1.16 mm (95% CI 0.62–1.69; p < 0.001) after surgery with a heterogeneity between studies of I2 = 74.7% (Fig. 6)
Diastolic function
E/A ratio
The E/A ratio represents the ratio of peak velocity blood flow from left ventricular relaxation in early diastole (the E wave) to peak velocity flow in late diastole (the A wave) caused by atrial contraction [61]. It is therefore a marker of the function of the left ventricle of the heart. Thirty-six studies including 680 patients reported on the E/A ratio before and after surgery, demonstrating an increase of 0.155 (95% CI 0.106–0.205; p < 0.001) with interstudy heterogeneity of I2 = 77.1%.
Left atrial diameter (LA diameter)
Twenty-five studies including 1125 patients reported on LA diameter with pooled analysis demonstrating a decrease of 2.012 mm (95% CI 1.356–2.669; p < 0.001) after surgery with a heterogeneity between studies of I2 = 82.1%.
Septal E/A ratio
Five studies including 209 patients reported on septal E/A ratio with pooled analysis demonstrating a decrease of 0.797 (95% CI 0.234–1.361; p < 0.001) after surgery.
Systolic function
Left ventricular ejection fraction (LVEF)
Forty-three studies including 1955 patients reported on LVEF with pooled analysis demonstrating an increase of 1.636% (95% CI 0.706–2.566 p < 0.001) after surgery, with interstudy heterogeneity of I2 = 84.2% (Fig. 7)
Stroke volume (SV)
Eight studies including 676 patients reported on SV demonstrating a decrease of 2.95 mL (95% CI –8.565 to 2.660; p < 0.001) after surgery with an interstudy heterogeneity of I2 = 72.6.
Cardiac output (CO)
Three studies including 151 patients reported on cardiac output demonstrating a decrease of 0.625 mL/min (95% CI –1.261 to 0.010; p = 0.089) after surgery with heterogeneity between studies of I2 = 58.7%.
Blood pressure
Resting systolic blood pressure was reported in 24 studies including 1066 patients. Pooled analysis demonstrated a decrease of 10.918 mmHg (95% CI 8.457–13.379; p < 0.001) after surgery with heterogeneity of I2 = 77.8% between studies. Resting diastolic blood pressure was reported in 23 studies including 1031 patients. Pooled analysis demonstrated a decrease of 5.921 mmHg (95% CI 4.646–7.795; p < 0.001) after surgery with heterogeneity of I2 = 75.8% between studies.
Heart rate
Resting heart rate was reported in 12 studies including 554 patients. Pooled analysis demonstrated a weighted mean decrease of 6.68 beats/min (95% CI 3.76–9.59; p < 0.001) after surgery with heterogeneity between studies of I2 = 86.1%.
Discussion
This study demonstrates an improvement in cardiac structures and function in patients after undergoing bariatric surgery. These statistically significant improvements are reported in cardiac geometry, systolic and diastolic function across cardiac imaging modalities. For example, this was reflected by bariatric surgery leading to an increase in LVEF and E/A ratio, and a decrease in LA diameter, LVDD and LVMI. This systematic review and meta-analysis including eighty studies represents findings confirming and extending previous evidence in improvements in cardiac geometry and function, demonstrating statistically significant changes across all parameters.
Cardiac geometry
Obesity has long been recognised as an independent risk factor to directly alter cardiac structure and function, including ventricular and septal hypertrophy leading to increased LVM and MACE (major adverse cardiovascular event) [5, 14]. Our results demonstrate a statistically significant weighted mean decrease of 12.2% in LVMI after bariatric surgery. There was also a mean weighted decrease of 31.27 g in LV mass as a measure of improvement of left ventricular hypertrophy (LVH). This supports the notion that bariatric surgery leads to LV reverse remodelling and plasticity, resulting in a long-term decrease in cardiovascular mortality in bariatric patients [3, 15].
The metabolic effects of bariatric surgery is thought to improve the haemodynamic parameters and lead to reverse remodelling by decreasing the inflammatory state that induces ventricular hypertrophy associated with pathological diastolic dysfunction and ventricular filling, reduced ventricular contractility, reduced coronary reserve, chronic overload and arrhythmogenic electrical dysfunction [16, 17].
The concept of beneficial reverse remodelling of LVH following bariatric surgery is further supported by our results, demonstrating an improvement in posterior wall thickness, interventricular septal thickness and relative wall thickness. In addition, our study demonstrated improvement in end-systolic and end-diastolic volume and diameter dimensions supporting increased filling and relaxation of the left ventricles after bariatric surgery.
Diastolic function
Our study demonstrates that bariatric surgery can lead to improvement in echocardiographic markers of diastolic dysfunction through reverse remodelling of obesity-related changes in cardiac structure and function influenced by neurohormonal and metabolic factors. Diastolic dysfunction with impaired myocardial relaxation is characterised by a decreased early (E wave) but enhanced atrial LV filling (A wave). Thus, atrial contraction basically compensates for the decrease in early passive filling and results in lower E/A ratio. Our study shows a statistically significant increase in E/A ratio post bariatric surgery; significant decrease in LA diameter, which is an important measure of diastolic function as LA enlargement reflects chronic exposure to LV filling pressure. In addition, LVMI is also a predictor of LA enlargement. An increased LA diameter is associated with an increased risk of developing atrial fibrillation so an improvement in these parameters after bariatric surgery reflects a positive impact on diastolic function and arrhythmogenesis. LA diameter is also known to be associated with obstructive sleep apnoea, which has also been demonstrated to be reported to improve in patients after undergoing bariatric surgery [18, 19].
Systolic function
The development of LV systolic dysfunction has long been recognised as a complication from severe obesity. Our meta-analysis demonstrated a significant increase in LVEF after bariatric surgery. While bariatric surgery has been shown to improve diastolic dysfunction, the effect on LV systolic function and mechanics has been little explored. Previous studies demonstrated that improvement in systolic function only occurs in obese patients with systolic dysfunction preoperatively and those individuals who have been severely obese for a longer period of time [20, 21]. More recently, rapid weight loss from bariatric surgery has been shown to be associated with a considerable increase in LVEF and significant improvement in systolic function, especially in patients with pre-existing heart failure [22] and even in those individuals indicated for a cardiac transplant [23].
Our systematic review and meta-analysis has also demonstrated an improvement in LVGLS post bariatric surgery, which is used as a marker of left systolic mechanics. Postoperative increase in GLS is associated with postoperative reduction in afterload, mean blood pressure and BMI [24]. Improvement in parameters of LV geometry and mechanics in our study demonstrated an improvement in systolic dysfunction in patients after bariatric surgery. These profound improvements in cardiac imaging after bariatric surgery can be related to the mechanisms in how these procedures achieve beneficial reverse remodelling.
Adipose tissue has been considered as an endocrine organ with the ability to produce a variety of hormones and cytokines [25, 26]. Therefore, the combined effects of weight loss and metabolic enhancement from bariatric surgery can result in reverse remodelling of cardiac geometry and function. Obesity is also linked with sympathetic nervous system overactivity [27] and baroreflex sensitivity [28], and this cardiac autonomic nervous system imbalance results in increased heart rate and hypertension. Our meta-analysis has demonstrated a significant reduction in heart rate and blood pressure.
The metabolic and haemodynamic hypothesis as described by le Roux et al. [29] may explain the beneficial surgical effects in morbid obesity; metabolites such as leptin and other adipokines are reported to induce ventricular hypertrophy in obesity and lead to increasing circulating blood volume contributing to ventricular dilatation and hypertrophy [30]. Bariatric surgery leads to reversal of metabolic dysfunction which in turn can contribute to improved cardiac structure and function.
Bariatric surgery causes several metabolic improvements described as BRAVE effect: bile flow alterations, reduction of gastric size, anatomical gut rearrangement and altered flow of nutrients, vagal manipulation, enteric gut modulation [31], which contributes to cardiac reverse remodelling. These effects occur almost simultaneously after surgery and may offer a paradigm to identify the profound mechanisms driving improvements in glucose metabolism, insulin resistance, gut hormone release, microbiota and adipokine modulation [32,33,34,35]; which may offer a resolution of obesity-linked cardiac dysfunction.
The impact of bariatric surgery on enteric gut hormones has been reported to have a beneficial effect on cardiac function via the entero-cardiac axis [36, 37]. By inducing changes in secretion of hormones such as secretin produced in the duodenum, glucagon produced in the pancreas and vasoactive intestinal peptide produced in pancreas, gut and brain, act as inotropes indirectly involved in cardiac cell communication by activating cardiac membrane adenylate cyclase. The exact mechanism is currently unclear, although it is thought that cardiac energy metabolism is enhanced through tricarboxylic acid (TCA) cycle intermediaries, cardiorenal protective activities and biochemical caloric restrictions [29, 38].
Anorexigenic gut hormones, mainly GLP-1 (glucagon-like peptide-1) and peptide YY associated with improvement in insulin secretion and sensitivity, have been found to favourably modulate cardiac function by reducing vascular tone and increase in myocardial contractility. More recently, cardiovascular outcome trials (CVOTs) of GLP-1 receptor agonists have demonstrated potential cardiovascular protective potential by retarding lipotoxicity and cardiac inflammation via pyroptotic cytokines but also improving cardiac energy metabolism [29, 37,38,39,40].
The orexigenic hormone ghrelin has been suggested to improve SV index, ejection fraction and cardiac index with a decrease in LV wall stress. Adipokines such as leptin, TNFα and adiponectin have demonstrated cardiovascular activity with circulating leptin levels correlating with LV mass in morbid obesity and may also be responsible in contributing to the beneficial cardiac effects of bariatric surgery [37, 41,42,43]. The renin-angiotensin-aldosterone system (RAAS) can counter haemodynamic stress. However, sustained activation of RAAS and angiotensin release worsens cardiac fibrosis, cellular necrosis and induces cardiomyocyte hypertrophy, inhibition of RAAS is linked with reverse cardiac remodelling. Bariatric surgery has been linked to changes in several RAAS components, including a significant decrease in plasma renin activity, aldosterone and angiotensin-converting enzyme-2 (ACE2) activity and increase in ACE/ACE2 ratio [44,45,46].
Beta-adrenergic receptor blockade has demonstrated reverse LV remodelling and improvement in EF linked to SERCA2a, which has been linked to reversing cardiac excitation-contraction-coupling abnormalities and associated chamber dilatation and dysfunction [14, 22, 47, 48, 62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111]. Osteopontin (OPN) has emerged as a key ubiquitous phosphoprotein involved in numerous biological functions, including biomineralisation, extracellular matrix and tissue remodelling and repair. Enhanced expression of OPN appears to play an important role in remodelling of the heart following myocardial infarction as well as LVH. Bariatric surgery has been shown to increase OPN plasma concentration [112].
Strengths and limitations
The strength of this study includes comprehensive data collection involving extensive data published in the literature and rigorous data extraction and statistical analysis. There are important limitations which must be addressed including the vulnerability of echocardiographic studies to subjectivity and reporting biases on interpretation by healthcare professionals undertaking and/or reporting cardiac imaging. In addition, the majority of the studies included were prospective studies, rather than RCTs, with a limited follow-up period.
Conclusions
This meta-analysis and systematic review on the effect of bariatric surgery on cardiac structure and function assessed on cardiac imaging suggests that bariatric surgery is associated with significant improvement in cardiac structure and function. Bariatric surgery can drive reverse cardiac remodelling improving cardiac dysfunction. Further RCTs are justified to describe the relationship between the improvement of cardiac structure and function as assessed by imaging modalities to mortality and morbidity when addressing obesity-associated cardiac disease.
References
World Obesity Atlas 2022. Obesity is a disease. https://s3-eu-west-1.amazonaws.com/wof-files/World_Obesity_Atlas_2022.pdf.
Powell-Wiley TM, Poirier P, Burke LE, Després JP, Gordon-Larsen P, Lavie CJ, et al. American Heart Association Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology; Council on Epidemiology and Prevention; and Stroke Council. Obesity and Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation. 2021;143:e984–1010.
Ashrafian H, le Roux CW, Darzi A, Athanasiou T. Effects of bariatric surgery on cardiovascular function. Circulation. 2008;118:2091–102.
Meigs JB, Wilson PW, Fox CS, Vasan RS, Nathan DM, Sullivan LM, et al. Body mass index, metabolic syndrome, and risk of type 2 diabetes or cardiovascular disease. J Clin Endocrinol Metab. 2006;91:2906–12.
Lauer MS, Anderson KM, Kannel WB, Levy D. The impact of obesity on left ventricular mass and geometry. The Framingham Heart Study. JAMA. 1991;266:231–6.
Kardassis D, Bech-Hanssen O, Schönander M, Sjöström L, Karason K. The influence of body composition, fat distribution, and sustained weight loss on left ventricular mass and geometry in obesity. Obesity. 2012;20:605611.
Tuah NA, Amiel C, Qureshi S, Car J, Kaur B, Majeed A. Transtheoretical model for dietary and physical exercise modification in weight loss management for overweight and obese adults. Cochrane Database Syst Rev. 2011;10:CD008066.
Gutierrez-Blanco D, Romero Funes D, Castillo M, Lo Menzo E, Szomstein S, Rosenthal RJ. Bariatric surgery reduces the risk of developing type 2 diabetes in severe obese subjects undergoing sleeve gastrectomy. Surg Obes Relat Dis. 2019;15:168–72.
Lakhani M, Fein S. Effects of obesity and subsequent weight reduction on left ventricular function. Cardiol Rev. 2011;19:14.
Cuspidi C, Rescaldani M, Tadic M, Sala C, Grassi G. Effects of bariatric surgery on cardiac structure and function: a systematic review and meta-analysis. Am J Hypertens. 2014;27:146–56.
Zhou N, Scoubeau C, Forton K, Loi P, Closset J, Deboeck G. Lean mass loss and altered muscular aerobic capacity after bariatric surgery. Obes Facts. 2022;15:248–56.
Aggarwal R, Harling L, Efthimiou E, Darzi A, Athanasiou T, Ashrafian H. The effects of bariatric surgery on cardiac structure and function: a systematic review of cardiac imaging outcomes. Obes Surg. 2016;26:1030–40.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
Hsuan CF, Huang CK, Lin JW, Lin LC, Lee TL, Tai CM, et al. The effect of surgical weight reduction on left ventricular structure and function in severe obesity. Obesity (Silver Spring). 2010;18:1188–93.
Gomez CO, Rammohan R, Romero-Funes D, Sarmiento-Cobos M, Gutierrez D, Menzo EL, et al. Bariatric surgery improves heart geometry and plasticity. Obes Surg. 2022;32:1–6.
Lascaris B, Pouwels S, Houthuizen P, Dekker LR, Nienhuijs SW, Bouwman RA, et al. Cardiac structure and function before and after bariatric surgery: a clinical overview. Clin Obes. 2018;8:434–43.
Paulus WJ, Tschöpe C, Sanderson JE, Rusconi C, Flachskampf FA, Rademakers FE, et al. How to diagnose diastolic heart failure: a consensus statement on the diagnosis of heart failure with normal left ventricular ejection fraction by the Heart Failure and Echocardiography Associations of the European Society of Cardiology. Eur Heart J. 2007;28:2539–50.
Ashrafian H, Toma T, Rowland SP, Harling L, Tan A, Efthimiou E, et al. Bariatric surgery or non-surgical weight loss for obstructive sleep apnoea? A systematic review and comparison of meta-analyses. Obes Surg. 2015;25:1239–50.
Donnellan E, Wazni OM, Elshazly M, Kanj M, Hussein AA, Baranowski B, et al. Impact of bariatric surgery on atrial fibrillation type. Circ Arrhythm Electrophysiol. 2020;13:e007626.
Alpert MA, Terry BE, Lambert CR, Kelly DL, Panayiotou H, Mukerji V, et al. Factors influencing left ventricular systolic function in nonhypertensive morbidly obese patients, and effect of weight loss induced by gastroplasty. Am J Cardiol. 1993;71:733–77.
Alpert MA, Lambert CR, Panayiotou H, Terry BE, Cohen MV, Massey CV, et al. Relation of duration of morbid obesity to left ventricular mass, systolic function, and diastolic filling, and effect of weight loss. Am J Cardiol. 1995;76:1194–7.
Sarmiento-Cobos M, Fonnegra CB, Montorfano L, Rivera C, Cogollo VJ, Lo Menzo E, et al. Short-term rapid weight loss induced by bariatric surgery improves ventricular ejection fraction in patients with severe obesity and heart failure. Surg Obes Relat Dis. 2021;17:1616–20.
Vest AR, Patel P, Schauer PR, Satava ME, Cavalcante JL, Brethauer S, et al. Clinical and echocardiographic outcomes after bariatric surgery in obese patients with left ventricular systolic dysfunction. Circ Heart Fail. 2016;9:e002260.
Grymyr LMD, Nadirpour S, Gerdts E, Nedrebø BG, Just Hjertaas JJ, Matre K. One-year impact of bariatric surgery on left ventricular mechanics: results from the prospective FatWest study. Eur Heart J Open. 2021;1:oeab024.
Morricone L, Malavazos AE, Coman C, Donati C, Hassan T, Vaciezel F. Echocardiographic abnormalities in normotensive obese patients: relationship with visceral fat. Obes Res. 2002;10:489–98.
Malavazos AE, Corsi MM, Ermetici F, Coman C, Sardanelli F, Rossi A, et al. Proinflammatory cytokines and cardiac abnormalities in uncomplicated obesity: relationship with abdominal fat deposition. Nutr Metab Cardiovasc Dis. 2007;17:294–302.
Quilliot D, Böhme P, Zannad F, Ziegler O. Sympathetic-leptin relationship in obesity: effect of weight loss. Metabolism. 2008;57:555–62.
Skrapari I, Tentolouris N, Katsilambros N. Baroreflex function: determinants in healthy subjects and disturbances in diabetes, obesity and metabolic syndrome. Curr Diabetes Rev. 2006;2:329–38.
Ashrafian H, Athanasiou T, le Roux CW. Heart remodelling and obesity: the complexities and variation of cardiac geometry. Heart. 2011;97:171–2.
Rider OJ, Petersen SE, Francis JM, Ali MK, Hudsmith LE, Robinson MR, et al. Ventricular hypertrophy and cavity dilatation in relation to body mass index in women with uncomplicated obesity. Heart. 2011;97:203–8.
Wabitsch M. Gastrointestinal endocrinology in bariatric surgery. Endocr Dev. 2017;32:124–38.
Ashrafian H, Bueter M, Ahmed K, Suliman A, Bloom SR, Darzi A, et al. Metabolic surgery: an evolution through bariatric animal models. Obes Rev. 2010;11:907–20.
Ashrafian H, Athanasiou T, Li JV, Bueter M, Ahmed K, Nagpal K, et al. Diabetes resolution and hyperinsulinaemia after metabolic Roux-en-Y gastric bypass. Obes Rev. 2011;12:e257–72.
Ashrafian H, Darzi A, Athanasiou T. Autobionics: a new paradigm in regenerative medicine and surgery. Regen Med. 2010;5:279–88.
Li JV, Ashrafian H, Bueter M, Kinross J, Sands C, le Roux CW, et al. Metabolic surgery profoundly influences gut microbial-host metabolic cross-talk. Gut. 2011;60:1214–23.
Vaz M, Pereira SS, Monteiro MP. Metabolomic signatures after bariatric surgery – a systematic review. Rev Endocr Metab Disord. 2022;23:503–19.
Ashrafian H, le Roux CW. Metabolic surgery and gut hormones – a review of bariatric entero-humoral modulation. Physiol Behav. 2009;97:620–31.
Ashrafian H, Li JV, Spagou K, Harling L, Masson P, Darzi A. et al. Bariatric surgery modulates circulating and cardiac metabolites. J Proteome Res. 2014;13:570–80.
Dimitriadis GK, Randeva MS, Miras AD. Potential hormone mechanisms of bariatric surgery. Curr Obes Rep. 2017;6:253–65.
Ma X, Liu Z, Ilyas I, Little PJ, Kamato D, Sahebka A, et al. GLP-1 receptor agonists (GLP-1RAs): cardiovascular actions and therapeutic potential. Int J Biol Sci. 2021;17:2050–68.
Nagaya N, Moriya J, Yasumura Y, Uematsu M, Ono F, Shimizu W, et al. Effects of ghrelin administration on left ventricular function, exercise capacity, and muscle wasting in patients with chronic heart failure. Circulation. 2004;110:3674–9.
Perego L, Pizzocri P, Corradi D, Maisano F, Paganelli M, Fiorina P, et al. Circulating leptin correlates with left ventricular mass in morbid (Grade III) obesity before and after weight loss induced by bariatric surgery: a potential role for leptin in mediating human left ventricular hypertrophy. J Clin Endocrinol Metab. 2005;90:4087–93.
Rodríguez A. Novel molecular aspects of ghrelin and leptin in the control of adipobiology and the cardiovascular system. Obes Facts. 2014;7:82–95.
Oliveras A, Molina L, Goday A, Sans L, Riera M, Vazquez S, et al. Effect of bariatric surgery on cardiac structure and function in obese patients: role of the renin-angiotensin system. J Clin Hypertens (Greenwich). 2021;23:181–92.
Koitabashi N, Kass DA. Reverse remodeling in heart failure–mechanisms and therapeutic opportunities. Nat Rev Cardiol. 2011;9:147–57.
Hayes H, Patz J, Corbett J, Afzal MZ, Strande J, Kindel TL. Sleeve gastrectomy in obese Wistar rats improves diastolic function and promotes cardiac recovery independent of weight loss. Surg Obes Relat Dis. 2019;15:837–42.
Koshino Y, Villarraga HR, Somers VK, Miranda WR, Garza CA, Hsiao JF, et al. Changes in myocardial mechanics in patients with obesity following major weight loss after bariatric surgery. Obesity (Silver Spring). 2013;21:1111–8.
Kaier TE, Morgan D, Grapsa J, Demir OM, Paschou SA, Sundar S, et al. Ventricular remodelling post-bariatric surgery: is the type of surgery relevant? A prospective study with 3D speckle tracking. Eur Heart J Cardiovasc Imaging. 2014;15:1256–62.
Hamo C, Wallace A, Shah A, Florido R, Tcheugui J, Matsushita K. et al. Correlation between echocardiographic measures and HS-CTNT levels pre- and post-bariatric surgery. J Am Coll Cardiol. 2020;75(11_Supplement_1):1988.
Frea S, Andreis A, Scarlatta V, Rovera C, Vairo A, Pistone E, et al. Subclinical left ventricular dysfunction in severe obesity and reverse cardiac remodeling after bariatric surgery. J Cardiovasc Echogr. 2020;30:22–8.
Büber İ, Aykota MR, Sevgican Cİ, Adalı MK. The effects of laparoscopic sleeve gastrectomy on cardiac diastolic function, aortic elasticity, and atrial electromechanics delay. Obes Surg. 2021;31:3571–8.
Meng W, Peng R, Du L, Zheng Y, Liu D, Qu S, et al. Weight loss after laparoscopic sleeve gastrectomy ameliorates the cardiac remodeling in obese Chinese. Front Endocrinol (Lausanne). 2022;12:799537.
El-Zawawy HT, El-Aghoury AA, Katri KM, El-Sharkawy EM, Gad SMS. Cortisol/DHEA ratio in morbidly obese patients before and after bariatric surgery: relation to metabolic parameters and cardiovascular performance. Int J Obes (Lond). 2022;46:381–92.
Baradaran A, Zamanifard S, Parham M, Nikdoust F. Evaluation of global longitudinal strain pattern (GLS) and echocardiographic parameters in the sleeve gastrectomy patients before and after surgery. J Adv Med Biomed Res. 2022;30:327–32.
Kokkinos A, Alexiadou K, Liaskos C, Argyrakopoulou G, Balla I, Tentolouris N. et al. Improvement in cardiovascular indices after Roux-en-Y gastric bypass or sleeve gastrectomy for morbid obesity. Obes Surg. 2013;23:31–8.
Dzenkeviciūte V, Petrulioniene Z, Sapoka V, Aidietiene S, Abaraviciute L. The effect of weight loss on the cardiac structure and function after laparoscopic adjustable gastric banding surgery in morbidly obese individuals. Obes Surg. 2014;24:1961–8.
Koc F, Kayaoglu HA, Celik A, Altunkas F, Karayakali M, Ozbek K, et al. Effect of weight loss induced by intragastric balloon therapy on cardiac function in morbidly obese individuals: a pilot study. Med Princ Pract. 2015;24:432–5.
Leung M, Xie M, Durmush E, Leung DY, Wong VW. Weight loss with sleeve gastrectomy in obese type 2 diabetes mellitus: impact on cardiac function. Obes Surg. 2016;26:321–6.
de Simone G, Daniels SR, Devereux RB, Meyer RA, Roman MJ, de Divitiis O, et al. Left ventricular mass and body size in normotensive children and adults: assessment of allometric relations and impact of overweight. J Am Coll Cardiol. 1992;20:1251–60.
National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents. The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics. 2004;114(2 Suppl 4th Report):555–76.
Nishimura RA, Tajik AJ. Evaluation of diastolic filling of left ventricle in health and disease: Doppler echocardiography is the clinician’s Rosetta Stone. J Am Coll Cardiol. 1997;30:8–18.
Kemaloğlu Öz, Ünal Dayı T, Seyit H, Öz A, Ösken A, Atasoy I, et al. The effects of weight loss after sleeve gastrectomy on left ventricular systolic function in men versus women. J Clin Ultrasound. 2016;44:492–9.
Brownell NK, Rodriguez-Flores M, Garcia-Garcia E, Ordoñez-Ortega S, Oseguera-Moguel J, Aguilar-Salinas CA. Impact of body mass index >50 on cardiac structural and functional characteristics and surgical outcomes after bariatric surgery. Obes Surg. 2016;26:2772–8.
Malekpour Alamdari N, Bayat F, Bakhtiyari M, Noori M. Effects of weight loss on echocardiographic parameters 1 year after sleeve gastrectomy. J Tehran Heart Cent. 2019;14:128–33.
Tuluce K, Kara C, Tuluce SY, Cetin N, Topaloglu C, Bozkaya YT, et al. Early reverse cardiac remodeling effect of laparoscopic sleeve gastrectomy. Obes Surg. 2017;27:364–75.
Shin SH, Lee YJ, Heo YS, Park SD, Kwon SW, Woo SI, et al. Beneficial effects of bariatric surgery on cardiac structure and function in obesity. Obes Surg. 2017;27:620–5.
Kurnicka K, Domienik-Karłowicz J, Lichodziejewska B, Bielecki M, Kozłowska M, Goliszek S, et al. Improvement of left ventricular diastolic function and left heart morphology in young women with morbid obesity six months after bariatric surgery. Cardiol J. 2018;25:97–105.
Mostfa SA. Impact of obesity and surgical weight reduction on cardiac remodeling. Indian Heart J. 2018;70(Suppl 3):S224–8.
Inci S, Gül M, Alsancak Y, Ozkan N. Short- and mid-term effects of sleeve gastrectomy on left ventricular function with two-dimensional speckle tracking echocardiography in obese patients. Echocardiography. 2019;36:2019–25.
Santos ECL, Del Castillo JM, Parente GBO, Pedrosa RP, Gadelha PS, Lopes RD, et al. Changes in left ventricular mechanics after sleeve gastrectomy. Obes Surg. 2020;30:580–6.
de Witte D, Wijngaarden LH, van Houten VAA, van den Dorpel MA, Bruning TA, van der Harst E, et al. Improvement of cardiac function after Roux-en-Y gastric bypass in morbidly obese patients without cardiac history measured by cardiac MRI. Obes Surg. 2020;30:2475–81.
Kaya BC, Elkan H. Impact of weight loss with laparoscopic sleeve gastrectomy on left ventricular diastolic function: a prospective study. Obes Surg. 2020;30:3280–6.
Sarmiento-Cobos M, Valera R, Botero Fonnegra C, Alonso M, Rivera C, Montorfano L, et al. Ventricular conduction improvement after pericardial fat reduction triggered by rapid weight loss in subjects with obesity undergoing bariatric surgery. Surg Obes Relat Dis. 2022;18:288–94.
Alqunai MS, Assakran BS, Widyan AM, Suresh A, Alharbi AA, Almosallam OI, et al. Effect of bariatric surgery on cardiac function in obese patients: an echocardiographic assessment. Saudi Med J. 2022;43:587–91.
Mukerji R, Petruc M, Fresen JL, Terry BE, Govindarajan G, Alpert MA. Effect of weight loss after bariatric surgery on left ventricular mass and ventricular repolarization in normotensive morbidly obese patients. Am J Cardiol. 2012;110:415–9.
Alpert MA, Nusair MB, Mukerji R, Omran J, Mehra A, Ardhanari S, et al. Effect of weight loss on ventricular repolarization in normotensive severely obese patients with and without heart failure. Am J Med Sci. 2015;349:17–23.
van Schinkel LD, Sleddering MA, Lips MA, Jonker JT, de Roos A, Lamb HJ, et al. Effects of bariatric surgery on pericardial ectopic fat depositions and cardiovascular function. Clin Endocrinol (Oxf). 2014;81:689–95.
Graziani F, Leone AM, Cialdella P, Basile E, Pennestrì F, Della Bona R, et al. Effects of bariatric surgery on cardiac remodeling: clinical and pathophysiologic implications. Int J Cardiol. 2013;168:4277–9.
Iancu ME, Copăescu C, Şerban M, Ginghină C. Favorable changes in arterial elasticity, left ventricular mass, and diastolic function after significant weight loss following laparoscopic sleeve gastrectomy in obese individuals. Obes Surg. 2014;24:364–70.
Martin J, Bergeron S, Pibarot P, Bastien M, Biertho L, Lescelleur O, et al. Impact of bariatric surgery on N-terminal fragment of the prohormone brain natriuretic peptide and left ventricular diastolic function. Can J Cardiol. 2013;29:969–75.
Kokkinos A, Alexiadou K, Liaskos C, Argyrakopoulou G, Balla I, Tentolouris N, et al. Improvement in cardiovascular indices after Roux-en-Y gastric bypass or sleeve gastrectomy for morbid obesity. Obes Surg. 2013;23:31–8.
Damiano S, De Marco M, Del Genio F, Contaldo F, Gerdts E, de Simone G, et al. Effect of bariatric surgery on left ventricular geometry and function in severe obesity. Obes Res Clin Pract. 2012;6:e175–262.
Luaces M, Martínez-Martínez E, Medina M, Miana M, González N, Fernández-Pérez C. et al. The impact of bariatric surgery on renal and cardiac functions in morbidly obese patients. Nephrol Dial Transplant. 2012;27(Suppl 4):iv53–7.
Cavarretta E, Casella G, Calì B, Dammaro C, Biondi-Zoccai G, Iossa A, et al. Cardiac remodeling in obese patients after laparoscopic sleeve gastrectomy. World J Surg. 2013;37:565–72.
Luaces M, Cachofeiro V, García-Muñoz-Najar A, Medina M, González N, Cancer E, et al. Anatomical and functional alterations of the heart in morbid obesity. changes after bariatric surgery. Rev Esp Cardiol (Engl Ed). 2012;65:14–21.
Michalsky MP, Raman SV, Teich S, Schuster DP, Bauer JA. Cardiovascular recovery following bariatric surgery in extremely obese adolescents: preliminary results using Cardiac Magnetic Resonance (CMR) Imaging. J Pediatr Surg. 2013;48:170–7.
Kardassis D, Bech-Hanssen O, Schönander M, Sjöström L, Petzold M, Karason K. Impact of body composition, fat distribution and sustained weight loss on cardiac function in obesity. Int J Cardiol. 2012;159:128–33.
McCloskey CA, Ramani GV, Mathier MA, Schauer PR, Eid GM, Mattar SG, et al. Bariatric surgery improves cardiac function in morbidly obese patients with severe cardiomyopathy. Surg Obes Relat Dis. 2007;3:503–7.
Lin CH, Kurup S, Herrero P, Schechtman KB, Eagon JC, Klein S, et al. Myocardial oxygen consumption change predicts left ventricular relaxation improvement in obese humans after weight loss. Obesity (Silver Spring). 2011;19:1804–12.
Valezi AC, Machado VH. Morphofunctional evaluation of the heart of obese patients before and after bariatric surgery. Obes Surg. 2011;21:1693–7.
Owan T, Avelar E, Morley K, Jiji R, Hall N, Krezowski J, et al. Favorable changes in cardiac geometry and function following gastric bypass surgery: 2-year follow-up in the Utah obesity study. J Am Coll Cardiol. 2011;57:732–9.
Algahim MF, Lux TR, Leichman JG, Boyer AF, Miller CC III, Laing ST, et al. Progressive regression of left ventricular hypertrophy two years after bariatric surgery. Am J Med. 2010;123:549–55. https://doi.org/10.1016/j.amjmed.2009.11.020. Erratum in: Am J Med. 2010;123(10):e13.
Syed M, Torosoff M, Rosati C, Alger S, Fein S. Effect of comorbidities and medications on left ventricular mass regression after bariatric surgery. J Clin Hypertens (Greenwich). 2010;12:223–7.
Garza CA, Pellikka PA, Somers VK, Sarr MG, Collazo-Clavell ML, Korenfeld Y, et al. Structural and functional changes in left and right ventricles after major weight loss following bariatric surgery for morbid obesity. Am J Cardiol. 2010;105:550–6.
Jhaveri RR, Pond KK, Hauser TH, Kissinger KV, Goepfert L, Schneider B, et al. Cardiac remodeling after substantial weight loss: a prospective cardiac magnetic resonance study after bariatric surgery. Surg Obes Relat Dis. 2009;5:648–52.
Leichman JG, Wilson EB, Scarborough T, Aguilar D, Miller CC III, Yu S, et al. Dramatic reversal of derangements in muscle metabolism and left ventricular function after bariatric surgery. Am J Med. 2008;121:966–73.
Ippisch HM, Inge TH, Daniels SR, Wang B, Khoury PR, Witt SA. Reversibility of cardiac abnormalities in morbidly obese adolescents. J Am Coll Cardiol. 2008;51:1342–8.
Di Bello V, Santini F, Di Cori A, Pucci A, Talini E, Palagi C. Effects of bariatric surgery on early myocardial alterations in adult severely obese subjects. Cardiology. 2008;109:241–8.
Nault I, Nadreau E, Paquet C, Brassard P, Marceau P, Marceau S, et al. Impact of bariatric surgery–induced weight loss on heart rate variability. Metabolism. 2007;56:1425–30.
Maniscalco M, Arciello A, Zedda A, Faraone S, Verde R, Giardiello C, et al. Right ventricular performance in severe obesity. Effect of weight loss. Eur J Clin Invest. 2007;37:270–5.
Cunha Lde C, da Cunha CL, de Souza AM, Chiminacio Neto N, Pereira RS, Suplicy HL. Evolutive echocardiographic study of the structural and functional heart alterations in obese individuals after bariatric surgery. Arq Bras Cardiol. 2006;87:615–22.
Ikonomidis I, Mazarakis A, Papadopoulos C, Patsouras N, Kalfarentzos F, Lekakis J, et al. Weight loss after bariatric surgery improves aortic elastic properties and left ventricular function in individuals with morbid obesity: a 3-year follow-up study. J Hypertens. 2007;25:439–47.
Leichman JG, Aguilar D, King TM, Mehta S, Majka C, Scarborough T, et al. Improvements in systemic metabolism, anthropometrics, and left ventricular geometry 3 months after bariatric surgery. Surg Obes Relat Dis. 2006;2:592–9.
Willens HJ, Chakko SC, Byers P, Chirinos JA, Labrador E, Castrillon JC, et al. Effects of weight loss after gastric bypass on right and left ventricular function assessed by tissue Doppler imaging. Am J Cardiol. 2005;95:1521–4.
Kanoupakis E, Michaloudis D, Fraidakis O, Parthenakis F, Vardas P, Melissas J. Left ventricular function and cardiopulmonary performance following surgical treatment of morbid obesity. Obes Surg. 2001;11:552–8.
Karason K, Wallentin I, Larsson B, Sjöström L. Effects of obesity and weight loss on cardiac function and valvular performance. Obes Res. 1998;6:422–9.
Gahtan V, Goode SE, Kurto HZ, Schocken DD, Powers P, Rosemurgy AS. Body composition and source of weight loss after bariatric surgery. Obes Surg. 1997;7:184–8.
Alpert MA, Terry BE, Mulekar M, Cohen MV, Massey CV, Fan TM, et al. Cardiac morphology and left ventricular function in normotensive morbidly obese patients with and without congestive heart failure, and effect of weight loss. Am J Cardiol. 1997;80:736–40.
Alpert MA, Lambert CR, Terry BE, Cohen MV, Mulekar M, Massey CV, et al. Effect of weight loss on left ventricular diastolic filling in morbid obesity. Am J Cardiol. 1995;76:1198–201.
Alpert MA, Lambert CR, Terry BE, Kelly DL, Panayiotou H, Mukerji V, et al. Effect of weight loss on left ventricular mass in nonhypertensive morbidly obese patients. Am J Cardiol. 1994;73:918–21.
Alaud-din A, Meterissian S, Lisbona R, MacLean LD, Forse RA. Assessment of cardiac function in patients who were morbidly obese. Surgery. 1990;108:809–18.
Schaller G, Aso Y, Schernthaner GH, Kopp HP, Inukai T, Kriwanek S, et al. Increase of osteopontin plasma concentrations after bariatric surgery independent from inflammation and insulin resistance. Obes Surg. 2009;19:351–6.
Author information
Authors and Affiliations
Contributions
NS, JYC, MF and HA conceptualised the study. NS, JYC, RA and MGF contributed to data collection. NS, MF and HA conducted the analyses. All authors contributed to the interpretation of the analyses and the presentation of the results. NS was responsible for drafting the manuscript. All authors contributed to reviewing and editing the manuscript, and all authors have agreed to the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sargsyan, N., Chen, J.Y., Aggarwal, R. et al. The effects of bariatric surgery on cardiac function: a systematic review and meta-analysis. Int J Obes 48, 166–176 (2024). https://doi.org/10.1038/s41366-023-01412-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-023-01412-3