Abstract
Background
International guidelines recommend children aged 9 months to 2 years consume whole (3.25%) fat cow’s milk, and children older than age 2 years consume reduced (0.1–2%) fat cow’s milk to prevent obesity. The objective of this study was to evaluate the longitudinal relationship between cow’s milk fat (0.1–3.25%) intake and body mass index z-score (zBMI) in childhood. We hypothesized that higher cow’s milk fat intake was associated with lower zBMI.
Methods
A prospective cohort study of children aged 9 months to 8 years was conducted through the TARGet Kids! primary care research network. The exposure was cow’s milk fat consumption (skim (0.1%), 1%, 2%, whole (3.25%)), measured by parental report. The outcome was zBMI. Height and weight were measured by trained research assistants and zBMI was determined according to WHO growth standards. A linear mixed effects model and logistic generalized estimating equations were used to determine the longitudinal association between cow’s milk fat intake and child zBMI.
Results
Among children aged 9 months to 8 years (N = 7467; 4699 of whom had repeated measures), each 1% increase in cow’s milk fat consumed was associated with a 0.05 lower zBMI score (95% CI −0.07 to −0.03, p < 0.0001) after adjustment for covariates including volume of milk consumed. Compared to children who consumed reduced fat (0.1–2%) milk, there was evidence that children who consumed whole milk had 16% lower odds of overweight (OR = 0.84, 95% CI 0.77 to 0.91, p < 0.0001) and 18% lower odds of obesity (OR = 0.82, 95% CI 0.68 to 1.00, p = 0.047).
Conclusions
Guidelines for reduced fat instead of whole cow’s milk during childhood may not be effective in preventing overweight or obesity.
Similar content being viewed by others
Introduction
One in three children in North America are living with overweight or obesity [1]. Childhood obesity also tracks into adulthood [2], increasing the risk of cardiovascular disease over the life course [3]. Current nutritional guidelines recommended limiting dietary fat in childhood, but evidence to support these guidelines is lacking [4]. The American Academy of Pediatrics and American Heart Association [5, 6], the Canadian Pediatric Society [7], and National Health Service (UK) [8] have recommended since the 1990’s that children consume whole (3.25% milk fat) cow’s milk until 2 years of age and then switch to reduced (0.1–2% milk fat) cow’s milk to lower the risk of overweight or obesity.
Cow’s milk is widely consumed in childhood [9, 10] and contains nutrients which support growth and development, such as protein, fat, calcium, phosphorous, and vitamin D. Cow’s milk has nutritional properties distinct from other dairy products such as yogurt, which often contains added sugar, or cheese, which contains a different quantity of fat and type of protein than fluid milk, and may have a unique relationship with adiposity [11, 12]. Two recent systematic reviews revealed that whole milk was associated with lower risk of child overweight or obesity among children aged 9 months to 18 years [13, 14]. These findings were largely based on low quality cross-sectional studies which did not adjust for potential confounding factors such as volume of milk consumed or previous child weight. Several systematic reviews have highlighted the need for additional high quality prospective cohort studies on this topic [15, 16]. This study is a longitudinal cohort study, including participants with and without multiple observations, in order to achieve the largest possible sample size and statistical power from this cohort. In contrast, our previous publication in this cohort was a cross-sectional analysis [17]. The current study was designed to overcome weaknesses of previous analyses and minimize risk of bias through a large prospective cohort study with adjustment for important potentially confounding factors.
The objective of this study was to evaluate the relationship between cow’s milk fat consumption and child adiposity, measured by body mass index z-score (zBMI) among healthy children aged 9 months to 8 years. We hypothesized that children who consumed higher intakes of cow’s milk fat would have lower zBMI and be less likely to be classified as overweight or obese compared to children who had lower intakes of cow’s milk fat.
Methods
Participants and setting
A prospective cohort study was conducted through the TARGet Kids! (The Applied Research Group for Kids) primary care research network in Toronto, Ontario, Canada. Children who were participating in regularly scheduled well-child visits, which occur at ages 2, 4, 6, 9, 12, 18, and 24 months, and annually thereafter, at 12 pediatric and family practices were recruited by trained research assistants in the clinic waiting room between August 2008 and April 2019 [18]. In TARGet Kids!, recruitment occurs between ages 0 and 5 years, and children are followed through subsequent well-child visits. Healthy children aged 9 months to 8 years who reported consuming cow’s milk were included in this analysis, in the interest of generating evidence about cow’s milk fat for children of all ages. Exclusion criteria were children with severe developmental delay, birth weight <2500 grams, chronic illness impacting metabolism (e.g., cystic fibrosis). All exposures, outcomes and covariates were measured at each well-child visit following enrollment in TARGet Kids!. All parents of children participating in this study provided informed written consent, and ethics approval was obtained from the Hospital for Sick Children (#10000-12436) and Unity Health Toronto (#17-335).
Exposure
The exposure was cow’s milk fat consumption. This was measured through a parent-completed dietary questionnaire, administered at a well-child visit, based on the Canadian Community Health Survey which has been in use since 2008 [19]. Parents were asked, “Please specify your child’s diet for the past 3 days: skim (0.1%), 1%, 2%, homo (3.25%) milk. Circle how many cups your child has currently in a typical day: 0, ½, 1, 2, 3, 4, 5+.” If parents indicated more than one milk fat, the mean cow’s milk fat intake was calculated. We also measured cow’s milk fat dichotomously as whole (3.25%) vs. reduced (0.1–2%) fat cow’s milk consumption.
Outcome
The outcome was body mass index z-score (zBMI), which is an age and sex adjusted measure of body weight. Trained research assistants measured child weight in kilograms and height in meters using a Healthometer digital baby scale and standardized recumbent length board (SECA, FL), respectively, for children younger than 2 years of age. Weight and height were measured using a Healthometer stadiometer (SECA, FL) for older children, according to standardized procedures [20]. Body mass index was calculated by dividing weight (kg) by height squared (m2) and z-scores were determined according to the World Health Organization (WHO) [21] growth standards. Categories for normal weight, overweight, and obesity were defined as: −2 ≤ zBMI ≤ 1 normal weight, 1 < zBMI ≤ 2 overweight, zBMI > 2 obesity, consistent with WHO recommended weight status categorization [21]. For the purpose of this manuscript and consistency across ages, we applied WHO weight classifications for children over age 5 years to all children in our sample [22, 23]. There is evidence that children younger than 5 years of age in the TARGet Kids! cohort exhibit functional outcomes of overweight, obesity, and severe obesity [24].
Covariates
Clinically relevant covariates which have been proposed in the literature to have a relationship with cow’s milk fat or childhood adiposity were specified a priori through a detailed literature review. These included volume of cow’s milk, volume of sugary drinks (including 100% juice, fruit drinks, and soft drinks) consumed in 250 ml cups per day, maternal ethnicity, self-reported family income, birth weight in kilograms, breastfeeding duration in months, and parent BMI. Dietary information, maternal ethnicity, family income, breastfeeding duration, and birth weight were collected using the same questionnaire as used to measure cow’s milk fat consumption. Volume of cow’s milk and sugary drinks was measured with the question, “Circle how many cups of each drink your child has currently in a typical day: 0, ½, 1, 2, 3, 4, 5+.” Daily physical activity was considered as a covariate but our previous research [17] demonstrated that it may not be a confounder in this relationship. Trained research assistants measured parental height and weight using a Healthometer stadiometer (SECA, FL) and BMI was calculated as weight (kg) divided by height squared (m2).
Statistical analysis
Descriptive statistics were used to characterize participants. To determine the longitudinal association between cow’s milk fat consumption and child zBMI as continuous variables, a linear mixed effects model was used. Linear mixed effects models have the advantage that all repeated measures provided by each participant can be used and within-subject correlation can be accounted for using subject specific random intercepts. Unadjusted and adjusted (see “Covariates” above) models were created with cow’s milk fat as the exposure and zBMI as the outcome. Restricted cubic splines for age with five knots were used to evaluate non-linear trajectories of the observed relationships in keeping with previous research in the same cohort [25]. Multinomial logistic generalized estimating equations (GEE) were used to examine the relationship between cow’s milk fat and odds of overweight excluding obesity (1 < zBMI ≤ 2) and obesity (zBMI >2) relative to normal weight (−2 ≤ zBMI ≤ 1), also adjusting for all covariates.
All covariates had <15% missing data. Missing data were handled using multiple imputation using 18 imputed datasets using all covariates [26]. Sensitivity analyses were conducted among children with and without missing data to assess for consistency of results. For all statistical tests, an alpha level of 0.05 was used and 95% confidence intervals were calculated. As the secondary objectives were hypothesis-generating, no adjustments for multiple comparisons were performed. The sample size for this analysis was dictated by the number of children enrolled in the ongoing registry. Multicollinearity was measured using the variance inflation factor, which remained under 1.5 for all covariates [27]. All analyses were conducted using the geepack [28], multgee [29], nlme [30], and lme4 [31] packages in R version 3.5.1 [32]. Statistical code used to produce the analysis is available upon request.
Results
A total of 7467 children aged 9 months to 8 years were included in the analysis (Fig. 1). Baseline characteristics of participating children are described in Table 1. Sixty-three percent (4699/7467) of children had repeated measures. At baseline, the mean child age was 2.6 (SD 1.5) years, and 53% of participants were male. The mean zBMI at baseline was 0.18 (SD 1.1); 77% of children had normal weight, 16% had overweight excluding obesity, 5% had obesity, and 2% had underweight. The majority (56%) of children consumed whole cow’s milk at baseline, followed by 2% milk (34% of children), 1% milk (8% of children), and skim milk (3% of children). On average, children consumed 1.9 (SD 1.2) 250-ml cups of cow’s milk per day at baseline. The mean follow-up duration was 2.7 years (SD 1.7).
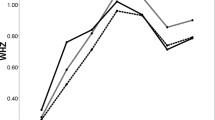
The linear mixed model revealed that with every 1% increase in cow’s milk fat consumed (for example, 2% vs. 1%, 1% vs. skim, etc.), children had 0.05 lower zBMI (95% CI −0.07 to −0.03, p < 0.0001), after adjustment for covariates including volume of milk consumed. Unadjusted and adjusted model results were unchanged (Table 2). For example, a 4-year-old child who consumed whole milk had on average 0.1 kg lower body weight than a 4-year-old child with the same height consuming reduced fat milk. Figure 2 shows the adjusted longitudinal relationship between cow’s milk fat, dichotomized into reduced (0.1–2%) fat and whole (3.25%) fat milk, and child zBMI.
Odds ratios for overweight and obesity among children who consumed whole vs. skim (0.1%), 1 and 2% cow’s milk were estimated (Table 3). When analyzed in a multinomial logistic GEE model, trends suggest that children who consumed whole cow’s milk had 16% lower odds of overweight (OR = 0.84, 95% CI 0.77–0.91, p < 0.0001) and 18% lower odds of obesity (OR = 0.82, 95% CI 0.68–1.00, p = 0.047) compared to children who consumed reduced (0.1–2%) fat cow’s milk, after adjustment.
To apply our analysis directly to current recommendations for children over age 2 years to consume reduced fat milk, we conducted a sensitivity analysis among children aged 2–8 years only. Among 6219 children aged 2–8 years of age, each 1% increase in cow’s milk fat consumed was associated with a 0.07 lower zBMI score (95% CI −0.09 to −0.05, p < 0.0001). Compared to children who consumed reduced fat (0.1–2%) cow’s milk, there was evidence that children who consumed whole cow’s milk had 10% lower odds of overweight (OR = 0.90, 95% CI 0.80–1.00, p = 0.05).
Discussion
In this prospective cohort study of 7467 healthy children aged 9 months to 8 years, higher cow’s milk fat consumption was associated with lower child zBMI after controlling for potential confounding factors including volume of milk consumed, parent BMI, and birth weight. On average, children who consumed whole milk had a 0.1 lower zBMI than children who consumed reduced fat milk, which is similar in magnitude to other dietary interventions for preventing obesity among children [33]. These findings were further supported by the evidence that children who consumed whole milk had 16% lower odds of overweight and 18% lower odds of obesity relative to children who consumed reduced fat (0.1–2%) milk.
Professional recommendations from the American Academy of Pediatrics [6], Academy of Nutrition and Dietetics [34], American Heart Association [5, 35], Health Canada [36] and the Canadian Pediatric Society [36], the National Health Service (UK) [8] and the Australian National Health and Medical Research Council [37] recommend that children consume whole cow’s milk until age 2 years, and then transition to reduced (0.1–2%) fat cow’s milk to reduce the risk of excess adiposity. These guidelines are based on consensus opinion [38] and evidence from the 1990s supporting that reduced fat cow’s milk is safe for children to consume [39, 40]. Our findings support the guideline for children aged 9 months to 2 years, but suggest that guidelines for older children may not be effective in preventing child overweight or obesity.
Findings from the present study are consistent with several other studies. A recent systematic review [14] and meta-analysis [13] identified observational studies which examined the relationship between cow’s milk fat and child adiposity among children aged 9 months to 18 years. An association between higher cow’s milk fat and lower adiposity was found in the majority of studies, and no study identified that reduced fat milk lowered the risk of child overweight or obesity. However, the majority of the studies were considered to have high risk of bias due to cross-sectional design or lack of adjustment for potential confounding factors such as volume of milk, prior measures of adiposity, and parent BMI [13]. The current study was designed to overcome these weaknesses through a large prospective cohort study with adjustment for important potentially confounding factors. Our findings are also consistent with a RCT of children aged 4–13 years which showed no evidence of a relationship between dairy fat (including milk, cheese, and yogurt) intake and child adiposity [41].
Possible mechanisms underlying the observed relationship include reverse causality, where parents of leaner children provide higher cow’s milk fat and vice versa. Another possibility is that children who consume higher cow’s milk fat may be more satiated than those who consume reduced fat cow’s milk, leading them to consume a lower quantity of cow’s milk or other energy dense foods contributing to higher energy intake [42]. Hormones secreted in response to whole milk consumption such as cholecystokinin and glucagon-like peptide 1 may also play a role in this relationship [43, 44]. Cow’s milk fat contains unique fatty acids such as trans-palmitoleic acid and conjugated linoleic acid [45], which may provide cardiometabolic benefits relative to other fatty acids [46]. This theory suggests that cow’s milk fat may not contribute to energy storage and adipose tissue as significantly as other types of dietary fat [47, 48]. Some evidence suggests that children who consume a lower proportion of dietary fat early in life may develop a “thrifty metabolism,” where the body adapts by storing excess energy as fat [49]. Conversely, a higher fat diet may metabolically program higher energy utilization and lower energy storage [49, 50]. A lower fat diet in early life may program the body to favor energy storage over utilization, which may increase the risk of obesity over the life course [51,52,53,54]. Future studies are needed to understand the mechanism underlying the relationship between cow milk fat and child adiposity, including randomized controlled trials to mitigate influence of confounding factors and establish temporality.
This study had several strengths. The large, diverse cohort of healthy children who were followed over many years provided rich information about nutrition through early and middle childhood. In addition, a number of clinically relevant covariates were controlled for which reduced the risk of confounding. Complex statistical modeling using random effects models minimized within-subject correlation and improved model estimate accuracy. Restricted cubic splines allowed for evaluation of non-linear trends over time. Repeated measures data allowed us to account for the directionality of this relationship, which improved upon the cross-sectional methods of previous studies by adding power and adjusting for within-subject measures, including changes in cow’s milk fat intake from one observation to the next.
This study had a number of weaknesses. The clinical significance of small differences in zBMI in children is not clear. However, the observed difference in weight for a 4-year-old child who consumed whole vs. reduced fat milk seems meaningful. Although cow’s milk volume and sugary drink consumption were accounted for, there were insufficient data to calculate total daily energy which would have improved our understanding of the mechanism underlying the observed relationship. The questionnaire used to capture participant diets was based on the Canadian Community Health Survey [55], but has not yet been validated. Though we were able to follow children over an average of 2.7 years, longer follow-up duration would have allowed a better understanding of the temporality of the observed relationships. While zBMI is a measure preferred by clinicians because it is simple to interpret and track over time, and the WHO growth standards are considered the gold standard for zBMI classification [56], BMI is not a direct measure of body composition [57]. Stadiometers used to measure height and weight in this study are routinely checked for accuracy but calibration protocols are not consistent across primary care practices, which may have led to measurement errors. Our sample included a relatively small proportion (5% of the total sample) of children with obesity, which may have limited our statistical power to explore the relationship between cow’s milk fat and adiposity among these children, especially in our secondary analysis which estimated odds of overweight and obesity among children who consumed skim, 1%, and 2% compared to whole milk. Imbalance between groups was expected, because obesity was classified based on z-score cut-offs at the tails. While participants in this study were from an ethnically diverse population of healthy of urban Canadian children, they may not be representative of other groups of children. Further, cow’s milk with different fat contents may have been offered to children based on parent perception of body size. Data on reasons for milk fat choices would have helped to clarify whether reverse causality contributed to the observed results, but this was not available.
In this prospective cohort study of healthy children age 9 months to 8 years, higher cow’s milk fat consumption was associated with lower zBMI and lower odds of overweight and obesity. Given that the majority of North American children consume cow’s milk daily, this study raises the possibility that whole cow’s milk may play a role in lowering the risk of childhood overweight or obesity. Randomized trial data showing the relationship between cow’s milk fat and child adiposity is needed to confirm these findings.
References
Abarca-Gómez L, Abdeen ZA, Hamid ZA, et al. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. The Lancet. 2017;390:2627–42. https://doi.org/10.1016/S0140-6736(17)32129-3.
Bancej C, Jayabalasingham B, Wall RW, Rao DP, Do MT, de Groh M, et al. Evidence brief—trends and projections of obesity among Canadians. Health Promot Chronic Dis Prev Can. 2015;35:109–12.
Public Health Agency of Canada. Actions taken and future directions 2011: curbing childhood obesity: a federal, provincial and territorial framework for action to promote healthy weights. Government of Canada. Ottawa, ON; 2011.
Harcombe Z. Dietary fat guidelines have no evidence base: where next for public health nutritional advice? Br J Sports Med. 2017;51:769–74.
Lott M, Callahan E, Welker Duffy E, Story M, Daniels S. Healthy Beverage Consumption in Early Childhood: Recommendations from Key National Health and Nutrition Organizations. Consensus Statement. Durham, NC: Healthy Eating Research, 2019. Available at https://healthyeatingresearch.org
Gidding SS, Dennison BA, Birch LL, Daniels SR, Gillman MW, Lichtenstein AH, et al. Dietary recommendations for children and adolescents: a guide for practitioners. Pediatrics. 2006;117:544–59.
Canadian Pediatric Society. Feeding your baby in the first year: Canadian Pediatric Society. 2014. Accessed May 2020. https://www.caringforkids.cps.ca/handouts/feeding_your_baby_in_the_first_year
National Health Service. What to feed young children UK. 2016. https://www.nhs.uk/conditions/pregnancy-and-baby/understanding-food-groups/-milk-and-dairy-products.
Maillot M, Rehm CD, Vieux F, Rose CM, Drewnowski A. Beverage consumption patterns among 4-19 y old children in 2009-14 NHANES show that the milk and 100% juice pattern is associated with better diets. Nutr J. 2018;17:54.
Garriguet D. Beverage consumption of children and teens. Statistics Canada, Catalogue no. 82-003-XPE. Health Reports. 2008;19:4.
Thorning TK, Bertram HC, Bonjour JP, de Groot L, Dupont D, Feeney E, et al. Whole dairy matrix or single nutrients in assessment of health effects: current evidence and knowledge gaps. Am J Clin Nutr. 2017;105:1033–45.
Moreno LA, Bel-Serrat S, Santaliestra-Pasias A, Bueno G. Dairy products, yogurt consumption, and cardiometabolic risk in children and adolescents. Nutr Rev. 2015;73(Suppl 1):8–14.
Vanderhout SM, Aglipay M, Torabi N, Juni P, da Costa BR, Birken CS, et al. Whole milk compared with reduced-fat milk and childhood overweight: a systematic review and meta-analysis. Am J Clin Nutr. 2019;111:266–79.
O'Sullivan TA, Schmidt KA, Kratz M. Whole-fat or reduced-fat dairy product intake, adiposity, and cardiometabolic health in children: a systematic review. Adv Nutr. 2020;11:928–50. https://doi.org/10.1093/advances/nmaa011.
Dror DK. Dairy consumption and pre-school, school-age and adolescent obesity in developed countries: a systematic review and meta-analysis. Obes Rev. 2014;15:516–27.
Louie JC, Flood VM, Hector DJ, Rangan AM, Gill TP. Dairy consumption and overweight and obesity: a systematic review of prospective cohort studies. Obes Rev. 2011;12:e582–92.
Vanderhout SM, Birken CS, Parkin PC, Lebovic G, Chen Y, O’Connor DL, et al. Relation between milk-fat percentage, vitamin D, and BMI z score in early childhood. Am J Clin Nutr. 2016;104:1657–64.
Carsley S, Borkhoff CM, Maguire JL, et al. Cohort Profile: The Applied Research Group for Kids (TARGet Kids!). International Journal of Epidemiology. 2015;44:776–88. https://doi.org/10.1093/ije/dyu123.
Statistics Canada. Canadian Community Health Survey—annual component (CCHS). Ottawa: Statistics Canada; 2013.
Centers for Disease Control and Prevention. Third National Health and Nutrition Examination (NHANES III). [Video]. Pittsburgh, PA: Centers for Disease Control and Prevention and National Center for Health Statistics; 2003.
Guideline: assessing and managing children at primary health-care facilities to prevent overweight and obesity in the context of the double burden of malnutrition. Updates for the Integrated Management of Childhood Illness (IMCI). Geneva: World Health Organization; 2017. Licence: CC BY-NC-SA 3.0 IGO.
Satkunam M, Anderson LN, Carsley S, Maguire JL, Parkin PC, Sprague AE, et al. Severe obesity in children 17 to 24 months of age: a cross-sectional study of TARGet Kids! and Better Outcomes Registry & Network (BORN) Ontario. Can J Public Health. 2018;109:489–97.
Carsley S, Pope EI, Anderson LN, Tremblay MS, Tu K, Birken CS, et al. Temporal trends in severe obesity prevalence in children and youth from primary care electronic medical records in Ontario: a repeated cross-sectional study. CMAJ Open. 2019;7:E351–9.
Carsley SE, Anderson LN, Plumptre L, Parkin PC, Maguire JL, Birken CS. Severe obesity, obesity, and cardiometabolic risk in children 0 to 6 years of age. Child Obes. 2017;13:415–24.
Eny KM, Chen S, Anderson LN, Chen Y, Lebovic G, Pullenayegum E, et al. Breastfeeding duration, maternal body mass index, and birth weight are associated with differences in body mass index growth trajectories in early childhood. Am J Clin Nutr. 2018;107:584–92.
van Buuren S. mice Package for R. 2019. https://cran.r-project.org/web/packages/mice/mice.pdf
O’Brien R. A caution regarding rules of thumb for variance inflation factors. Qual Quant. 2007;41:673–90.
Hojsgaard SHU, Yan J. Package ‘geepack’. 1.2-1 ed. 2016. https://cran.r-project.org/web/packages/geepack/geepack.pdf
Touloumis A. multgee: GEE Solver for Correlated Nominal or Ordinal Multinomial Responses. 1.6.0 ed. 2017. https://cran.rproject.org/web/packages/multgee/multgee.pdf
Pinheiro JBD, DebRoy S, Sarkar D, Heisterkamp S, Van Willigen B. Package ‘nlme’. 3.1-141 ed. 2019. https://cran.rproject.org/web/packages/nlme/nlme.pdf
Bates DMM, Bolker B, Walker S, Christensen RHB, Singmann H, Daj B, et al. Package ‘lme4’. 1.1-12 ed. 2019. https://cran.r-project.org/web/packages/lme4/lme4.pdf
R Core Team. R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2014. https://www.r-project.org/
Brown T, Moore TH, Hooper L, Gao Y, Zayegh A, Ijaz S, et al. Interventions for preventing obesity in children. Cochrane Database Syst Rev. 2019;7:CD001871.
Hoelscher DM, Kirk S, Ritchie L, Cunningham-Sabo L,Academy Positions Committee.Position of the academy of nutrition and dietetics: interventions for the prevention and treatment of pediatric overweight and obesity. J Acad Nutr Diet. 2013;113:1375–94.
Sacks FM, Lichtenstein AH, Wu JHY, Appel LJ, Creager MA, Kris-Etherton PM, et al. Dietary fats and cardiovascular disease: a presidential advisory from the American Heart Association. Circulation. 2017;136:e1–23.
Critch JN, Canadian Paediatric Society; Nutrition and Gastroenterology Committee. Nutrition for healthy term infants, six to 24 months: an overview. Paediatr Child Health. 2014;19:547–52.
NHMRC (National Health and Medical Research Council). Infant Feeding Guidelines: Summary. Canberra: National Health and Medical Research Council. 2013.
Allen RE, Myers AL. Nutrition in toddlers. Am Fam Physician. 2006;74:1527–32.
Obarzanek E, Hunsberger SA, Van Horn L, Hartmuller VV, Barton BA, Stevens VJ, et al. Safety of a fat-reduced diet: the Dietary Intervention Study in Children (DISC). Pediatrics. 1997;100:51–9.
Niinikoski H, Viikari J, Ronnemaa T, Helenius H, Jokinen E, Lapinleimu H, et al. Regulation of growth of 7- to 36-month-old children by energy and fat intake in the prospective, randomized STRIP baby trial. Pediatrics. 1997;100:810–6.
Hendrie GA, Golley RK. Changing from regular-fat to low-fat dairy foods reduces saturated fat intake but not energy intake in 4-13-y-old children. Am J Clin Nutr. 2011;93:1117–27.
Kavezade SAM, Asemi M, Salehi-Abargouei A. The effects of whole milk compared to skim milk and apple juice consumption in breakfast on appetite and energy intake in obese children: a three-way randomized crossover clinical trial. BMC Nutr. 2018;4:1–10.
Montmayeur, J.-P. Fat Detection: Taste, Texture, and Post Ingestive Effects (1st ed.). CRC Press, Boca Raton. 2009. https://doi.org/10.1201/9781420067767.
Naslund E, Bogefors J, Skogar S, Gryback P, Jacobsson H, Holst JJ, et al. GLP-1 slows solid gastric emptying and inhibits insulin, glucagon, and PYY release in humans. Am J Physiol. 1999;277:R910–6.
Helena Lindmark Månsson. Fatty acids in bovine milk fat, Food & Nutrition Research. 2008;52:1,1821. https://doi.org/10.3402/fnr.v52i0.1821.
Haug A, Hostmark AT, Harstad OM. Bovine milk in human nutrition-a review. Lipids Health Dis. 2007;6:25.
Mozaffarian D, Cao H, King IB, Lemaitre RN, Song X, Siscovick DS, et al. Trans-palmitoleic acid, metabolic risk factors, and new-onset diabetes in U.S. adults: a cohort study. Ann Intern Med. 2010;153:790–9.
Racine NM, Watras AC, Carrel AL, Allen DB, McVean JJ, Clark RR, et al. Effect of conjugated linoleic acid on body fat accretion in overweight or obese children. Am J Clin Nutr. 2010;91:1157–64.
Rolland-Cachera MF, Akrout M, Peneau S. Nutrient intakes in early life and risk of obesity. Int J Environ Res Public Health. 2016;13. https://doi.org/10.3390/ijerph13060564.
Rolland-Cachera MF, Maillot M, Deheeger M, Souberbielle JC, Peneau S, Hercberg S. Association of nutrition in early life with body fat and serum leptin at adult age. Int J Obes. 2013;37:1116–22.
Giudici KV, Rolland-Cachera MF, Gusto G, Goxe D, Lantieri O, Hercberg S, et al. Body mass index growth trajectories associated with the different parameters of the metabolic syndrome at adulthood. Int J Obes. 2017;41:1518–25.
Monteiro PO, Victora CG, Barros FC, Monteiro LM. Birth size, early childhood growth, and adolescent obesity in a Brazilian birth cohort. Int J Obes Relat Metab Disord. 2003;27:1274–82.
Ong KK, Ahmed ML, Emmett PM, Preece MA, Dunger DB. Association between postnatal catch-up growth and obesity in childhood: prospective cohort study. BMJ. 2000;320:967–71.
Peneau S, Giudici KV, Gusto G, Goxe D, Lantieri O, Hercberg S, et al. Growth trajectories of body mass index during childhood: associated factors and health outcome at adulthood. J Pediatr. 2017;186:64–71 e1.
Statistics Canada. Canadian Community Health Survey—Annual Component (CCHS): Government of Canada. 2014. http://www23.statcan.gc.ca/imdb/p2SV.pl?Function=getSurvey&SDDS=3226.
Shields M, Tremblay MS. Canadian childhood obesity estimates based on WHO, IOTF and CDC cut-points. Int J Pediatr Obes. 2010;5:265–73.
Dietz WH, Story MT, Leviton LC. Issues and implications of screening, surveillance, and reporting of children’s BMI. Pediatrics. 2009;124(Suppl 1):S98–101.
Acknowledgements
Funding was provided by the Canadian Institutes of Health Research (CIHR) Institute of Human Development, Child and Youth Health (grant number MOP-333560). The funding agency had no role in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
SMV and JLM conceptualized and designed the research study, performed initial statistical analyses, drafted the manuscript, approved the final manuscript as submitted, and had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. CSB and DLO assisted in refining the study design, reviewed and revised the manuscript, and approved the final manuscript as submitted. KET and CDGKS reviewed and revised statistical analysis as well as the manuscript, and approved the final manuscript as submitted.
Corresponding author
Ethics declarations
Competing interests
JLM received an unrestricted research grant for a completed investigator-initiated study from the Dairy Farmers of Canada (2011–2012) and Ddrops provided non-financial support (vitamin D supplements) for an investigator-initiated study on vitamin D and respiratory tract infections (2011–2015). All other authors have no potential conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vanderhout, S.M., Keown-Stoneman, C.D.G., Birken, C.S. et al. Cow’s milk fat and child adiposity: a prospective cohort study. Int J Obes 45, 2623–2628 (2021). https://doi.org/10.1038/s41366-021-00948-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-021-00948-6
This article is cited by
-
The role of dairy consumption in the relationship between wealth and early life physical growth in India: evidence from multiple national surveys
BMC Public Health (2024)
-
Centre-based childcare in early childhood and growth in later childhood: a prospective cohort study
International Journal of Obesity (2023)