Abstract
Fundamental questions remain about the optimal temperature, duration, and mode of delivery that provide the best striatal neuroprotection from hypothermia after perinatal hypoxia-ischemia. This study used stereological methods to investigate whether a mild (i.e. 2°C) or a moderate (5°C) decrease in whole body temperature, for 6 h immediately postinsult, was neuroprotective for striatal medium-spiny neurons after perinatal hypoxia-ischemia in the rat. This study also investigated whether moderate hypothermia had any effect on normal striatal development. Hypoxia-ischemia or sham hypoxia-ischemia was induced on postnatal day (PN) 7. Pups were kept either normothermic, mildly hypothermic, or moderately hypothermic for 6 h immediately postinsult. The absolute number of striatal medium-spiny neurons was calculated using modern stereological methods. There was no significant difference in the absolute number of medium-spiny neurons in the right striatum after either mild hypothermia or moderate hypothermia. There was also no significant difference in the absolute number of medium-spiny neurons between the control normothermic and the control moderately hypothermic pups. The latter results suggest that moderate hypothermia for 6 h immediately postinsult may be a safe treatment for striatal medium-spiny neurons. Yet, neither mild nor moderate hypothermia alone for 6 h immediately posthypoxia-ischemia is neuroprotective for striatal medium-spiny neurons.
Similar content being viewed by others
Main
Perinatal cerebral hypoxia-ischemia is a leading cause of acute brain injury, which can lead ultimately to cerebral palsy (1). Recent clinical trials in hypoxic-ischemic (H/I) neonates indicate that posttreatment with mild to moderate hypothermia can improve the combined outcome of death or severe disability for some infants (2,3). The improved outcomes were evident after treatment with mild or moderate hypothermia for 48–72 h, with the onset of treatment commencing within 6 h of birth or H/I event (2,3). While these findings are promising, fundamental important questions remain about the optimal temperature, duration, and mode of delivery that provide the best neuroprotection from hypothermia (4). Thus, the first aim of this study was to investigate whether mild or moderate hypothermia for less than 48 h immediately after hypoxia-ischemia was neuroprotective when administered systemically (i.e. as whole-body cooling).
More specifically, this study investigated whether a mild (i.e. 2°C) or a moderate (5°C) decrease in whole-body temperature for 6 h immediately postinsult was neuroprotective for striatal medium-spiny neurons after perinatal hypoxia-ischemia in the rat. Mild hypothermia for 6 h was investigated because mild hypothermia for 3 h postinsult delayed striatal injury but was not neuroprotective (5). Moderate hypothermia for 6 h was investigated because moderate hypothermia for 3–6 h has yielded conflicting evidence on striatal protection (6,7). The striatum was chosen because it is one of the main sites of injury after acute perinatal hypoxia-ischemia in both the rat (8,9) and human (10). In humans, striatal injury is associated with the choreoathetotic movement disorders of cerebral palsy (10). The predominant medium-spiny neurons of the striatum were investigated because they comprise more than 97% of all striatal neurons (11). The absolute number of surviving striatal medium-spiny neurons was measured in each animal using modern stereological methods. This allowed reliable comparisons to be made (9,11,12).
The second aim of this study was to investigate whether moderate hypothermia for 6 h immediately postinsult had any effect on normal striatal development. It is possible that hypothermia-induced decreases in blood flow, oxygen consumption and cerebral energy utilization rate (13,14) could have affected normal programmed cell death during development. Such effects could have resulted in changes in the absolute number of medium-spiny neurons in the striatum. To the best of our knowledge, there have been no previous experiments that have examined the effect of moderate hypothermia on the normal development of the striatum.
MATERIALS AND METHODS
Pregnant albino Sprague-Dawley rats from the University of Otago Animal Breeding Station were housed individually, under controlled environmental conditions, with food and water freely available. All procedures were approved by the Committee on Ethics in the Care and Use of Laboratory Animals at the University of Otago.
On postnatal day 7 (PN7), the 10 pups per litter were separated from the dam, sexed, and the males weighed. The heaviest male pups were paired according to body weight. A difference of ≤4% in body weight was deemed acceptable for pairing. The heaviest pup in each pair was randomly assigned to a treatment or control group. The second pup in the pair was assigned to the alternate group. There were two matched pairs of pups per litter. The four male pups per litter were then subjected to a variation of the Rice et al. (8) model of acute H/I brain injury during the human third trimester equivalent. These methods have been previously described (9,11,12). Briefly, each pup was anesthetized with ether and the right common carotid artery was permanently ligated using sterile 5/0 surgical silk. Two and a half hours after completion of the last surgery, the pups were exposed to a hypoxic environment of humidified 8% oxygen/92% nitrogen for 1.5 h at 37°C. A hypoxic exposure of 1.5 h was chosen since it induces brain pathology with minimal mortality (9,11,12). This procedure and the subsequent procedures were repeated on a total of 32 pups from eight litters.
Sixteen pups from four of these litters were subsequently exposed to either mild hypothermia (n = 8 pups) or normothermia (n = 8). Another 16 pups from another four litters were exposed to either moderate hypothermia (n = 8 pups) or normothermia (n = 8). A statistical power calculation showed that eight H/I pups per treatment group would give a 70% chance of detecting, at the 5% level for a two-tailed test, a treatment difference of 33% or more (15). This power calculation was based on the variability of previous estimates of the absolute number of striatal medium-spiny neurons at PN14 in rats exposed to H/I at PN7 (12). Eight H/I pups per treatment group has yielded statistically significant neuroprotection of striatal medium-spiny neurons (12).
To investigate the effect of moderate hypothermia on normal brain development, another group of six male pups from two litters were subjected to sham-ligation of the right common carotid artery followed by 1.5 h of hypoxia. Sham-ligation followed by hypoxia does not reduce the absolute number of striatal medium-spiny neurons compared with naive control animals (9). The pups were then exposed immediately to either moderate hypothermia or normothermia for 6 h to yield three moderate hypothermia-treated normal pups and three normothermia-treated normal pups.
At the completion of 1.5 h of hypoxia for the 38 pups from 10 litters, the rectal core temperature was measured before the start, and then during the hypothermia or normothermia, to assess the degree of hypothermia that was induced. A correlation between rectal core temperature and brain temperature in the PN7 rat has previously been established (6,14). At the cessation of hypoxia, each pup was briefly transferred on a 37°C gel pad to a rectal probe station. As soon as the initial measurement of the rectal temperature had been completed, each pup was transferred on a 37°C gel pad to an individual click-clack box in a waterbath to commence the 6 h of hypothermia or normothermia. The bath water was maintained at 31°C to achieve mild hypothermia, 26°C to achieve moderate hypothermia, or 37°C to achieve normothermia. Rectal temperatures were also measured at 0.5, 3, and 6 h for each litter. After the last temperature measurement at 6 h, the pups were returned to their littermates and dam.
From PN7 to PN14, each experimental pup was weighed daily to monitor their general well being. On PN14, the rats were anesthetized, perfused intracardially, and each brain was processed, as previously described (12). Each right cerebral hemisphere was embedded, in toto, in glycolmethacrylate (Technovit 7100, Kulzer and Co., Wehrhein, Germany) as previously described (16). Serial, 40-μm coronal sections were cut through the entire right striatum of each animal on a Microm HM355S rotary microtome. After a random start, every fourth section was collected on gelatin-coated slides until the striatum was no longer present, stained with 0.003% cresyl violet, dehydrated, and coverslipped as previously described (16).
Stereological quantitation was carried out on every eighth section of the brain containing the striatum. All slides were coded before stereological analyses. The Cavalieri and optical disector methods (17,18) were used to measure the total number of medium-spiny neurons in the striatum. The Cavalieri method was used to measure the total reference volume (Vref). The number of medium-spiny neurons per unit volume of the striatum (i.e. the neuronal density or NV) was measured using the optical disector method. The boundaries of the striatum and the identification of a medium-spiny neuron were as defined by Oorschot (16). Specific details on the stereological equipment and sampling parameters used are described in Table 1 (see footnote). An estimate of the total (i.e. absolute) number of medium-spiny neurons (N) was then obtained by N = Vref × NV.
With data derived from a set of systematically sampled sections from an individual, it is possible to estimate the precision of the estimates made on that individual. These estimates of precision, termed the coefficient of the error (CE) of the estimates, were calculated for Vref and N using the nugget variance (19). The CE of NV was calculated using 1/(ΣQ−)1/2. All quantitative data, except rectal temperature and body weight, were compared using the Mann-Whitney U test. A repeated measures ANOVA was used to compare the effect of hypothermia on rectal temperature for 6 h on PN7 and on body weight from PN7 to PN14.
RESULTS
An average temperature differential of 2.17°C ± 0.84 (mean ± SD) was evident between 30 min and 6 h for the H/I pups in mild hypothermia compared with the H/I pups in normothermia (e.g. at 6 h, H/I mild hypothermia 33.18°C ± 0.39 versus H/I normothermia 35.39°C ± 0.74, Fig. 1A). Statistical analysis of these temperatures over 0–6 h revealed a significant difference between the H/I normothermia and H/I mild hypothermic groups (F (1,6) = 9.413, p < 0.022, ANOVA repeated measures). An average temperature differential of 5.43°C ± 0.15 was evident between 30 min and 6 h for the H/I pups in moderate hypothermia compared with the H/I pups in normothermia (e.g. at 6 h, H/I moderate hypothermia 28.80°C ± 0.77 versus H/I normothermia 34.15°C ± 0.58, Fig. 1B). Statistical analysis of these temperatures over 0 to 6 h revealed a significant difference between the H/I normothermia and H/I moderate hypothermic groups (F (1,6) = 75.057, p < 0.001, ANOVA repeated measures). For the sham-ligated control pups, moderate hypothermia resulted in a temperature differential of 5.58°C ± 0.41 between 30 min and 6 h compared with the sham-ligated pups in normothermia (e.g. at 6 h, moderate hypothermia 28.65°C ± 0.28 versus normothermia 34.68°C ± 0.50, Fig. 1C). Statistical analysis of these temperatures over 0–6 h revealed a significant difference between the sham-ligated normothermic and moderate hypothermic groups (F (1,2) = 19.870, p < 0.047, ANOVA repeated measures).
There was no significant difference in the average body weight from PN7 until PN14 when the H/I-normothermia-treated and the H/I-hypothermia-treated groups were compared after either mild or moderate hypothermia for 6 h on PN7 (e.g. H/I normothermia versus H/I moderate hypothermia, F (1,14) = 0.073, p < 0.791, ANOVA repeated measures). Both the H/I-normothermia- and H/I-hypothermia-treated pups also showed a significant overall increase in body weight from PN7 to PN14 after mild or moderate hypothermia for 6 h on PN7 (e.g. H/I normothermia versus H/I moderate hypothermia, F (7,98) = 486.944, p < 0.001, ANOVA repeated measures). For the sham-ligated hypoxia-alone normal animals, there was no significant difference in the average body weight from PN7 until PN14 when the normothermia-treated and the moderate hypothermia-treated groups were compared after moderate hypothermia for 6 h on PN7 (F (1,14) = 0.191, p < 0.685, ANOVA repeated measures). Both the normothermic- and moderate-hypothermia-treated normal pups also showed a significant overall increase in body weight from PN7 to PN14 after moderate hypothermia for 6 h on PN7 (e.g. F (7,28) = 484.351, p < 0.001, ANOVA repeated measures). Thus, mild or moderate hypothermia for 6 h on PN7 did not have any adverse effects on the general health of the animals.
Comparison of the weight of the right cerebral hemispheres revealed that there was no significant difference between the H/I mild hypothermic pups and the H/I normothermic pups (p = 0.798, two-tailed Mann-Whitney U test, Table 1), the H/I moderate hypothermic pups and their own control group of H/I normothermic pups (p = 0.798), and the sham-ligated moderate hypothermic and normothermic pups (p = 0.400).
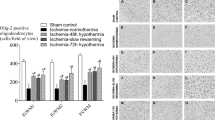
Statistical comparison of the total volume (Vref) of the right striatum, and the neuronal density (NV) of striatal medium-spiny neurons, was undertaken (see Table 1) to enable a comparison with the findings from the absolute number data (see Discussion). The absolute number data showed that there was no significant difference between the H/I mild hypothermic rat pups and the H/I normothermic pups (p = 1.000, two-tailed Mann-Whitney U test, Table 1). There was also no significant difference in the absolute number of striatal medium-spiny neurons when the H/I moderate hypothermic rat pups were compared with their H/I normothermic group (p = 0.442, Table 1, Fig. 2). There was some variability in the average absolute number of striatal medium-spiny neurons in the two H/I normothermic groups of this study (Table 1, Fig. 2), yet the difference in the average absolute number was not statistically significant (p = 0.160, two-tailed Mann-Whitney U test). To control for inter-litter variation in striatal injury due to H/I, hypothermic-treated pups were always matched with normothermic-treated pups from the same litter.
Scatter plots of the total striatal volume (A), number of striatal medium-spiny neurons per mm3, NV (B) and total number of striatal medium-spiny neurons, N (C) data in Table 1. Mild, mild hypothermia; N, normothermia; Mod, moderate hypothermia; Sham, sham-ligated control.
In the control experiment to assess the effect of moderate hypothermia on normal striatal development, it was found that there was no significant difference in the absolute number of striatal medium-spiny neurons when the sham-ligated hypoxia-alone, moderate hypothermia rat pups were compared with the sham-ligated hypoxia-alone, normothermia pups (p = 0.500, one-tailed Mann-Whitney U test, Table 1, Fig. 2). There were significantly more surviving neurons in the sham-ligated normothermia control pups compared with each H/I normothermia control group (see Table 1 footnote, Fig. 2). This supports previous findings that around 50% of striatal medium-spiny neurons are injured, on average, after neonatal H/I (9,11,12).
The stereological measurements generally fulfilled the guidelines in the literature (see Table 1 footnote). This yielded reliable estimates, with mean coefficients of error for the sum of the points counted (Vref), the number of disector neurons sampled per disector volume (NV), and the absolute number of striatal medium-spiny neurons, that were below 0.10 (Table 2).
The estimate of the mean total volume of the striatum, and the mean NV and the mean total number of striatal medium-spiny neurons, in all the H/I hypothermia-treated animals and the H/I normothermia-treated animals was also considered precise enough. This is because the mean variability of the individual estimates for each specific parameter in a group (i.e. the CE2, Table 2) was generally less than half of the variability for the estimate of the group (i.e. the CV2, Table 1). For example, the observed mean variance of the individual absolute number estimates (e.g. CE2 = 0.0622 and 0.0752, respectively, for the H/I moderate-hypothermia-treated group and its H/I normothermia control, Table 2) were less than half the variance of the group (CV2 = 0.2602 and 0.4212, respectively; Table 1; see also Table 1 footnote). Therefore, in general, for every H/I group, the major factor contributing to the overall variability was the true interanimal differences in the total volume, NV, and total neuronal number, and not the precision of the estimates made with the stereological techniques used (16,19).
For the sham-ligated, control groups exposed to either moderate hypothermia or normothermia, the variability (i.e. CV) for the total volume, NV, and absolute number measurements was low (i.e. 1.5–4.2%, Table 1). This may reflect the lower number of animals and litters in these groups. A low CV meant that the CE2 was not less than half the CV2 for the stereological measurement of the total volume, NV and absolute number for each of these groups (Table 2). Yet, the average absolute number of striatal medium-spiny neurons obtained for each group (i.e. moderate hypothermia, 2,616,000 ± 82,000, mean ± SD; normothermia, 2,585,000 ± 102,000; Table 1) is statistically comparable to previous results for normal control animals [2,791,000 ± 188,000, Oorschot (16); 2,583,000 ± 287,000, 2,684,000 ± 167,000, Galvin and Oorschot (9)]. In these studies, there were a greater number of animals and litters per group, a higher CV per group, and all stereological measurements fulfilled the requirement that the CE2 was less than half the CV2 (9,16). Thus, the average absolute number of striatal medium-spiny neurons is likely to be reliable for the sham-ligated control groups of the current study.
DISCUSSION
This unique stereological study examined the effect of mild or moderate hypothermia, administered immediately postinsult, on the absolute number of surviving striatal medium-spiny neurons after acute neonatal hypoxia-ischemia. Neither mild nor moderate hypothermia for 6 h immediately postinsult significantly increased the absolute number of striatal medium-spiny neurons. Moderate hypothermia for 6 h immediately postinsult had no adverse effects on the development of the striatal medium-spiny neurons in normal rat pups. These stereological data are important because it helps to establish the precise hypothermia dose-response relationship for the striatum. This is needed so that optimal neuroprotective treatment strategies can be refined and designed for the striatum after neonatal hypoxia-ischemia.
The effect of mild hypothermia for 6 h immediately postinsult was investigated stereologically because mild hypothermia for 3 h postinsult delayed rat striatal injury but was not neuroprotective (5). The failure to observe striatal neuroprotection after mild hypothermia for 6 h suggests that mild hypothermia for a longer duration warrants investigation. It is possible that mild hypothermia for 6 h is not sufficient to cause a significant decrease in the severity of the secondary energy failure after a H/I insult. Although hypothermia has been found to modulate secondary energy failure (13,14), it is possible that a 2°C drop in body temperature for 6 h is not enough. Thoresen et al. (20) have theorized that sedation may be a necessary component in the reduction of neuropathological damage by mild hypothermia. The increase in stress due to hypoxia and hypothermia may be the cause of the failure of mild hypothermia to induce neuroprotection (20). It is possible that stress could play a factor in our study. The pups are individually housed for the length of the hypothermic exposure and separation stress could adversely influence the results. The effect of sedation (e.g. xenon, 21) and mild hypothermia for 6 h on the survival of neonatal rat striatal medium-spiny neurons now warrants investigation.
There was no significant neuroprotection of rat striatal medium-spiny neurons after moderate hypothermia for 6 h immediately postinsult. Bona et al. (7) also reported no significant neuroprotection for the rat basal ganglia after 6 h of moderate hypothermia immediately postinsult. In contrast, Thoresen et al. (6) reported significant protection of the rat basal ganglia after 3 h of moderate hypothermia immediately postinsult. Since the current study used rigorous stereological quantitation of total number, rather than a qualitative neuropathological scoring technique (6,7), it seems likely that moderate hypothermia for 6 h immediately postinsult is not significantly neuroprotective for striatal medium-spiny neurons.
We have published a positive control for the same experimental paradigm that shows statistically significant neuroprotection of striatal medium-spiny neurons by brain derived neurotrophic factor or neurotrophin-3 (12). Thus, neuroprotection is possible in this experimental paradigm.
At the time of this study, a duration of hypothermia of longer than 6 h was not ethically possible for individually housed PN7 rat pups at the University of Otago. Yet, a more recent study has exposed PN7 rats to a 10-h exposure of moderate hypothermia and reported protection for hippocampal neurons after semi-quantitative analyses (22). This study may provide an ethical precedent for future stereological studies on the effect of 10 h of moderate hypothermia at PN7 on the total number of surviving striatal medium-spiny neurons. It needs to be acknowledged that the PN7 rats in Zhu et al. (22) were group-housed during hypothermia, rather than individually housed as in the current study. Yet the clinical relevance of individual housing during hypothermia seems to justify investigation of the effect of 10 h of moderate, or mild, hypothermia on individually housed PN7 rats.
Investigation of the effect of hypothermia for longer durations also merits investigation because moderate hypothermia for 6 h had no adverse effects on the normal number of striatal medium-spiny neurons. Previously there have been no experiments which have examined the effect of moderate hypothermia on the normal development of the striatum (23). It is possible to conclude from the total number data, in conjunction with the body weight data, of the current study that moderate hypothermia for 6 h has no adverse effects on the normal development of rat pups and is a safe potential therapy for striatal medium-spiny neurons.
A lack of effect on normal striatal development was only evident when the absolute number of medium-spiny neurons in the right striatum was compared (Table 1, Fig. 2). When the total volume and neuronal density was compared between the sham-ligated groups, an effect of moderate hypothermia seemed apparent (Table 1, Fig. 2). Yet, the total volume and neuronal density are indirect measures of neuronal survival that can be affected by tissue processing. Comparison of the absolute number of surviving neurons is preferable since this measure was shrinkage-independent. These results highlight that neuronal density and total volume data, in the absence of absolute neuronal number data, can be misleading (9,11,12).
During the 6 h of normothermia, the average rectal temperature of approximately 34.5°C was 2.5°C cooler than the average “normothermic” rectal temperature reported in earlier studies (7,24). Thus, the present study may represent mild hypothermia in the normothermic group. However, the normal rectal temperature of a rat pup can be as low as 33°C (25). Previous studies using 37°C as “normothermia” may have exposed their normothermic pups to hyperthermia, which may exacerbate the H/I brain injury (26).
Others have investigated the effects of 24–26 h of moderate hypothermia, either as immediate posttreatment or after a 2-h delay, on H/I PN7 rat brain injury (27–29). These studies have yielded promising results, including decreased brain edema, decreased brain lesion volume and decreased spatial memory deficits in the moderately hypothermic animals (27,28). Yet, during the 24–26 h of hypothermia the pups were housed either with the dam (27,28), or group-housed (29), which limits its clinical relevance.
An alternative strategy to prolonging the duration of hypothermia is the use of combinatorial treatments. A multifocal approach using antioxidants, caspase inhibitors or growth factors in combination with moderate hypothermia may prove to be effective. Recent preliminary evidence supports this contention. Combined treatment with moderate hypothermia for 6 h and an antioxidant is significantly neuroprotective for striatal medium-spiny neurons at PN14, after an H/I brain injury at PN8 (30).
A reduction in the absolute number of striatal medium-spiny neurons would be clinically insignificant unless it correlated with a significant impairment in long-term functioning. In a preliminary study (30), a significant decrease in the absolute number of medium-spiny neurons in the right striatum at PN14, and at 11 wk, after neonatal H/I injury correlated with a statistically significant reduction in left forepaw ability on the staircase test at 9–11 wk. The investigation of potential treatments to rescue striatal medium-spiny neurons is therefore likely to be of functional and clinical significance.
Two key findings from this study are that 6 h of mild hypothermia, or moderate hypothermia, immediately postinsult is not neuroprotective for striatal medium-spiny neurons after perinatal hypoxia-ischemia. A third key finding is that 6 h of moderate hypothermia after sham ligation and hypoxia has no effect on the absolute number of medium-spiny neurons in the normal striatum. Further stereological studies are now needed on treatment of individual rats with hypothermia alone for longer than 6 h, and on combinatorial treatments involving hypothermia, to reveal the optimal striatal neuroprotective strategy that treatment with hypothermia can achieve after perinatal hypoxia-ischemia.
Abbreviations
- CE:
-
coefficient of error
- H/I:
-
hypoxic/ischemic
- N:
-
total (i.e. absolute) number of neurons
- NV:
-
neuronal density
- PN7:
-
postnatal day 7
- PN14:
-
postnatal day 14
- Vref:
-
total reference volume
References
Berger R, Garnier Y 1999 Pathophysiology of perinatal brain damage. Brain Res Brain Res Rev 30: 107–134
Eicher DJ, Wagner CL, Katikaneni LP, Hulsey TC, Bass WT, Kaufman DA, Horgan MJ, Languani S, Bhatia JJ, Givelichian LM, Sankaran K, Yager JY 2005 Moderate hypothermia in neonatal encephalopathy: efficacy outcomes. Pediatr Neurol 32: 11–17
Shankaran S, Laptook AR, Ehrenkranz RA, Tyson JE, McDonald SA, Donovan EF, Fanaroff AA, Poole WK, Wright LL, Higgins RD, Finer NN, Waldemar AC, Duara S, Oh W, Cotten CM, Stevenson DK, Stoll BJ, Lemons JA, Guillet R, Jobe AH 2005 Whole body hypothermia for neonates with hypoxic-ischemic encephalopathy. N Engl J Med 353: 1574–1584
O'Brien FE, Iwata O, Thornton JS, De Vita E, Sellwood MW, Iwata S, Sakata YS, Charman S, Ordridge R, Cady EB, Wyatt JS, Robertson NJ 2006 Delayed whole-body cooling to 33 or 35°C and the development of impaired energy generation consequential to transient cerebral hypoxia-ischemia in the newborn piglet. Pediatrics 117: 1549–1559
Trescher WH, Ishiwa S, Johnston MV 1997 Brief post-hypoxic-ischemic hypothermia markedly delays neonatal brain injury. Brain Dev 19: 326–338
Thoresen M, Bagenholm R, Loberg EM, Apricena F, Kjellmer I 1996 Posthypoxic cooling of neonatal rats provides protection against brain injury. Arch Dis Child Fetal Neonatal Ed 74: F3–F9
Bona E, Hagberg H, Loberg EM, Bagenholm R, Thoresen M 1998 Protective effects of moderate hypothermia after neonatal hypoxia-ischemia: short- and long-term outcome. Pediatr Res 43: 738–745
Rice JE, Vannucci RC, Brierley JB 1981 The influence of immaturity on hypoxic-ischemic brain damage in the rat. Ann Neurol 9: 131–141
Galvin KA, Oorschot DE 1998 Postinjury magnesium sulfate treatment is not markedly neuroprotective for striatal medium spiny neurons after perinatal hypoxia/ischemia in the rat. Pediatr Res 44: 740–745
Volpe JJ 1992 Perinatal hypoxic-ischemic brain injury: overview. In: Fukuyama Y, Suzuki Y, Kamoshita S, Casaer P (eds) Fetal and Perinatal Neurology. Karger, Tokyo, pp 232–252
Covey MV, Murphy MP, Hobbs CE, Smith RA, Oorschot DE 2006 Effect of the mitochondrial antioxidant, Mito Vitamin E, on hypoxic-ischemic striatal injury in neonatal rats: a dose-response and stereological study. Exp Neurol 199: 513–519
Galvin KA, Oorschot DE 2003 Continuous low-dose treatment with brain-derived neurotrophic factor or neurotrophin-3 protects striatal medium spiny neurons from mild neonatal hypoxia/ischemia: a stereological study. Neuroscience 118: 1023–1032
Laptook AR, Corbett RJ, Sterett R, Garcia D, Tollefsbol G 1995 Quantitative relationship between brain temperature and energy utilization rate measured in vivo using 31P and 1H magnetic resonance spectroscopy. Pediatr Res 38: 919–925
Laptook AR, Shalak L, Corbett RJ 2001 Differences in brain temperature and cerebral blood flow during selective head versus whole-body cooling. Pediatrics 108: 1103–1110
Cochran WG, Cox GM 1992 Experimental Designs. John Wiley & Sons, New York, pp. 17–23
Oorschot DE Total number of neurons in the neostriatal, pallidal, subthalamic, and substantia nigral nuclei of the rat basal ganglia: a stereological study using the Cavalieri and optical disector methods. J Comp Neurol 366:580-599 Erratum: 1998 J Comp N. eurol 396: 556
Gundersen HJ, Jensen EB 1987 The efficiency of systematic sampling in stereology and its prediction. J Microsc 147: 229–263
Gundersen HJ 1986 Stereology of arbitrary particles. A review of unbiased number and size estimators and the presentation of some new ones, in memory of William R. Thompson. J Microsc 143: 3–45
Gundersen HJ, Jensen EB, Kieu K, Nielsen J 1999 The efficiency of systematic sampling in stereology-reconsidered. J Microsc 193: 199–211
Thoresen M, Satas S, Loberg EM, Whitelaw A, Acolet D, Lindgren C, Penrice J, Robertson N, Haug E, Steen PA 2001 Twenty-four hours of mild hypothermia in unsedated newborn pigs starting after a severe global hypoxic-ischemic insult is not neuroprotective. Pediatr Res 50: 405–411
Ma D, Hossain M, Chow A, Arshad M, Battson RM, Sanders RD, Mehmet H, Edwards AD, Franks NP, Maze M 2005 Xenon and hypothermia combine to provide neuroprotection from neonatal asphyxia. Ann Neurol 58: 182–193
Zhu C, Wang X, Cheng X, Qiu L, Xu F, Simbruner G, Blomgren K 2004 Post-ischemic hypothermia-induced tissue protection and diminished apoptosis after neonatal cerebral hypoxia-ischemia. Brain Res 996: 67–75
Yang X-F, Kennedy BR, Lomber SG, Schmidt RE, Rothman SM 2006 Cooling produces minimal neuropathology in neocortex and hippocampus. Neurobiol Dis 23: 637–643
Busto R, Dietrich WD, Globus MY, Valdes I, Scheinberg P, Ginsberg MD 1987 Small differences in intraischemic brain temperature critically determine the extent of ischemic neuronal injury. J Cereb Blood Flow Metab 7: 729–738
Rogalska J, Caputa M, Wentowska K, Nowakowska A 2004 Stress-induced behaviour in juvenile rats: effects of neonatal asphyxia, body temperature and chelation of iron. Behav Brain Res 154: 321–329
Mishima K, Ikeda T, Yoshikawa T, Aoo N, Egashira N, Xia YX, Ikenoue T, Iwasaki K, Fujiwara M 2004 Effects of hypothermia and hyperthermia on attentional and spatial learning deficits following neonatal hypoxia-ischemic insult in rats. Behav Brain Res 151: 209–217
Nedelcu J, Klein MA, Aguzzi A, Martin E 2000 Resuscitative hypothermia protects the neonatal rat brain from hypoxic-ischemic injury. Brain Pathol 10: 61–71
Wagner BP, Nedelcu J, Martin E 2002 Delayed postischemic hypothermia improves long-term behavioral outcome after cerebral hypoxia-ischemia in neonatal rats. Pediatr Res 51: 354–360
Tomimatsu T, Fukuda H, Endoh M, Mu J, Kanagawa T, Hosono T, Kanzaki T, Doi K, Kubo T, Murata Y 2003 Long-term neuroprotective effects of hypothermia on neonatal hypoxic-ischemic brain injury in rats, assessed by auditory brainstem response. Pediatr Res 53: 57–61
Hobbs C, Oorschot D 2004 Long-term rescue of striatal neurons and motor skills by combined antioxidant-hypothermia treatment after neonatal hypoxic-ischemic brain injury in the rat. N Z Med J 117: 3. Retrieved from http://www.nzma.org.nz/journal/117-1202/1086/content.pdf. Accessed on January 16 2007.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the University of Otago in the form of a PhD Postgraduate Scholarship to MVC.
Rights and permissions
About this article
Cite this article
Covey, M., Oorschot, D. Effect of Hypothermic Post-Treatment on Hypoxic-Ischemic Striatal Injury, and Normal Striatal Development, in Neonatal Rats: A Stereological Study. Pediatr Res 62, 646–651 (2007). https://doi.org/10.1203/PDR.0b013e318157d1fe
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/PDR.0b013e318157d1fe
This article is cited by
-
Hypothermia combined with extracellular vesicles from clonally expanded immortalized mesenchymal stromal cells improves neurodevelopmental impairment in neonatal hypoxic-ischemic brain injury
Journal of Neuroinflammation (2023)
-
Treatment temperature and insult severity influence the neuroprotective effects of therapeutic hypothermia
Scientific Reports (2016)
-
Effects of combination therapy using hypothermia and erythropoietin in a rat model of neonatal hypoxia–ischemia
Pediatric Research (2013)