Abstract
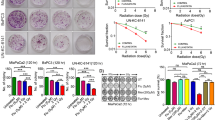
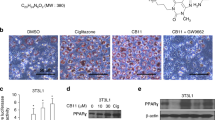
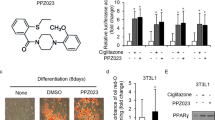
Radiotherapy is emerging as an important modality for the local control of pancreatic cancer, but pancreatic cancer cell radioresistance remains a serious concern. Peroxisome proliferator-activated receptor α (PPARα) is a member of the PPAR nuclear hormone receptor superfamily, which can be activated by fibrate ligands. The clinical relevance of PPARα and its biological function in pancreatic cancer radiosensitivity have not been previously described. In this study, we examined PPARα expression in tissue samples of pancreatic cancer patients. We found significantly higher expression of PPARα in pancreatic cancer tissues than in tumor-adjacent tissues and that the PPARα expression level is inversely associated with higher overall patient survival rate. We further observed that PPARα activation by its agonist clofibrate sensitizes pancreatic cancer cells to radiation by modulating cell cycle progression and apoptosis in several pancreatic cancer cell lines. Small interfering RNA-mediated PPARα silencing and PPARα blockade by the antagonist GW6471 abolish the effect of clofibrate on radiosensitization. An in vivo study showed that PANC1 xenografts treated with clofibrate are more sensitive to radiation than untreated xenografts. mRNA profiling by microarray analysis revealed that the expression of PTPRZ1 and Wnt8a, two core components of the β-catenin pathway, is downregulated by clofibrate. Chromatin immunoprecipitation analysis confirmed that clofibrate abrogates the binding of nuclear factor-κB to the PTPRZ1 and Wnt8a promoters, ultimately decreasing Wnt/β-catenin signaling activity, which is associated with radiosensitivity. Overall, we demonstrate that PPARα is overexpressed in pancreatic cancer tissues and clofibrate-mediated PPARα activation sensitizes pancreatic cancer cells to radiation through the Wnt/β-catenin pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F et al. Cancer statistics in China, 2015. CA Cancer J Clin 2016; 66: 115–132.
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 2016; 66: 271–289.
Midha S, Chawla S, Garg PK . Modifiable and non-modifiable risk factors for pancreatic cancer: a review. Cancer Lett 2016; 381: 269–277.
Polireddy K, Chen Q . Cancer of the pancreas: molecular pathways and current advancement in treatment. J Cancer 2016; 7: 1497–1514.
Changela K, Patil R . Endoscopic ultrasound-guided radiofrequency ablation of the pancreatic tumors: a promising tool in management of pancreatic tumors. Can J Gastroenterol Hepatol 2016; 2016: 4189358.
Hajj C, Goodman KA . Pancreatic cancer and SBRT: a new potential option? Rep Pract Oncol Radiother 2015; 20: 377–384.
Goodman KA . Stereotactic body radiation therapy for pancreatic cancer. Cancer J 2016; 22: 290–295.
Hoffe S, Rao N, Shridhar R . Neoadjuvant vs adjuvant therapy for resectable pancreatic cancer: the evolving role of radiation. Semin Radiat Oncol 2014; 24: 113–125.
Pisters PW, Wolff RA, Janjan NA, Cleary KR, Charnsangavej C, Crane CN et al. Preoperative paclitaxel and concurrent rapid-fractionation radiation for resectable pancreatic adenocarcinoma: toxicities, histologic response rates, and event-free outcome. J Clin Oncol 2002; 20: 2537–2544.
Evans DB, Varadhachary GR, Crane CH, Sun CC, Lee JE, Pisters PW et al. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J Clin Oncol 2008; 26: 3496–3502.
Mandard S, Muller M, Kersten S . Peroxisome proliferator-activated receptor alpha target genes. Cell Mol Life Sci 2004; 61: 393–416.
Retraction notice to ‘Peroxisome proliferator-activated receptor alpha controls hepatic heme biosynthesis through ALAS1’ J Mol Biol 2010; 388: 225–238. J Mol Biol 2010; 404: 172.
Kostapanos MS, Kei A, Elisaf MS . Current role of fenofibrate in the prevention and management of non-alcoholic fatty liver disease. World J Hepatol 2013; 5: 470–478.
Wright MB, Bortolini M, Tadayyon M, Bopst M . Minireview: Challenges and opportunities in development of PPAR agonists. Mol Endocrinol 2014; 28: 1756–1768.
Gao J, Yuan S, Jin J, Shi J, Hou Y . PPARalpha regulates tumor progression, foe or friend? Eur J Pharmacol 2015; 765: 560–564.
Wysowski DK, Kennedy DL, Gross TP . Prescribed use of cholesterol-lowering drugs in the United States, 1978 through 1988. JAMA 1990; 263: 2185–2188.
Maggiora M, Oraldi M, Muzio G, Canuto RA . Involvement of PPARalpha and PPARgamma in apoptosis and proliferation of human hepatocarcinoma HepG2 cells. Cell Biochem Funct 2010; 28: 571–577.
Zhou J, Zhang S, Xue J, Avery J, Wu J, Lind SE et al. Activation of peroxisome proliferator-activated receptor alpha (PPARalpha) suppresses hypoxia-inducible factor-1alpha (HIF-1alpha) signaling in cancer cells. J Biol Chem 2012; 287: 35161–35169.
Bell D, Berchuck A, Birrer M, Chien J, Cramer D, Dao F et al. Integrated genomic analyses of ovarian carcinoma. Nature 2011; 474: 609–615.
Urbanska K, Pannizzo P, Grabacka M, Croul S, Del Valle L, Khalili K et al. Activation of PPARalpha inhibits IGF-I-mediated growth and survival responses in medulloblastoma cell lines. Int J Cancer 2008; 123: 1015–1024.
Coleman TR, Dunphy WG . Cdc2 regulatory factors. Curr Opin Cell Biol 1994; 6: 877–882.
Green DR, Kroemer G . The pathophysiology of mitochondrial cell death. Science 2004; 305: 626–629.
Liu YT, Shang D, Akatsuka S, Ohara H, Dutta KK, Mizushima K et al. Chronic oxidative stress causes amplification and overexpression of ptprz1 protein tyrosine phosphatase to activate beta-catenin pathway. Am J Pathol 2007; 171: 1978–1988.
Shang D, Xu X, Wang D, Li Y, Liu Y . Protein tyrosine phosphatase zeta enhances proliferation by increasing beta-catenin nuclear expression in VHL-inactive human renal cell carcinoma cells. World J Urol 2013; 31: 1547–1554.
Saitoh T, Mine T, Katoh M . Expression and regulation of WNT8A and WNT8B mRNAs in human tumor cell lines: up-regulation of WNT8B mRNA by beta-estradiol in MCF-7 cells, and down-regulation of WNT8A and WNT8B mRNAs by retinoic acid in NT2 cells. Int J Oncol 2002; 20: 999–1003.
Cunningham TJ, Kumar S, Yamaguchi TP, Duester G . Wnt8a and Wnt3a cooperate in the axial stem cell niche to promote mammalian body axis extension. Dev Dyn 2015; 244: 797–807.
Lucero OM, Dawson DW, Moon RT, Chien AJ . A re-evaluation of the ‘oncogenic’ nature of Wnt/beta-catenin signaling in melanoma and other cancers. Curr Oncol Rep 2010; 12: 314–318.
Karim R, Tse G, Putti T, Scolyer R, Lee S . The significance of the Wnt pathway in the pathology of human cancers. Pathology 2004; 36: 120–128.
Chen J, Li J, Miao Z, Xu X, Liu CF . XAV939, a small molecular inhibitor, provides neuroprotective effects on oligodentrocytes. J Neurosci Res 2014; 92: 1252–1258.
Afifi MM, Austin LA, Mackey MA, El-Sayed MA . XAV939: from a small inhibitor to a potent drug bioconjugate when delivered by gold nanoparticles. Bioconjug Chem 2014; 25: 207–215.
Chandran K, Goswami S, Sharma-Walia N . Implications of a peroxisome proliferator-activated receptor alpha (PPARalpha) ligand clofibrate in breast cancer. Oncotarget 2016; 7: 15577–15599.
Chen H, Shen ZY, Xu W, Fan TY, Li J, Lu YF et al. Expression of P450 and nuclear receptors in normal and end-stage Chinese livers. World J Gastroenterol 2014; 20: 8681–8690.
Segawa Y, Yoshimura R, Hase T, Nakatani T, Wada S, Kawahito Y et al. Expression of peroxisome proliferator-activated receptor (PPAR) in human prostate cancer. Prostate 2002; 51: 108–116.
Misra P, Reddy JK . Peroxisome proliferator-activated receptor-alpha activation and excess energy burning in hepatocarcinogenesis. Biochimie 2014; 98: 63–74.
Yamazaki K, Shimizu M, Okuno M, Matsushima-Nishiwaki R, Kanemura N, Araki H et al. Synergistic effects of RXR alpha and PPAR gamma ligands to inhibit growth in human colon cancer cells—phosphorylated RXR alpha is a critical target for colon cancer management. Gut 2007; 56: 1557–1563.
Skrypnyk N, Chen X, Hu W, Su Y, Mont S, Yang S et al. PPARalpha activation can help prevent and treat non-small cell lung cancer. Cancer Res 2014; 74: 621–631.
Yokoyama Y, Xin B, Shigeto T, Umemoto M, Kasai-Sakamoto A, Futagami M et al. Clofibric acid, a peroxisome proliferator-activated receptor alpha ligand, inhibits growth of human ovarian cancer. Mol Cancer Ther 2007; 6: 1379–1386.
Ge Y, Liu J, Yang X, Zhu H, Yang B, Zhao K et al. Fenofibrate enhances radiosensitivity of esophageal squamous cell carcinoma by suppressing hypoxia-inducible factor-1alpha expression. Tumour Biol 2014; 35: 10765–10771.
Li XQ, Zhou JD, Zou ST, Yu J, Meng XJ, Wu JC . Enhancement of radiosensitivity in human esophageal carcinoma cells by fenofibrate and its potential mechanism. Tumori 2015; 101: 123–130.
Lin H, Yu CH, Jen CY, Cheng CF, Chou Y, Chang CC et al. Adiponectin-mediated heme oxygenase-1 induction protects against iron-induced liver injury via a PPARalpha dependent mechanism. Am J Pathol 2010; 177: 1697–1709.
Yin WH, Chen JW, Chen YH, Lin SJ . Fenofibrate modulates HO-1 and ameliorates endothelial expression of cell adhesion molecules in systolic heart failure. Acta Cardiol Sin 2013; 29: 251–260.
Han D, Wei W, Chen X, Zhang Y, Wang Y, Zhang J et al. NF-kappaB/RelA-PKM2 mediates inhibition of glycolysis by fenofibrate in glioblastoma cells. Oncotarget 2015; 6: 26119–26128.
Zhao W, Iskandar S, Kooshki M, Sharpe JG, Payne V, Robbins ME . Knocking out peroxisome proliferator-activated receptor (PPAR) alpha inhibits radiation-induced apoptosis in the mouse kidney through activation of NF-kappaB and increased expression of IAPs. Radiat Res 2007; 167: 581–591.
Clevers H . Wnt/beta-catenin signaling in development and disease. Cell 2006; 127: 469–480.
Yang W, Yu H, Shen Y, Liu Y, Yang Z, Sun T . MiR-146b-5p overexpression attenuates stemness and radioresistance of glioma stem cells by targeting HuR/lincRNA-p21/beta-catenin pathway. Oncotarget 2016; 7: 41505–41526.
Zhang Q, Gao M, Luo G, Han X, Bao W, Cheng Y et al. Enhancement of radiation sensitivity in lung cancer cells by a novel small molecule inhibitor that targets the beta-catenin/Tcf4 interaction. PLoS ONE 2016; 11: e0152407.
Cheng R, Ding L, He X, Takahashi Y, Ma JX . Interaction of PPARalpha with the canonic Wnt pathway in the regulation of renal fibrosis. Diabetes 2016; 65: 3730–3743.
Luo J, Zhou X, Ge X, Liu P, Cao J, Lu X et al. Upregulation of Ying Yang 1 (YY1) suppresses esophageal squamous cell carcinoma development through heme oxygenase-1. Cancer Sci 2013; 104: 1544–1551.
Tang Y, Geng Y, Luo J, Shen W, Zhu W, Meng C et al. Downregulation of ubiquitin inhibits the proliferation and radioresistance of non-small cell lung cancer cells in vitro and in vivo. Sci Rep 2015; 5: 9476.
Zhang S, Hao J, Xie F, Hu X, Liu C, Tong J et al. Downregulation of miR-132 by promoter methylation contributes to pancreatic cancer development. Carcinogenesis 2011; 32: 1183–1189.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81522039, 81573080, 81673100 and 31670857), the Key Scientific Development Program of China (2016YFC0904700 and 2016YFC0904702), the Suzhou Key Medical Center (SZZX201506) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Xue, J., Zhu, W., Song, J. et al. Activation of PPARα by clofibrate sensitizes pancreatic cancer cells to radiation through the Wnt/β-catenin pathway. Oncogene 37, 953–962 (2018). https://doi.org/10.1038/onc.2017.401
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2017.401
This article is cited by
-
ACOX2 is a prognostic marker and impedes the progression of hepatocellular carcinoma via PPARα pathway
Cell Death & Disease (2021)
-
Exosomes overexpressing miR-34c inhibit malignant behavior and reverse the radioresistance of nasopharyngeal carcinoma
Journal of Translational Medicine (2020)
-
CLPTM1L induces estrogen receptor β signaling-mediated radioresistance in non-small cell lung cancer cells
Cell Communication and Signaling (2020)
-
B7-H3 regulates KIF15-activated ERK1/2 pathway and contributes to radioresistance in colorectal cancer
Cell Death & Disease (2020)
-
CD59 is a potential biomarker of esophageal squamous cell carcinoma radioresistance by affecting DNA repair
Cell Death & Disease (2018)