Abstract
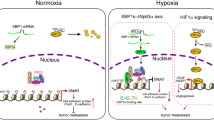
Hypoxia is a driver of cell movement in processes such as development and tumor progression. The cellular response to hypoxia involves a transcriptional program mediated by hypoxia-inducible factors, but translational control has emerged as a significant contributor. In this study, we demonstrate that a cell–cell adhesion molecule, cadherin-22, is upregulated in hypoxia via mTORC1-independent translational control by the initiation factor eIF4E2. We identify new functions of cadherin-22 as a hypoxia-specific cell-surface molecule involved in cancer cell migration, invasion and adhesion. Silencing eIF4E2 or cadherin-22 significantly impaired MDA-MB-231 breast carcinoma and U87MG glioblastoma cell migration and invasion only in hypoxia, while reintroduction of the respective exogenous gene restored the normal phenotype. Cadherin-22 was evenly distributed throughout spheroids and required for their formation and support of a hypoxic core. Conversely, E-cadherin translation was repressed by hypoxia and only expressed in the oxygenated cells of U87MG spheroids. Furthermore, immunofluorescence on paraffin-embedded human tissue from 40 glioma and 40 invasive ductal breast carcinoma patient specimens revealed that cadherin-22 expression colocalized with areas of hypoxia and significantly correlated with tumor grade and progression-free survival or stage and tumor size, respectively. This study broadens our understanding of tumor progression and metastasis by highlighting cadherin-22 as a potential new target of cancer therapy to disable hypoxic cancer cell motility and adhesion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Gebauer F, Hentze MW . Molecular mechanisms of translational control. Nat Rev Mol Cell Biol 2004; 5: 827–835.
Lin TA, Kong X, Haystead TA, Pause A, Belsham G, Sonenberg N et al. PHAS-I as a link between mitogen-activated protein kinase and translation initiation. Science 1994; 266: 653–656.
Uniacke J, Holterman CE, Lachance G, Franovic A, Jacob MD, Fabian MR et al. An oxygen-regulated switch in the protein synthesis machinery. Nature 2012; 486: 126–129.
Ho JJ, Wang M, Audas TE, Kwon D, Carlsson SK, Timpano S et al. Systemic reprogramming of translation efficiencies on oxygen stimulus. Cell Rep 2016; 14: 1293–1300.
Rom E, Kim HC, Gingras AC, Marcotrigiano J, Favre D, Olsen H et al. Cloning and characterization of 4EHP, a novel mammalian eIF4E-related cap-binding protein. J Biol Chem 1998; 273: 13104–13109.
Uniacke J, Perera JK, Lachance G, Francisco CB, Lee S . Cancer cells exploit eIF4E2-directed synthesis of hypoxia response proteins to drive tumor progression. Cancer Res 2014; 74: 1379–1389.
Ramaswamy S, Ross KN, Lander ES, Golub TR . A molecular signature of metastasis in primary solid tumors. Nat Genet 2003; 33: 49–54.
Timpano S, Uniacke J . Human cells cultured under physiological oxygen utilize two cap-binding proteins to recruit distinct mRNAs for translation. J Biol Chem 2016; 291: 10772–10782.
Gumbiner BM . Cell adhesion: the molecular basis of tissue architecture and morphogenesis. Cell 1996; 84: 345–357.
Christofori G, Semb H . The role of the cell-adhesion molecule E-cadherin as a tumour-suppressor gene. Trends Biochem Sci 1999; 24: 73–76.
Jeanes A, Gottardi CJ, Yap AS . Cadherins and cancer: how does cadherin dysfunction promote tumor progression? Oncogene 2008; 27: 6920–6929.
De Leeuw WJ, Berx G, Vos CB, Peterse JL, Van de Vijver MJ, Litvinov S et al. Simultaneous loss of E-cadherin and catenins in invasive lobular breast cancer and lobular carcinoma in situ. J Pathol 1997; 183: 404–411.
Guilford P, Hopkins J, Harraway J, McLeod M, McLeod N, Harawira P et al. E-cadherin germline mutations in familial gastric cancer. Nature 1998; 392: 402–405.
Sawada K, Mitra AK, Radjabi AR, Bhaskar V, Kistner EO, Tretiakova M et al. Loss of E-cadherin promotes ovarian cancer metastasis via alpha 5-integrin, which is a therapeutic target. Cancer Res 2008; 68: 2329–2339.
Batlle E, Sancho E, Franci C, Dominguez D, Monfar M, Baulida J et al. The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat Cell Biol 2000; 2: 84–89.
Comijn J, Berx G, Vermassen P, Verschueren K, van Grunsven L, Bruyneel E et al. The two-handed E box binding zinc finger protein SIP1 downregulates E-cadherin and induces invasion. Mol Cell 2001; 7: 1267–1278.
Lombaerts M, van Wezel T, Philippo K, Dierssen JW, Zimmerman RM, Oosting J et al. E-cadherin transcriptional downregulation by promoter methylation but not mutation is related to epithelial-to-mesenchymal transition in breast cancer cell lines. Br J Cancer 2006; 94: 661–671.
Imai T, Horiuchi A, Wang C, Oka K, Ohira S, Nikaido T et al. Hypoxia attenuates the expression of E-cadherin via up-regulation of SNAIL in ovarian carcinoma cells. Am J Pathol 2003; 163: 1437–1447.
Krishnamachary B, Zagzag D, Nagasawa H, Rainey K, Okuyama H, Baek JH et al. Hypoxia-inducible factor-1-dependent repression of E-cadherin in von Hippel-Lindau tumor suppressor-null renal cell carcinoma mediated by TCF3, ZFHX1A, and ZFHX1B. Cancer Res 2006; 66: 2725–2731.
Wheelock MJ, Shintani Y, Maeda M, Fukumoto Y, Johnson KR . Cadherin switching. J Cell Sci 2008; 121 (Pt 6): 727–735.
Sugimoto K, Honda S, Yamamoto T, Ueki T, Monden M, Kaji A et al. Molecular cloning and characterization of a newly identified member of the cadherin family, PB-cadherin. J Biol Chem 1996; 271: 11548–11556.
Wu J, Jester WF Jr, Laslett AL, Meinhardt A, Orth JM . Expression of a novel factor, short-type PB-cadherin, in Sertoli cells and spermatogenic stem cells of the neonatal rat testis. J Endocrinol 2003; 176: 381–391.
Zhou J, Li J, Chen J, Liu Y, Gao W, Ding Y . Over-expression of CDH22 is associated with tumor progression in colorectal cancer. Tumour Biol 2009; 30: 130–140.
Indovina P, Rainaldi G, Santini MT . Hypoxia increases adhesion and spreading of MG-63 three-dimensional tumor spheroids. Anticancer Res 2008; 28 (2A): 1013–1022.
Zhong H, De Marzo AM, Laughner E, Lim M, Hilton DA, Zagzag D et al. Overexpression of hypoxia-inducible factor 1alpha in common human cancers and their metastases. Cancer Res 1999; 59: 5830–5835.
Zhu Y, Yang P, Wang Q, Hu J, Xue J, Li G et al. The effect of CXCR4 silencing on epithelial-mesenchymal transition related genes in glioma U87 cells. Anat Rec (Hoboken) 2013; 296: 1850–1856.
Franko AJ, Sutherland RM . Oxygen diffusion distance and development of necrosis in multicell spheroids. Radiat Res 1979; 79: 439–453.
Tee AR, Tee JA, Blenis J . Characterizing the interaction of the mammalian eIF4E-related protein 4EHP with 4E-BP1. FEBS Lett 2004; 564: 58–62.
Chen J, Imanaka N, Chen J, Griffin JD . Hypoxia potentiates Notch signaling in breast cancer leading to decreased E-cadherin expression and increased cell migration and invasion. Br J Cancer 2010; 102: 351–360.
Krishnamachary B, Berg-Dixon S, Kelly B, Agani F, Feldser D, Ferreira G et al. Regulation of colon carcinoma cell invasion by hypoxia-inducible factor 1. Cancer Res 2003; 63: 1138–1143.
Zagzag D, Lukyanov Y, Lan L, Ali MA, Esencay M, Mendez O et al. Hypoxia-inducible factor 1 and VEGF upregulate CXCR4 in glioblastoma: implications for angiogenesis and glioma cell invasion. Lab Invest 2006; 86: 1221–1232.
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Labernadie A, Kato T, Brugues A, Serra-Picamal X, Derzsi S, Arwert E et al. A mechanically active heterotypic E-cadherin/N-cadherin adhesion enables fibroblasts to drive cancer cell invasion. Nat Cell Biol 2017; 19: 224–237.
Friedl P, Gilmour D . Collective cell migration in morphogenesis, regeneration and cancer. Nat Rev Mol Cell Biol 2009; 10: 445–457.
Friedl P, Locker J, Sahai E, Segall JE . Classifying collective cancer cell invasion. Nat Cell Biol 2012; 14: 777–783.
Rorth P . Collective cell migration. Annu Rev Cell Dev Biol 2009; 25: 407–429.
Giampieri S, Manning C, Hooper S, Jones L, Hill CS, Sahai E . Localized and reversible TGFbeta signalling switches breast cancer cells from cohesive to single cell motility. Nat Cell Biol 2009; 11: 1287–1296.
Kitamura T, Kometani K, Hashida H, Matsunaga A, Miyoshi H, Hosogi H et al. SMAD4-deficient intestinal tumors recruit CCR1+ myeloid cells that promote invasion. Nat Genet 2007; 39: 467–475.
Wolf K, Wu YI, Liu Y, Geiger J, Tam E, Overall C et al. Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nat Cell Biol 2007; 9: 893–904.
Hidalgo-Carcedo C, Hooper S, Chaudhry SI, Williamson P, Harrington K, Leitinger B et al. Collective cell migration requires suppression of actomyosin at cell-cell contacts mediated by DDR1 and the cell polarity regulators Par3 and Par6. Nat Cell Biol 2011; 13: 49–58.
Martinez-Gonzalez A, Calvo GF, Perez Romasanta LA, Perez-Garcia VM . Hypoxic cell waves around necrotic cores in glioblastoma: a biomathematical model and its therapeutic implications. Bull Math Biol 2012; 74: 2875–2896.
Rong Y, Durden DL, Van Meir EG, Brat DJ . 'Pseudopalisading' necrosis in glioblastoma: a familiar morphologic feature that links vascular pathology, hypoxia, and angiogenesis. J Neuropathol Exp Neurol 2006; 65: 529–539.
Holmquist-Mengelbier L, Fredlund E, Lofstedt T, Noguera R, Navarro S, Nilsson H et al. Recruitment of HIF-1alpha and HIF-2alpha to common target genes is differentially regulated in neuroblastoma: HIF-2alpha promotes an aggressive phenotype. Cancer Cell 2006; 10: 413–423.
Grabmaier K, AdW MC, Verhaegh GW, Schalken JA, Oosterwijk E . Strict regulation of CAIX(G250/MN) by HIF-1alpha in clear cell renal cell carcinoma. Oncogene 2004; 23: 5624–5631.
Espina V, Wysolmerski J, Edmiston K, Liotta LA . Attacking breast cancer at the preinvasion stage by targeting autophagy. Womens Health (Lond) 2013; 9: 157–170.
Feng M, Chen JY, Weissman-Tsukamoto R, Volkmer JP, Ho PY, McKenna KM et al. Macrophages eat cancer cells using their own calreticulin as a guide: roles of TLR and Btk. Proc Natl Acad Sci USA 2015; 112: 2145–2150.
Piche B, Khosravi S, Martinka M, Ho V, Li G . CDH22 expression is reduced in metastatic melanoma. Am J Cancer Res 2011; 1: 233–239.
Bhat M, Robichaud N, Hulea L, Sonenberg N, Pelletier J, Topisirovic I . Targeting the translation machinery in cancer. Nat Rev Drug Discov 2015; 14: 261–278.
Kuehn BM . Genomics illuminates a deadly brain cancer. JAMA 2010; 303: 925–927.
Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 1989; 244: 707–712.
Efeyan A, Sabatini DM . mTOR and cancer: many loops in one pathway. Curr Opin Cell Biol 2010; 22: 169–176.
Lundgren K, Nordenskjold B, Landberg G . Hypoxia, Snail and incomplete epithelial-mesenchymal transition in breast cancer. Br J Cancer 2009; 101: 1769–1781.
Hwang-Verslues WW, Chang PH, Wei PC, Yang CY, Huang CK, Kuo WH et al. miR-495 is upregulated by E12/E47 in breast cancer stem cells, and promotes oncogenesis and hypoxia resistance via downregulation of E-cadherin and REDD1. Oncogene 2011; 30: 2463–2474.
Blaschuk OW, Devemy E . Cadherins as novel targets for anti-cancer therapy. Eur J Pharmacol 2009; 625: 195–198.
Beasley GM, McMahon N, Sanders G, Augustine CK, Selim MA, Peterson B et al. A phase 1 study of systemic ADH-1 in combination with melphalan via isolated limb infusion in patients with locally advanced in-transit malignant melanoma. Cancer 2009; 115: 4766–4774.
Perotti A, Sessa C, Mancuso A, Noberasco C, Cresta S, Locatelli A et al. Clinical and pharmacological phase I evaluation of Exherin (ADH-1), a selective anti-N-cadherin peptide in patients with N-cadherin-expressing solid tumours. Ann Oncol 2009; 20: 741–745.
Vinci M, Box C, Eccles SA . Three-dimensional (3D) tumor spheroid invasion assay. J Vis Exp 2015; 99: e52686.
Chaudary N, Hill RP . Increased expression of metastasis-related genes in hypoxic cells sorted from cervical and lymph nodal xenograft tumors. Lab Invest 2009; 89: 587–596.
Russell J, Carlin S, Burke SA, Wen B, Yang KM, Ling CC . Immunohistochemical detection of changes in tumor hypoxia. Int J Radiat Oncol Biol Phys 2009; 73: 1177–1186.
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T et al. Fiji: an open-source platform for biological-image analysis. Nat Methods 2012; 9: 676–682.
Acknowledgements
We thank Terry Van Raay for the use of his microscope, Scott Ryan and John Vessey for the use of their cryostat, and Marc Coppolino for technical advice and reagents. This work was funded by grants from the Canadian Institutes of Health Research (PJT 152925) and the Cancer Research Society to JU.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Kelly, N., Varga, J., Specker, E. et al. Hypoxia activates cadherin-22 synthesis via eIF4E2 to drive cancer cell migration, invasion and adhesion. Oncogene 37, 651–662 (2018). https://doi.org/10.1038/onc.2017.372
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2017.372