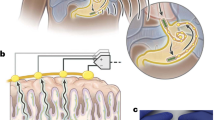
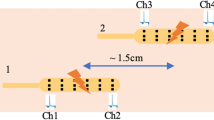
Abstract
Motility patterns of the gastrointestinal tract are important for efficient processing of nutrients and waste. Peristalsis and segmentation are based on rhythmic electrical slow waves that generate the phasic contractions fundamental to gastrointestinal motility. Slow waves are generated and propagated actively by interstitial cells of Cajal (ICC), and these events conduct to smooth muscle cells to elicit excitation–contraction coupling. Extracellular electrical recording has been utilized to characterize slow-wave generation and propagation and abnormalities that might be responsible for gastrointestinal motility disorders. Electrode array recording and digital processing are being used to generate data for models of electrical propagation in normal and pathophysiological conditions. Here, we discuss techniques of extracellular recording as applied to gastrointestinal organs and how mechanical artefacts might contaminate these recordings and confound their interpretation. Without rigorous controls for movement, current interpretations of extracellular recordings might ascribe inaccurate behaviours and electrical anomalies to ICC networks and gastrointestinal muscles, bringing into question the findings and validity of models of gastrointestinal electrophysiology developed from these recordings.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Alvarez, W. C. & Mahoney, L. J. Action currents in stomach and intestine. Am. J. Physiol. 58, 476–493 (1922).
Richter, C. P. Action currents from the stomach. Am. J. Physiol. 67, 612–633 (1924).
Janse, M. J. & Rosen, M. R. History of arrhythmias. Handb. Exp. Pharmacol. 171, 1–39 (2006).
O'Grady, G. et al. Abnormal initiation and conduction of slow-wave activity in gastroparesis, defined by high-resolution electrical mapping. Gastroenterology 143, 589–598 (2012).
Angeli, T. R. et al. Loss of interstitial cells of Cajal and patterns of gastric dysrhythmia in patients with chronic unexplained nausea and vomiting. Gastroenterology 149, 56–66.e5 (2015).
O'Grady, G. et al. Origin and propagation of human gastric slow-wave activity defined by high-resolution mapping. Am. J. Physiol. Gastrointest. Liver Physiol. 299, G585–G592 (2010).
Lammers, W. J. Normal and abnormal electrical propagation in the small intestine. Acta Physiol. (Oxf.) 213, 349–359 (2015).
Lammers, W. J., Ver Donck, L., Stephen, B., Smets, D. & Schuurkes, J. A. Origin and propagation of the slow wave in the canine stomach: the outlines of a gastric conduction system. Am. J. Physiol. Gastrointest. Liver Physiol. 296, G1200–G1210 (2009).
Nakayama, S. Frequency analysis may distinguish the effects of calcium antagonists on mechanical and electrical activity. Neurogastroenterol. Motil. 24, 397; author reply 398 (2012).
O'Grady, G. Gastrointestinal extracellular electrical recordings: fact or artifact? Neurogastroenterol. Motil. 24, 1–6 (2012).
O'Grady, G., Pullan, A. J. & Cheng, L. K. The analysis of human gastric pacemaker activity. J. Physiol. 590, 1299–1300; author reply 1301–1302 (2012).
O'Grady, G., Angeli, T., Du, P. & Cheng, L. K. Concerning the validity of gastrointestinal extracellular recordings. Physiol. Rev. 95, 691–692 (2015).
Sanders, K. M., Ward, S. M. & Koh, S. D. Reply to O'Grady et al. Physiol. Rev. 95, 693–694 (2015).
Chowdhury, R. H. et al. Surface electromyography signal processing and classification techniques. Sensors (Basel) 13, 12431–12466 (2013).
Bayguinov, O., Hennig, G. W. & Sanders, K. M. Movement based artifacts may contaminate extracellular electrical recordings from gastrointestinal muscles. Neurogastroenterol. Motil. 23, 1029–1042 (2011).
Park, K. J. et al. Spatial and temporal mapping of pacemaker activity in interstitial cells of Cajal in mouse ileum in situ. Am. J. Physiol. 290, C1411–C1427 (2006).
Szurszewski, J. H. in Physiology of the Gastrointestinal Tract (ed. Johnson, L. R.) 1435–1466 (Raven Press, 1981).
Sanders, K. M., Ward, S. M. & Koh, S. D. Interstitial cells: regulators of smooth muscle function. Physiol. Rev. 94, 859–907 (2014).
Vogalis, F., Publicover, N. G., Hume, J. R. & Sanders, K. M. Relationship between calcium current and cytosolic calcium in canine gastric smooth muscle cells. Am. J. Physiol. 260, C1012–C1018 (1991).
Ozaki, H., Stevens, R. J., Blondfield, D. P., Publicover, N. G. & Sanders, K. M. Simultaneous measurement of membrane potential, cytosolic Ca2+, and tension in intact smooth muscles. Am. J. Physiol. 260, C917–C925 (1991).
Sanders, K. M., Koh, S. D., Ro, S. & Ward, S. M. Regulation of gastrointestinal motility-insights from smooth muscle biology. Nat. Rev. Gastroenterol. Hepatol. 9, 633–645 (2012).
Farrugia, G. Interstitial cells of Cajal in health and disease. Neurogastroenterol. Motil. 20 (Suppl. 1), 54–63 (2008).
Koh, S. D., Ward, S. M. & Sanders, K. M. Ionic conductances regulating the excitability of colonic smooth muscles. Neurogastroenterol. Motil. 24, 705–718 (2012).
Bülbring, E. Smooth muscle potentials recorded in the taenia coli of the guineapig. J. Physiol. 123, 55P–56P (1954).
Dickens, E. J., Hirst, G. D. & Tomita, T. Identification of rhythmically active cells in guinea-pig stomach. J. Physiol. 514, 515–531 (1999).
Kito, Y., Kurahashi, M., Mitsui, R., Ward, S. M. & Sanders, K. M. Spontaneous transient hyperpolarizations in the rabbit small intestine. J. Physiol. 592, 4733–4745 (2014).
Kito, Y., Mitsui, R., Ward, S. M. & Sanders, K. M. Characterization of slow waves generated by myenteric interstitial cells of Cajal of the rabbit small intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 308, G378–G388 (2015).
Kito, Y. & Suzuki, H. Properties of pacemaker potentials recorded from myenteric interstitial cells of Cajal distributed in the mouse small intestine. J. Physiol. 553, 803–818 (2003).
Kito, Y., Ward, S. M. & Sanders, K. M. Pacemaker potentials generated by interstitial cells of Cajal in the murine intestine. Am. J. Physiol. Cell Physiol. 288, C710–C720 (2005).
Cousins, H. M., Edwards, F. R., Hickey, H., Hill, C. E. & Hirst, G. D. Electrical coupling between the myenteric interstitial cells of Cajal and adjacent muscle layers in the guinea-pig gastric antrum. J. Physiol. 550, 829–844 (2003).
Sanders, K. M., Stevens, R., Burke, E. & Ward, S. M. Slow waves actively propagate at submucosal surface of circular layer in canine colon. Am. J. Physiol. 259, G258–G263 (1990).
Bortoff, A. Configuration of intestinal slow waves obtained by monopolar recording techniques. Am. J. Physiol. 213, 157–162 (1967).
Hoffman, B. F., Cranefield, P. F., Lepeschkin, E., Surawicz, B. & Herrlich, H. C. Comparison of cardiac monophasic action potentials recorded by intracellular and suction electrodes. Am. J. Physiol. 196, 1297–1301 (1959).
Szurszewski, J. H. Mechanism of action of pentagastrin and acetylcholine on the longitudinal muscle of the canine antrum. J. Physiol. 252, 335–361 (1975).
Sarna, S. K. The gold standard for interpretation of slow wave frequency in in vitro and in vivo recordings by extracellular electrodes. J. Physiol. 591, 4373–4374 (2013).
Daniel, E. E. The electrical and contractile activity of the pyloric region in dogs and the effects of drugs. Gastroenterology 49, 403–418 (1965).
Daniel, E. E., Honour, A. J. & Bogoch, A. Electrical activity of the longitudinal muscle of dog small intestine studied in vivo using microelectrodes. Am. J. Physiol. 198, 113–118 (1960).
Kelly, K. A., Code, C. F. & Elveback, L. R. Patterns of canine gastric electrical activity. Am. J. Physiol. 217, 461–470 (1969).
Szurszewski, J. H. A migrating electric complex of canine small intestine. Am. J. Physiol. 217, 1757–1763 (1969).
Lammers, W. J., al-Kais, A., Singh, S., Arafat, K. & el-Sharkawy, T. Y. Multielectrode mapping of slow-wave activity in the isolated rabbit duodenum. J. Appl. Physiol. 74, 1454–1461 (1993).
Erickson, J. C. et al. Falling-edge, variable threshold (FEVT) method for the automated detection of gastric slow wave events in high-resolution serosal electrode recordings. Ann. Biomed. Eng. 38, 1511–1529 (2010).
Angeli, T. R. et al. The bioelectrical basis and validity of gastrointestinal extracellular slow wave recordings. J. Physiol. 591, 4567–4579 (2013).
Hille, B. Ion Channels of Excitable Membranes (Sinauer Associates Inc, 2001).
Connor, J. A., Prosser, C. L. & Weems, W. A. A study of pace-maker activity in intestinal smooth muscle. J. Physiol. 240, 671–701 (1974).
Ordog, T. et al. Quantitative analysis by flow cytometry of interstitial cells of Cajal, pacemakers, and mediators of neurotransmission in the gastrointestinal tract. Cytometry A 62, 139–149 (2004).
Furness, J. B. The enteric nervous system and neurogastroenterology. Nat. Rev. Gastroenterol. Hepatol. 9, 286–294 (2012).
el-Sharkawy, T. Y., Morgan, K. G. & Szurszewski, J. H. Intracellular electrical activity of canine and human gastric smooth muscle. J. Physiol. 279, 291–307 (1978).
Nakayama, S., Ohishi, R., Sawamura, K., Watanabe, K. & Hirose, K. Microelectrode array evaluation of gut pacemaker activity in wild-type and W/W(v) mice. Biosens. Bioelectron. 25, 61–67 (2009).
Seerden, T. C., Lammers, W. J., De Winter, B. Y., De Man, J. G. & Pelckmans, P. A. Spatiotemporal electrical and motility mapping of distension-induced propagating oscillations in the murine small intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 289, G1043–G1051 (2005).
Bass, P., Code, C. F. & Lambert, E. H . Electric activity of gastroduodenal junction. Am. J. Physiol. 201, 587–592 (1961).
Papasova, M. & Boev, K. in Physiology of Smooth Muscle (eds Bulbring, E. & Shuba, M. F.) 209–216 (Raven Press, 1976).
el-Sharkawy, T. Y. & Szurszewski, J. H. Modulation of canine antral circular smooth muscle by acetylcholine, noradrenaline and pentagastrin. J. Physiol. 279, 309–320 (1978).
Hennig, G. W. et al. Propagation of pacemaker activity in the guinea-pig antrum. J. Physiol. 556, 585–599 (2004).
Paskaranandavadivel, N., O'Grady, G., Du, P. & Cheng, L. K. Comparison of filtering methods for extracellular gastric slow wave recordings. Neurogastroenterol. Motil. 25, 79–83 (2013).
Youm, J. B. et al. A mathematical model of pacemaker activity recorded from mouse small intestine. Philos. Trans. A Math. Phys. Eng. Sci. 364, 1135–1154 (2006).
Edwards, F. R. & Hirst, G. D. An electrical description of the generation of slow waves in the antrum of the guinea-pig. J. Physiol. 564, 213–232 (2005).
Goto, K., Matsuoka, S. & Noma, A. Two types of spontaneous depolarizations in the interstitial cells freshly prepared from the murine small intestine. J. Physiol. 559, 411–422 (2004).
Imtiaz, M. S., von der Weid, P. Y. & van Helden, D. F. Synchronization of Ca2+ oscillations: a coupled oscillator-based mechanism in smooth muscle. FEBS J. 277, 278–285 (2010).
van Helden, D. F., Laver, D. R., Holdsworth, J. & Imtiaz, M. S. Generation and propagation of gastric slow waves. Clin. Exp. Pharmacol. Physiol. 37, 516–524 (2010).
Singh, R. D. et al. Ano1, a Ca2+-activated Cl− channel, coordinates contractility in mouse intestine by Ca2+ transient coordination between interstitial cells of Cajal. J. Physiol. 592, 4051–4068 (2014).
Aliev, R. R., Richards, W. & Wikswo, J. P. A simple nonlinear model of electrical activity in the intestine. J. Theor. Biol. 204, 21–28 (2000).
Buist, M. L., Corrias, A. & Poh, Y. C. A model of slow wave propagation and entrainment along the stomach. Ann. Biomed. Eng. 38, 3022–3030 (2010).
Pullan, A., Cheng, L., Yassi, R. & Buist, M. Modelling gastrointestinal bioelectric activity. Prog. Biophys. Mol. Biol. 85, 523–550 (2004).
Du, P. et al. The impact of surgical excisions on human gastric slow wave conduction, defined by high-resolution electrical mapping and in silico modeling. Neurogastroenterol. Motil. 27, 1409–1422 (2015).
Du, P., Paskaranandavadivel, N., O'Grady, G., Tang, S. J. & Cheng, L. K. A theoretical study of the initiation, maintenance and termination of gastric slow wave re-entry. Math. Med. Biol. 32, 405–423 (2014).
Kim, J. H., Du, P. & Cheng, L. K. Reconstruction of normal and abnormal gastric electrical sources using a potential based inverse method. Physiol. Meas. 34, 1193–1206 (2013).
Lammers, W. J. et al. Slow wave propagation and plasticity of interstitial cells of Cajal in the small intestine of diabetic rats. Exp. Physiol. 96, 1039–1048 (2011).
O'Grady, G. et al. Rapid high-amplitude circumferential slow wave propagation during normal gastric pacemaking and dysrhythmias. Neurogastroenterol. Motil. 24, e299–e312 (2012).
Hirst, G. D., Garcia-Londono, A. P. & Edwards, F. R. Propagation of slow waves in the guinea-pig gastric antrum. J. Physiol. 571, 165–177 (2006).
Dinning, P. G., Arkwright, J. W., Gregersen, H., O'Grady, G. & Scott, S. M. Technical advances in monitoring human motility patterns. Neurogastroenterol. Motil. 22, 366–380 (2010).
Baker, S. A. et al. Spontaneous Ca2+ transients in interstitial cells of Cajal located within the deep muscular plexus of the murine small intestine. J. Physiol. 594, 3317–3338 (2016).
Rhee, P. L. et al. Analysis of pacemaker activity in the human stomach. J. Physiol. 589, 6105–6118 (2011).
Acknowledgements
The authors are grateful to Y. Bayguinov for providing the images in Fig. 2 and Y. Shen and Y.-D. Luo from the Departments of Electrical and Biomedical Engineering at the University of Nevada, USA, for discussions and help with the signal processing used in Fig. 5. The authors are grateful to Y. Kito (Department of Pharmacology, Saga University, Japan) for providing the slow-wave data used in Fig. 5. The authors are also grateful to R. Mathias (State University of New York at Stony Brook, USA), D. Eisner (University of Manchester, UK) and A. Rich (State University of New York at Brockport, USA) for reading and commenting on this Perspectives article. The authors are supported by: R37 DK-40569 to K.M.S; R01 DK-057236 to S.M.W. and P01 DK-41315 to K.M.S. and S.M.W.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Sanders, K., Ward, S. & Hennig, G. Problems with extracellular recording of electrical activity in gastrointestinal muscle. Nat Rev Gastroenterol Hepatol 13, 731–741 (2016). https://doi.org/10.1038/nrgastro.2016.161
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2016.161
This article is cited by
-
Scalable ultrasmall three-dimensional nanowire transistor probes for intracellular recording
Nature Nanotechnology (2019)
-
Correct techniques for extracellular recordings of electrical activity in gastrointestinal muscle
Nature Reviews Gastroenterology & Hepatology (2017)
-
The powerful advantages of extracellular electrical recording
Nature Reviews Gastroenterology & Hepatology (2017)
-
Extracellular gastrointestinal electrical recordings: movement not electrophysiology
Nature Reviews Gastroenterology & Hepatology (2017)
-
Power comes from technical fidelity, not from ease of use
Nature Reviews Gastroenterology & Hepatology (2017)