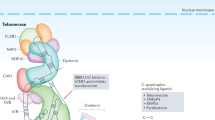
Abstract
Tumour growth is an evolutionary process that is characterized by the selection of clonal populations of cells that acquire distinct genetic changes. Many cancer therapies aim to exploit the specific changes that occur in cancer cells, but understanding the underlying mechanisms of genomic instability that cause these mutations could lead to more effective therapies. If common mechanisms exist for initiating genomic instability in tumours, selection could explain the differences in specific gene mutations that accumulate in different tumour types. The cause of genomic instability in human tumours is unclear, although there is evidence to indicate that telomere dysfunction could make an important contribution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mitelman, F. (ed. Bertil Johansson, F. M.) Catalog of Chromosome Aberrations in Cancer (Wiley–Liss, New York, 1994).
Loeb, L. A., Springgate, C. F. & Battula, N. Errors in DNA replication as a basis of malignant changes. Cancer Res. 34, 2311–2321 (1974).
Shih, I. M. et al. Evidence that genetic instability occurs at an early stage of colorectal tumorigenesis. Cancer Res. 61, 818–822 (2001).
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instability in colorectal cancers. Nature 386, 623–627 (1997).
Phear, G., Bhattacharyya, N. P. & Meuth, M. Loss of heterozygosity and base substitution at the APRT locus in mismatch-repair-proficient and-deficient colorectal carcinoma cell lines. Mol. Cell. Biol. 16, 6516–6523 (1996).
Cahill, D. P. et al. Mutations of mitotic checkpoint genes in human cancers. Nature 392, 300–303 (1998).
Da Costa L. T. & Lengauer, C. Exploring and exploiting instability. Cancer Biol. Ther. 1, 212–225 (2002).
Maser, R. S. & DePinho, R. A. Connecting chromosomes, crisis, and cancer. Science 297, 565–569 (2002).
Muller, H. J. The remaking of chromosomes. Collecting Net. 13, 181–198 (1938).
McClintock, B. The stability of broken ends of chromosomes in Zea mays. Genetics 26, 234–282 (1941).
Blasco, M. A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997).
Hemann, M. T., Strong, M. A., Hao, L. Y. & Greider, C. W. The shortest telomere, not average telomere length, is critical for cell viability and chromosome stability. Cell 107, 67–77 (2001).
Hackett, J. A., Feldser, D. M. & Greider, C. W. Telomere dysfunction increases mutation rate and genomic instability. Cell 106, 275–286 (2001).
Gisselsson, D. et al. Telomere dysfunction triggers extensive DNA fragmentation and evolution of complex chromosome abnormalities in human malignant tumors. Proc. Natl Acad. Sci. USA 98, 12683–12688 (2001).
Lengauer, C. How do tumors make ends meet? Proc. Natl Acad. Sci. USA 98, 12331–12333 (2001).
Baumann, P. & Cech, T. R. Pot1, the putative telomere end-binding protein in fission yeast and humans. Science 292, 1171–1175 (2001).
Carson, M. & Hartwell, L. CDC 17: an essential gene that prevents telomere elongation in yeast. Cell 42, 249–257 (1985).
Garvik, B., Carson, M. & Hartwell, L. Single-stranded DNA arising at telomeres in cdc13 mutants may constitute a specific signal for the RAD9 checkpoint. Mol. Cell. Biol. 15, 6128–6138 (1995).
O'Hagan, R. C. et al. Telomere dysfunction provokes regional amplification and deletion in cancer genomes. Cancer Cell 2, 149–155 (2002).
Artandi, S. E. et al. Telomere dysfunction promotes non-reciprocal translocations and epithelial cancers in mice. Nature 406, 641–645 (2000).
McClintock, B. The behavior in successive nuclear divisions of a chromosome broken at meiosis. Proc. Natl Acad. Sci. USA 25, 405–416 (1939).
Nowak, M. A. et al. The role of chromosomal instability in tumor initiation. Proc. Natl Acad. Sci. USA 99, 16226–16231 (2002).
Tomlinson, I. P., Novelli, M. R. & Bodmer, W. F. The mutation rate and cancer. Proc. Natl Acad. Sci. USA 93, 14800–14803 (1996).
Rudolph, K. L. et al. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 96, 701–712 (1999).
Chin, L. et al. p53 deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcinogenesis. Cell 97, 527–538 (1999).
Rudolph, K. L., Millard, M., Bosenberg, M. W. & DePinho, R. A. Telomere dysfunction and evolution of intestinal carcinoma in mice and humans. Nature Genet. 28, 155–159 (2001).
Chang, S., Khoo, C. & DePinho, R. A. Modeling chromosomal instability and epithelial carcinogenesis in the telomerase-deficient mouse. Semin. Cancer Biol. 11, 227–239 (2001).
Harley, C. B., Futcher, A. B. & Greider, C. W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990).
de Lange, T. et al. Structure and variability of human chromosome ends. Mol. Cell. Biol. 10, 518–527 (1990).
de Lange, T. in Telomeres (eds. Blackburn, E. H. & Greider, C. W.) 265–293 (Cold Spring Harbor Laboratory Press, New York, 1995).
Meeker, A. K. et al. Telomere length assessment in human archival tissues: combined telomere fluorescence in situ hybridization and immunostaining. Am. J. Pathol. 160, 1259–1268 (2002).
van Heek, N. T. et al. Telomere shortening is nearly universal in pancreatic intraepithelial neoplasia. Am. J. Pathol. 161, 1541–1547 (2002).
Meeker, A. K. et al. Telomere shortening is an early somatic DNA alteration in human prostate tumorigenesis. Cancer Res. 62, 6405–6409 (2002).
Ekbom, A., Helmick, C., Zack, M. & Adami, H. O. Ulcerative colitis and colorectal cancer. A population-based study. N. Engl. J. Med. 323, 1228–1233 (1990).
Kinouchi, Y. et al. Telomere shortening in the colonic mucosa of patients with ulcerative colitis. J. Gastroenterol. 33, 343–348 (1998).
Kitada, T., Seki, S., Kawakita, N., Kuroki, T. & Monna, T. Telomere shortening in chronic liver diseases. Biochem. Biophys. Res. Commun. 211, 33–39 (1995).
O'Sullivan, J. N. et al. Chromosomal instability in ulcerative colitis is related to telomere shortening. Nature Genet. 32, 280–284 (2002).
Engelhardt, M., Drullinsky, P., Guillem, J. & Moore, M. A. Telomerase and telomere length in the development and progression of premalignant lesions to colorectal cancer. Clin. Cancer Res. 3, 1931–1941 (1997).
Campisi, J. Replicative senescence: an old lives' tale? Cell 84, 497–500 (1996).
Greider, C. W. Cellular responses to telomere shortening: cellular senescence as a tumor suppressor mechanism. Harvey Lect. 96, 33–50 (2000).
Hayflick, L. & Moorhead, P. S. Exp. Cell Res. 25, 585–621 (1961).
Counter, C. M. et al. Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. EMBO J. 11, 1921–1929 (1992).
Sherwood, S. W., Rush, D., Ellsworth, J. L. & Schimke, R. T. Defining cellular senescence in IMR-90 cells: a flow cytometric analysis. Proc. Natl Acad. Sci. USA 85, 9086–9090 (1988).
Benn, P. A. Specific chromosome aberrations in senescent fibroblast cell lines derived from human embryos. Am. J. Hum. Genet. 28, 465–473 (1976).
Cahill, D. P., Kinzler, K. W., Vogelstein, B. & Lengauer, C. Genetic instability and darwinian selection in tumours. Trends Cell Biol. 9, M57–M60 (1999).
Hackett, J. A. & Greider, C. W. Balancing instability: dual roles for telomerase and telomere dysfunction in tumorigenesis. Oncogene 21, 619–626 (2002).
Kim, N. W. et al. Specific association of human telomerase activity with immortal cells and cancer. Science 266, 2011–2015 (1994).
Shay, J. W. & Bacchetti, S. A survey of telomerase activity in human cancer. Eur. J. Cancer 33, 787–791 (1997).
Chadeneau, C., Hay, K., Hirte, H. W., Gallinger, S. & Bacchetti, S. Telomerase activity associated with acquisition of malignancy in human colorectal cancer. Cancer Res. 55, 2533–2536 (1995).
Tang, R., Cheng, A. J., Wang, J. Y. & Wang, T. C. Close correlation between telomerase expression and adenomatous polyp progression in multistep colorectal carcinogenesis. Cancer Res. 58, 4052–4054 (1998).
Bryan, T. M., Englezou, A., Dunham, M. A. & Reddel, R. R. Telomere length dynamics in telomerase-positive immortal human cell populations. Exp. Cell Res. 239, 370–378 (1998).
Myung, K., Chen, C. & Kolodner, R. D. Multiple pathways cooperate in the suppression of genome instability in Saccharomyces cerevisiae. Nature 411, 1073–1076 (2001).
Maringele, L. & Lydall, D. EXO1-dependent single-stranded DNA at telomeres activates subsets of DNA damage and spindle checkpoint pathways in budding yeast yku70Delta mutants. Genes Dev. 16, 1919–1933 (2002).
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instabilities in human cancers. Nature 396, 643–649 (1998).
Acknowledgements
We thank members of the Greider lab for reading the manuscript and A. I. Meeker for providing the image in figure 3. This work is supported by grants from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
Cancer.gov
LocusLink
OMIM
FURTHER INFORMATION
Rights and permissions
About this article
Cite this article
Feldser, D., Hackett, J. & Greider, C. Telomere dysfunction and the initiation of genome instability. Nat Rev Cancer 3, 623–627 (2003). https://doi.org/10.1038/nrc1142
Issue Date:
DOI: https://doi.org/10.1038/nrc1142
This article is cited by
-
Telomere length and epigenetic clocks as markers of cellular aging: a comparative study
GeroScience (2022)
-
TRIM28 inhibits alternative lengthening of telomere phenotypes by protecting SETDB1 from degradation
Cell & Bioscience (2021)
-
Pwp1 regulates telomere length by stabilizing shelterin complex and maintaining histone H4K20 trimethylation
Cell Discovery (2019)
-
Generation of an Rtel1-CreERT2 knock-in mouse model for lineage tracing RTEL1+ stem cells during development
Transgenic Research (2018)
-
Transient ALT activation protects human primary cells from chromosome instability induced by low chronic oxidative stress
Scientific Reports (2017)