Abstract
Engineered nanoparticles have been used to provide diagnostic1,2,3, therapeutic4,5 and prognostic information6,7 about the status of disease. Nanoparticles developed for these purposes are typically modified with targeting ligands (such as antibodies8,9,10, peptides11,12 or small molecules13) or contrast agents14,15,16 using complicated processes and expensive reagents. Moreover, this approach can lead to an excess of ligands on the nanoparticle surface, and this causes non-specific binding17,18,19,20 and aggregation of nanoparticles18,19,20, which decreases detection sensitivity17,18,19,20. Here, we show that magnetoferritin nanoparticles (M-HFn) can be used to target and visualize tumour tissues without the use of any targeting ligands or contrast agents. Iron oxide nanoparticles are encapsulated inside a recombinant human heavy-chain ferritin (HFn) protein shell, which binds to tumour cells that overexpress transferrin receptor 1 (TfR1). The iron oxide core catalyses the oxidation of peroxidase substrates in the presence of hydrogen peroxide to produce a colour reaction that is used to visualize tumour tissues. We examined 474 clinical specimens from patients with nine types of cancer and verified that these nanoparticles can distinguish cancerous cells from normal cells with a sensitivity of 98% and specificity of 95%.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
22 October 2012
Editorial note: A potential breach of our editorial policies has emerged following the publication of this paper. Yongxin Pan and Changqian Cao, who are listed as contributing authors, have declared no knowledge of the submission of the paper, and their disagreement with its publication. They have pointed out several errors in the description of the synthesis and characterization of the magnetoferritin nanoparticles used in the study. We take breaches of editorial policies very seriously and have therefore informed the listed authors' institutions and the Chinese Academy of Sciences.
27 November 2012
Editorial note: Following an investigation by the Chinese Academy of Sciences under the directive of Professor Chunli Bai, the investigating committee consisting of Professor Tao Xu, Professor Rixiang Zhu, Professor Jinghui Guo and Professor Ruiming Xu concluded that, owing to lack of communication between the authors, the original paper published online on 17 June 2012 contained errors but the central findings of the paper remain valid. The authors have reconciled their opinions and relevant sections of the paper have now been corrected through a Corrigendum.
27 November 2012
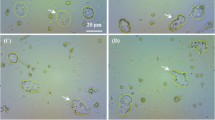
In the version of this Letter originally published, Fig. 1a was incorrect and in Fig. 1c the wrong TEM image was used. In the Supplementary Information, one of the authors was not mentioned in the author list: Lina Song has now been added. In the section 'Preparation and characterization of M-HFn particles' the column used for size-exclusion chromatography was incorrect: it should have been 'Sepharose 6B'. The synthesis procedure for M-HFn nanoparticles was incorrect: it should have read 'HFn protein shells were used as a reaction template to synthesize iron oxide nanoparticles according to the method reported by Cao et al.2 with some modification. The solution of 50 ml 100 mM NaCl with HFn (1 mg ml−1) was added to the reaction vessel, synthesized at 65 °C and pH 8.5. Fe(II) (25 mM (NH4)2Fe(SO4)2•6H2O) and stoichiometric equivalents (1:3 H2O2:Fe2+) of freshly prepared H2O2 (8.33 mM) were added. Fe(II) was added in a rate of 100 Fe/(protein min) using a dosing device (800 Dosino) connected with 842 Titrando. After theoretical 5000 Fe/ protein cage were added to the reaction vessel, the reaction was continued for another 5 min. Finally, 200 μl of 300 mM sodium citrate was added to chelate any free iron. The synthesized magnetite-containing HFn (M-HFn) nanoparticles were centrifuged and purified through size exclusion chromatography to remove the aggregated nanoparticles. The concentration of M-HFn nanoparticles was assumed to be the same as that of HFn protein and was determined using a BCA protein assay kit (Pierce). Purified M-HFn nanoparticles were obtained with a yield of about 75%.' Reference 2 was incorrect and should have read Cao, C. Q. et al. Magnetic characterization of noninteracting, randomly oriented, nanometer-scale ferrimagnetic particles. J. Geophys. Res. 115, B07103 (2010). The aforementioned errors did not affect the main conclusions of the paper. These errors have now been corrected in the HTML and PDF versions.
05 December 2012
Nature Nanotechnology 7, 459–464 (2012); published online 17 June 2012; corrected after print 22 October 2012; corrected after print 27 November 2012. In the version of this Letter originally published, Fig. 1a was incorrect and in Fig. 1c the wrong TEM image was used, they should have appeared as shown below.
References
Galanzha, E. I. et al. In vivo magnetic enrichment and multiplex photoacoustic detection of circulating tumour cells. Nature Nanotech. 4, 855–860 (2009).
Winter, P. M. et al. Molecular imaging of angiogenesis in nascent Vx-2 rabbit tumors using a novel ανβ3-targeted nanoparticle and 1.5 tesla magnetic resonance imaging. Cancer Res. 63, 5838–5843 (2003).
Stoeva, S. I., Lee, J. S., Smith, J. E., Rosen, S. T. & Mirkin, C. A. Multiplexed detection of protein cancer markers with biobarcoded nanoparticle probes. J. Am. Chem. Soc. 128, 8378–8379 (2006).
Janib, S. M., Moses, A. S. & Mackay, J. A. Imaging and drug delivery using theranostic nanoparticles. Adv. Drug Del. Rev. 3, 1–2 (2010).
Zrazhevskiy, P., Sena, M. & Gao, X. H. Designing multifunctional quantum dots for bioimaging, detection, and drug delivery. Chem. Soc. Rev. 3, 1–2 (2010).
Li, R. L. et al. Prognostic value of Akt-1 in human prostate cancer: a computerized quantitative assessment with quantum dot technology. Clin. Cancer Res. 15, 3568–3573 (2009).
Karathanasis, E. et al. Multifunctional nanocarriers for mammographic quantification of tumor dosing and prognosis of breast cancer therapy. Biomaterials 29, 4815–4822 (2008).
Jaetao, J. E. et al. Enhanced leukemia cell detection using a novel magnetic needle and nanoparticles. Cancer Res. 69, 8310–8316 (2009).
Corsi, F. et al. HER2 expression in breast cancer cells is downregulated upon active targeting by antibody-engineered multifunctional nanoparticles in mice. ACS Nano 5, 6383–6393 (2011).
Xu, H. Y. et al. Antibody conjugated magnetic iron oxide nanoparticles for cancer cell separation in fresh whole blood. Biomaterials 32, 9758–9765 (2011).
Park, Y. et al. A new atherosclerotic lesion probe based on hydrophobically modified chitosan nanoparticles functionalized by the atherosclerotic plaque targeted peptides. J. Control. Rel. 128, 217–223 (2008).
Kumar, A. et al. Gold nanoparticles functionalized with therapeutic and targeted peptides for cancer treatment. Biomaterials 33, 1180–1189 (2012).
Weissleder, R., Kelly, K., Sun, E. Y., Shtatland, T. & Josephson, L. Cell-specific targeting of nanoparticles by multivalent attachment of small molecules. Nature Biotechnol. 23, 1418–1423 (2005).
Gao, X., Cui, Y., Levenson, R. M., Chung, L. W. & Nie, S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nature Biotechnol. 22, 969–976 (2004).
Michalet, X. et al. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 307, 538–544 (2005).
Jin, Y. D., Jia, C. X., Huang, S. W., O'Donnell, M. & Gao, X. H. Multifunctional nanoparticles as coupled contrast agents. Nature Commun. 1, 1–8 (2010).
Chen, H. W. et al. Reducing non-specific binding and uptake of nanoparticles and improving cell targeting with an antifouling PEO-b-PγMPS copolymer coating. Biomaterials 31, 5397–5407 (2010).
Bagwe, R. P., Hilliard, L. R. & Tan, W. Surface modification of silica nanoparticles to reduce aggregation and nonspecific binding. Langmuir 22, 4357–4362 (2006).
Grubbs, R. B. Roles of polymer ligands in nanoparticle stabilization. Polym. Rev. 47, 197–215 (2007).
Doshi, N. & Mitragotri, S. Designer biomaterials for nanomedicine. Adv. Funct. Mater. 19, 3843–3854 (2009).
Theil, E. C. Ferritin-structure, gene-regulation, and cellular function in animals, plants, and microorganisms. Annu. Rev. Biochem. 56, 289–315 (1987).
Meldrum, F. C., Heywood, B. R. & Mann, S. Magnetoferritin: in vitro synthesis of a novel magnetic protein. Science 257, 522–523 (1992).
Douglas, T. & Young, M. Host–guest encapsulation of materials by assembled virus protein cages. Nature 393, 152–155 (1998).
Allen, M., Willits, D., Mosolf, J., Young, M. & Douglas, T. Protein cage constrained synthesis of ferrimagnetic iron oxide nanoparticles. Adv. Mater. 14, 1562–1565 (2002).
Li, L. et al. Binding and uptake of H-ferritin are mediated by human transferrin receptor-1. Proc. Natl Acad. Sci. USA 107, 3505–3510 (2010).
Sutherland, R. et al. Ubiquitous cell-surface glycoprotein on tumor-cells is proliferation-associated receptor for transferrin. Proc. Natl Acad. Sci. USA 78, 4515–4519 (1981).
Ryschich, E. et al. Transferrin receptor is a marker of malignant phenotype in human pancreatic cancer and in neuroendocrine carcinoma of the pancreas. Eur. J. Cancer 40, 1418–1422 (2004).
Walker, R. A. & Day, S. J. Transferrin receptor expression in non-malignant and malignant human breast tissue. J. Pathol. 148, 217–224 (1986).
Daniels, T. R., Delgado, T., Rodriguez, J. A., Helguera, G. & Penichet, M. L. The transferrin receptor part I: biology and targeting with cytotoxic antibodies for the treatment of cancer. Clin. Immunol. 121, 144–158 (2006).
Uchida, M. et al. Targeting of cancer cells with ferrimagnetic ferritin cage nanoparticles. J. Am. Chem. Soc. 128, 16626–16633 (2006).
Lin, X. et al. Chimeric ferritin nanocages for multiple function loading and multimodal imaging. Nano Lett. 11, 814–819 (2011).
Lin, X. et al. Hybrid ferritin nanoparticles as activatable probes for tumor imaging. Angew. Chem. Int. Ed. 50, 1569–1572 (2011).
Gao, L. Z. et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nature Nanotech. 2, 577–583 (2007).
Cao, C. Q. et al. Magnetic characterization of noninteracting, randomly oriented, nanometer-scale ferrimagnetic particles. J. Geophys. Res. 115, B07103 (2010).
Mann, S., Bannister, J. V. & Williams, R. J. Structure and composition of ferritin cores isolated from human spleen, limpet (Patella vulgata) hemolymph and bacterial (Pseudomonas aeruginosa) cells. J. Mol. Biol. 188, 225–232 (1986).
Acknowledgements
This work was partially supported by grants from the National Science and Technology Major Project (2012ZX10002009-016), the Knowledge Innovation Program of the Chinese Academy of Sciences (KJCX2-YW-M15), 973 Program (2011CB933500, 2012CB934003), and the National Defense Science and Technology Innovation Fund of Chinese Academy of Sciences (CXJJ-11-M61).
Author information
Authors and Affiliations
Contributions
M.L. conceived and designed the experiments. K.F. and M.L. performed the experiments. M.L. and X.Y. reviewed, analysed and interpreted the data. C.C. and Y.P. synthesized the nanoparticles. D.L., D.Y., J.F. and L.S. cultured the cancer cells. M.L. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 3243 kb)
Rights and permissions
About this article
Cite this article
Fan, K., Cao, C., Pan, Y. et al. Magnetoferritin nanoparticles for targeting and visualizing tumour tissues. Nature Nanotech 7, 459–464 (2012). https://doi.org/10.1038/nnano.2012.90
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2012.90
This article is cited by
-
Impact of doxorubicin-loaded ferritin nanocages (FerOX) vs. free doxorubicin on T lymphocytes: a translational clinical study on breast cancer patients undergoing neoadjuvant chemotherapy
Journal of Nanobiotechnology (2024)
-
A natural biogenic nanozyme for scavenging superoxide radicals
Nature Communications (2024)
-
Machine-learning-assisted single-vessel analysis of nanoparticle permeability in tumour vasculatures
Nature Nanotechnology (2023)
-
Supercritical fluid-assisted fabrication of C-doped Co3O4 nanoparticles based on polymer-coated metal salt nanoreactors for efficient enzyme-mimicking and glucose sensor properties
Nano Research (2023)
-
Copper-induced synthesis of versatile FeOx nanozymes for catalytic cancer therapy
Journal of Materials Science (2023)