Abstract
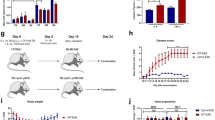
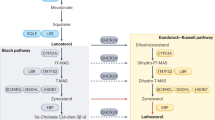
Duplication of PLP1 (proteolipid protein gene 1) and the subsequent overexpression of the myelin protein PLP (also known as DM20) in oligodendrocytes is the most frequent cause of Pelizaeus-Merzbacher disease (PMD), a fatal leukodystrophy1 without therapeutic options2,3. PLP binds cholesterol and is contained within membrane lipid raft microdomains4. Cholesterol availability is the rate-limiting factor of central nervous system myelin synthesis5. Transgenic mice with extra copies of the Plp1 gene6 are accurate models of PMD. Dysmyelination6,7,8 followed by demyelination9,10, secondary inflammation and axon damage contribute to the severe motor impairment in these mice9,10. The finding that in Plp1-transgenic oligodendrocytes, PLP and cholesterol accumulate in late endosomes and lysosomes (endo/lysosomes)9,11,12,13, prompted us to further investigate the role of cholesterol in PMD. Here we show that cholesterol itself promotes normal PLP trafficking and that dietary cholesterol influences PMD pathology. In a preclinical trial, PMD mice were fed a cholesterol-enriched diet. This restored oligodendrocyte numbers and ameliorated intracellular PLP accumulation. Moreover, myelin content increased, inflammation and gliosis were reduced and motor defects improved. Even after onset of clinical symptoms, cholesterol treatment prevented disease progression. Dietary cholesterol did not reduce Plp1 overexpression but facilitated incorporation of PLP into myelin membranes. These findings may have implications for therapeutic interventions in patients with PMD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Seitelberger, F. Neuropathology and genetics of Pelizaeus-Merzbacher disease. Brain Pathol. 5, 267–273 (1995).
Garbern, J.Y. Pelizaeus-Merzbacher disease: Genetic and cellular pathogenesis. Cell Mol. Life Sci. 64, 50–65 (2007).
Woodward, K.J. The molecular and cellular defects underlying Pelizaeus-Merzbacher disease. Expert Rev. Mol. Med. 10, e14 (2008).
Simons, M., Kramer, E.M., Thiele, C., Stoffel, W. & Trotter, J. Assembly of myelin by association of proteolipid protein with cholesterol- and galactosylceramide-rich membrane domains. J. Cell Biol. 151, 143–154 (2000).
Saher, G. et al. High cholesterol level is essential for myelin membrane growth. Nat. Neurosci. 8, 468–475 (2005).
Readhead, C., Schneider, A., Griffiths, I. & Nave, K.A. Premature arrest of myelin formation in transgenic mice with increased proteolipid protein gene dosage. Neuron 12, 583–595 (1994).
Anderson, T.J. et al. Distinct phenotypes associated with increasing dosage of the PLP gene: implications for CMT1A due to PMP22 gene duplication. Ann. NY Acad. Sci. 883, 234–246 (1999).
Anderson, T.J. et al. Late-onset neurodegeneration in mice with increased dosage of the proteolipid protein gene. J. Comp. Neurol. 394, 506–519 (1998).
Edgar, J.M. et al. Demyelination and axonal preservation in a transgenic mouse model of Pelizaeus-Merzbacher disease. EMBO Mol. Med. 2, 42–50 (2010).
Karim, S.A. et al. PLP/DM20 expression and turnover in a transgenic mouse model of Pelizaeus-Merzbacher disease. Glia 58, 1727–1738 (2010).
Krämer, E.M., Schardt, A. & Nave, K.A. Membrane traffic in myelinating oligodendrocytes. Microsc. Res. Tech. 52, 656–671 (2001).
Karim, S.A. et al. PLP overexpression perturbs myelin protein composition and myelination in a mouse model of Pelizaeus-Merzbacher disease. Glia 55, 341–351 (2007).
Simons, M. et al. Overexpression of the myelin proteolipid protein leads to accumulation of cholesterol and proteolipid protein in endosomes/lysosomes: implications for Pelizaeus-Merzbacher disease. J. Cell Biol. 157, 327–336 (2002).
Björkhem, I. & Meaney, S. Brain cholesterol: long secret life behind a barrier. Arterioscler. Thromb. Vasc. Biol. 24, 806–815 (2004).
Alberti, S. et al. Hepatic cholesterol metabolism and resistance to dietary cholesterol in LXRbeta-deficient mice. J. Clin. Invest. 107, 565–573 (2001).
Ip, C.W. et al. Origin of CD11b+ macrophage-like cells in the CNS of PLP-overexpressing mice: low influx of haematogenous macrophages and unchanged blood-brain-barrier in the optic nerve. Mol. Cell Neurosci. 38, 489–494 (2008).
Stolp, H.B. & Dziegielewska, K.M. Review: Role of developmental inflammation and blood-brain barrier dysfunction in neurodevelopmental and neurodegenerative diseases. Neuropathol. Appl. Neurobiol. 35, 132–146 (2009).
Xie, C., Lund, E.G., Turley, S.D., Russell, D.W. & Dietschy, J.M. Quantitation of two pathways for cholesterol excretion from the brain in normal mice and mice with neurodegeneration. J. Lipid Res. 44, 1780–1789 (2003).
Liu, B. et al. Reversal of defective lysosomal transport in NPC disease ameliorates liver dysfunction and neurodegeneration in the Npc1−/− mouse. Proc. Natl. Acad. Sci. USA 106, 2377–2382 (2009).
Brown, M.S. & Goldstein, J.L. The SREBP pathway: regulation of cholesterol metabolism by proteolysis of a membrane-bound transcription factor. Cell 89, 331–340 (1997).
Saher, G. et al. Cholesterol regulates the endoplasmic reticulum exit of the major membrane protein P0 required for peripheral myelin compaction. J. Neurosci. 29, 6094–6104 (2009).
Qin, C., Nagao, T., Grosheva, I., Maxfield, F.R. & Pierini, L.M. Elevated plasma membrane cholesterol content alters macrophage signaling and function. Arterioscler. Thromb. Vasc. Biol. 26, 372–378 (2006).
Shepard, L.A. et al. Identification of a membrane-spanning domain of the thiol-activated pore-forming toxin Clostridium perfringens perfringolysin O: an α-helical to β-sheet transition identified by fluorescence spectroscopy. Biochemistry 37, 14563–14574 (1998).
Karten, B., Peake, K.B. & Vance, J.E. Mechanisms and consequences of impaired lipid trafficking in Niemann-Pick type C1–deficient mammalian cells. Biochim. Biophys. Acta 1791, 659–670 (2009).
Otomo, T., Higaki, K., Nanba, E., Ozono, K. & Sakai, N. Lysosomal storage causes cellular dysfunction in mucolipidosis II skin fibroblasts. J. Biol. Chem. 286, 35283–35290 (2011).
Tang, Y., Leao, I.C., Coleman, E.M., Broughton, R.S. & Hildreth, J.E. Deficiency of niemann-pick type C-1 protein impairs release of human immunodeficiency virus type 1 and results in Gag accumulation in late endosomal/lysosomal compartments. J. Virol. 83, 7982–7995 (2009).
Sobo, K. et al. Late endosomal cholesterol accumulation leads to impaired intra-endosomal trafficking. PLoS ONE 2, e851 (2007).
Hawes, C.M., Wiemer, H., Krueger, S.R. & Karten, B. Pre-synaptic defects of NPC1-deficient hippocampal neurons are not directly related to plasma membrane cholesterol. J. Neurochem. 114, 311–322 (2010).
Bauer, N.G., Richter-Landsberg, C. & Ffrench-Constant, C. Role of the oligodendroglial cytoskeleton in differentiation and myelination. Glia 57, 1691–1705 (2009).
Prusky, G.T., Alam, N.M., Beekman, S. & Douglas, R.M. Rapid quantification of adult and developing mouse spatial vision using a virtual optomotor system. Invest. Ophthalmol. Vis. Sci. 45, 4611–4616 (2004).
Goetze, B. et al. Vision and visual cortical maps in mice with a photoreceptor synaptopathy: reduced but robust visual capabilities in the absence of synaptic ribbons. Neuroimage 49, 1622–1631 (2010).
Stromnes, I.M. & Goverman, J.M. Active induction of experimental allergic encephalomyelitis. Nat. Protoc. 1, 1810–1819 (2006).
Jung, M., Sommer, I., Schachner, M. & Nave, K.A. Monoclonal antibody O10 defines a conformationally sensitive cell-surface epitope of proteolipid protein (PLP): evidence that PLP misfolding underlies dysmyelination in mutant mice. J. Neurosci. 16, 7920–7929 (1996).
Hölttä-Vuori, M. et al. BODIPY-cholesterol: a new tool to visualize sterol trafficking in living cells and organisms. Traffic 9, 1839–1849 (2008).
Möbius, W. et al. Immunoelectron microscopic localization of cholesterol using biotinylated and non-cytolytic perfringolysin O. J. Histochem. Cytochem. 50, 43–55 (2002).
Waheed, A.A. et al. Selective binding of perfringolysin O derivative to cholesterol-rich membrane microdomains (rafts). Proc. Natl. Acad. Sci. USA 98, 4926–4931 (2001).
Acknowledgements
We dedicate this work to the memory of Jim Garbern for his support of PMD research. We are grateful to A. Fahrenholz, T. Freerck, M. Schildmann, M. Matthes and A. Kanbach for technical support and thank J. Edgar for helpful comments on the manuscript. We thank M. Grebe (Umea University) for providing the BCθ expression construct and Pfizer for the generous gift of the squalestatin CP-340868. This work was funded by the PMD foundation and the Deutsche Forschungsgemeinschaft (SA2114/1-1) to G.S. K.-A.N. is supported by the EU-FP7 (Leukotreat, Ngidd), the Bundesministerium für Bildung und Forschung (BMBF; Leukonet) and a European Research Council (ERC) Advanced Grant. Support of the BMBF to S.L. and K.-F.S. is gratefully acknowledged (01GQ0810).
Author information
Authors and Affiliations
Contributions
G.S. supervised the project, conducted experiments and wrote the manuscript; F.R., K.C. and T.R. conducted experiments, K.-F.S. and S.L. performed visual acuity measurements, P.D. conducted the two-photon microscopic analysis, B.B. implemented the beam test, W.M. supervised and contributed to electron microscopic studies, and K.-A.N. contributed to the discussion and the writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Table 1 (PDF 1330 kb)
Supplementary Video 1
Motor performance of treated and untreated PMD mice. This video shows an example of a beam test that was used to evaluate motor abilities of PMD mice. (MPG 3093 kb)
Supplementary Video 2
Motor disabilities of cSQS KO and cSQS KO*PLP+/66 mice. (WMV 3826 kb)
Rights and permissions
About this article
Cite this article
Saher, G., Rudolphi, F., Corthals, K. et al. Therapy of Pelizaeus-Merzbacher disease in mice by feeding a cholesterol-enriched diet. Nat Med 18, 1130–1135 (2012). https://doi.org/10.1038/nm.2833
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2833
This article is cited by
-
In conversation with Klaus-Armin Nave
Nature Neuroscience (2023)
-
Genotype–phenotype correlation and natural history analyses in a Chinese cohort with pelizaeus–merzbacher disease
Orphanet Journal of Rare Diseases (2022)
-
Hypomyelinating leukodystrophies — unravelling myelin biology
Nature Reviews Neurology (2021)
-
Suppression of proteolipid protein rescues Pelizaeus–Merzbacher disease
Nature (2020)
-
Ketogenic diet ameliorates axonal defects and promotes myelination in Pelizaeus–Merzbacher disease
Acta Neuropathologica (2019)