Abstract
The balance of myeloid populations and lymphoid populations must be well controlled. Here we found that osteopontin (OPN) skewed this balance during pathogenic conditions such as infection and autoimmunity. Notably, two isoforms of OPN exerted distinct effects in shifting this balance through cell-type-specific regulation of apoptosis. Intracellular OPN (iOPN) diminished the population size of myeloid progenitor cells and myeloid cells, and secreted OPN (sOPN) increase the population size of lymphoid cells. The total effect of OPN on skewing the leukocyte population balance was observed as host sensitivity to early systemic infection with Candida albicans and T cell–mediated colitis. Our study suggests previously unknown detrimental roles for two OPN isoforms in causing the imbalance of leukocyte populations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
References
Manz, M.G. & Boettcher, S. Emergency granulopoiesis. Nat. Rev. Immunol. 14, 302–314 (2014).
Takizawa, H., Boettcher, S. & Manz, M.G. Demand-adapted regulation of early hematopoiesis in infection and inflammation. Blood 119, 2991–3002 (2012).
Patchen, M.L., MacVittie, T.J., Williams, J.L., Schwartz, G.N. & Souza, L.M. Administration of interleukin-6 stimulates multilineage hematopoiesis and accelerates recovery from radiation-induced hematopoietic depression. Blood 77, 472–480 (1991).
Boettcher, S. & Manz, M.G. Regulation of inflammation- and infection-driven hematopoiesis. Trends Immunol. 38, 345–357 (2017).
Lieschke, G.J. et al. Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood 84, 1737–1746 (1994).
Zhan, Y. & Cheers, C. Haemopoiesis in mice genetically lacking granulocyte-macrophage colony stimulating factor during chronic infection with Mycobacterium avium. Immunol. Cell Biol. 78, 118–123 (2000).
Bernad, A. et al. Interleukin-6 is required in vivo for the regulation of stem cells and committed progenitors of the hematopoietic system. Immunity 1, 725–731 (1994).
Zhan, Y., Lieschke, G.J., Grail, D., Dunn, A.R. & Cheers, C. Essential roles for granulocyte-macrophage colony-stimulating factor (GM-CSF) and G-CSF in the sustained hematopoietic response of Listeria monocytogenes-infected mice. Blood 91, 863–869 (1998).
Bugl, S. et al. Steady-state neutrophil homeostasis is dependent on TLR4/TRIF signaling. Blood 121, 723–733 (2013).
Cain, D.W., Snowden, P.B., Sempowski, G.D. & Kelsoe, G. Inflammation triggers emergency granulopoiesis through a density-dependent feedback mechanism. PLoS One 6, e19957 (2011).
Inoue, M. et al. T cells down-regulate macrophage TNF production by IRAK1-mediated IL-10 expression and control innate hyperinflammation. Proc. Natl. Acad. Sci. USA 111, 5295–5300 (2014).
Shinohara, M.L., Kim, J.H., Garcia, V.A. & Cantor, H. Engagement of the type I interferon receptor on dendritic cells inhibits T helper 17 cell development: role of intracellular osteopontin. Immunity 29, 68–78 (2008).
Inoue, M. et al. Cutting edge: critical role of intracellular osteopontin in antifungal innate immune responses. J. Immunol. 186, 19–23 (2011).
Leavenworth, J.W., Verbinnen, B., Wang, Q., Shen, E. & Cantor, H. Intracellular osteopontin regulates homeostasis and function of natural killer cells. Proc. Natl. Acad. Sci. USA 112, 494–499 (2015).
Rittling, S.R. & Singh, R. Osteopontin in immune-mediated diseases. J. Dent. Res. 94, 1638–1645 (2015).
Uede, T. Osteopontin, intrinsic tissue regulator of intractable inflammatory diseases. Pathol. Int. 61, 265–280 (2011).
Shevde, L.A. & Samant, R.S. Role of osteopontin in the pathophysiology of cancer. Matrix Biol. 37, 131–141 (2014).
Kreke, M.R., Huckle, W.R. & Goldstein, A.S. Fluid flow stimulates expression of osteopontin and bone sialoprotein by bone marrow stromal cells in a temporally dependent manner. Bone 36, 1047–1055 (2005).
Stier, S. et al. Osteopontin is a hematopoietic stem cell niche component that negatively regulates stem cell pool size. J. Exp. Med. 201, 1781–1791 (2005).
Nilsson, S.K. et al. Osteopontin, a key component of the hematopoietic stem cell niche and regulator of primitive hematopoietic progenitor cells. Blood 106, 1232–1239 (2005).
Grassinger, J. et al. Thrombin-cleaved osteopontin regulates hemopoietic stem and progenitor cell functions through interactions with α9β1 and α4β1 integrins. Blood 114, 49–59 (2009).
Inoue, M. & Shinohara, M.L. Intracellular osteopontin (iOPN) and immunity. Immunol. Res. 49, 160–172 (2011).
Shinohara, M.L., Kim, H.J., Kim, J.H., Garcia, V.A. & Cantor, H. Alternative translation of osteopontin generates intracellular and secreted isoforms that mediate distinct biological activities in dendritic cells. Proc. Natl. Acad. Sci. USA 105, 7235–7239 (2008).
Leavenworth, J.W., Verbinnen, B., Yin, J., Huang, H. & Cantor, H. A p85α-osteopontin axis couples the receptor ICOS to sustained Bcl-6 expression by follicular helper and regulatory T cells. Nat. Immunol. 16, 96–106 (2015).
Shinohara, M.L. et al. Osteopontin expression is essential for interferon-α production by plasmacytoid dendritic cells. Nat. Immunol. 7, 498–506 (2006).
Ashkar, S. et al. Eta-1 (osteopontin): an early component of type-1 (cell-mediated) immunity. Science 287, 860–864 (2000).
Sekido, Y. Molecular pathogenesis of malignant mesothelioma. Carcinogenesis 34, 1413–1419 (2013).
Robinson, B.S. & Moberg, K.H. Cell-cell junctions: α-catenin and E-cadherin help fence in Yap1. Curr. Biol. 21, R890–R892 (2011).
Kim, N.G., Koh, E., Chen, X. & Gumbiner, B.M. E-cadherin mediates contact inhibition of proliferation through Hippo signaling-pathway components. Proc. Natl. Acad. Sci. USA 108, 11930–11935 (2011).
Qin, F., Tian, J., Zhou, D. & Chen, L. Mst1 and Mst2 kinases: regulations and diseases. Cell Biosci. 3, 31 (2013).
Kanayama, M. et al. Autophagy enhances NFκB activity in specific tissue macrophages by sequestering A20 to boost antifungal immunity. Nat. Commun. 6, 5779 (2015).
Basu, S. et al. “Emergency” granulopoiesis in G-CSF-deficient mice in response to Candida albicans infection. Blood 95, 3725–3733 (2000).
Lai, A.Y., Lin, S.M. & Kondo, M. Heterogeneity of Flt3-expressing multipotent progenitors in mouse bone marrow. J. Immunol. 175, 5016–5023 (2005).
Surh, C.D. & Sprent, J. Homeostasis of naive and memory T cells. Immunity 29, 848–862 (2008).
Kieper, W.C. et al. Recent immune status determines the source of antigens that drive homeostatic T cell expansion. J. Immunol. 174, 3158–3163 (2005).
Yang, B. et al. Inhibitors directed towards caspase-1 and -3 are less effective than pan caspase inhibition in preventing renal proximal tubular cell apoptosis. Nephron Exp. Nephrol. 96, e39–e51 (2004).
Kourepini, E. et al. Osteopontin expression by CD103- dendritic cells drives intestinal inflammation. Proc. Natl. Acad. Sci. USA 111, E856–E865 (2014).
Oz, H.S., Zhong, J. & de Villiers, W.J. Osteopontin ablation attenuates progression of colitis in TNBS model. Dig. Dis. Sci. 57, 1554–1561 (2012).
Heilmann, K. et al. Osteopontin as two-sided mediator of intestinal inflammation. J. Cell. Mol. Med. 13, 1162–1174 (2009).
Toyonaga, T. et al. Osteopontin deficiency accelerates spontaneous colitis in mice with disrupted gut microbiota and macrophage phagocytic activity. PLoS One 10, e0135552 (2015).
Da Silva, A.P. et al. Exacerbated tissue destruction in DSS-induced acute colitis of OPN-null mice is associated with downregulation of TNF-α expression and non-programmed cell death. J. Cell. Physiol. 208, 629–639 (2006).
Hur, E.M. et al. Osteopontin-induced relapse and progression of autoimmune brain disease through enhanced survival of activated T cells. Nat. Immunol. 8, 74–83 (2007).
Kanayama, M., He, Y.W. & Shinohara, M.L. The lung is protected from spontaneous inflammation by autophagy in myeloid cells. J. Immunol. 194, 5465–5471 (2015).
Shinohara, M.L. et al. T-bet-dependent expression of osteopontin contributes to T cell polarization. Proc. Natl. Acad. Sci. USA 102, 17101–17106 (2005).
Chinen, T. et al. Prostaglandin E2 and SOCS1 have a role in intestinal immune tolerance. Nat. Commun. 2, 190 (2011).
Macosko, E.Z. et al. Highly parallel genome-wide expression profiling of individual cells using nanoliter droplets. Cell 161, 1202–1214 (2015).
Acknowledgements
We thank M. Ciofani and M. Zuberbuehler for help in an experiment with fetal liver; M. Kondo for analysis of ex vivo progenitor-differentiation assays; G. Kucera for generating the LSL-iOPN bacterial artificial chromosome construct; the staff at the David H Murdock Research Institute for generating the sequence data; K. Abramson and E. Grass for generating the 10× single-cell libraries; and T. Kadota for help with in vitro experiments with BMMs in this study. Supported by the US National Institutes of Health (R01AI088100 and R21AI103584 to M.L.S.) and the US Department of Defense (PC100266 to M.L.S.).
Author information
Authors and Affiliations
Contributions
M.K. performed the majority of experiments; S.X. performed parts of the experiments assessing cell-density-mediated upregulation of OPN and confirmation of LSL-iOPN mice; K.D. performed parts of the BM-chimera experiments; J.R.G. and S.G.G. performed single-cell analysis, analyzed data and provided editorial help; M.I. provided experimental help in generating and confirming the LSL-iOPN mouse line; and M.K. and M.L.S. conceived of the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
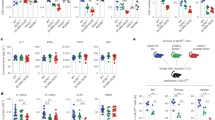
Supplementary Figure 1 Cellularity of leukocytes and their progenitors in naive wild-type and Spp1−/− mice.
(a, b) Gating strategies for differentiated cells including PMN (CD11b+Ly6Ghi and CD11b+Ly6Glo), monocytes (CD11b+Ly6C+), B cells (CD19+B220hi or lo), and T cells (CD3+) in the BM (a); and for progenitors including MPPs (Lin−Sca-1+c-kit+Flt3+CD34+), CMPs (Lin−Sca-1−c-kit+FcγRintCD34+), GMPs (Lin−Sca-1−c-kit+FcγRhiCD34+), and CLPs (Lin−Sca-1intc-kitintIL-7R+) in the BM (b). (c) Total cell numbers in the BM and spleen from naive WT and Spp1−/− mice. (d) Proportions of PMN, monocytes, B cells, T cells, and their progenitors in the BM of 7 weeks old naive WT and Spp1−/− mice. (e) Proportions of Neutrophils, Ly6C+ macrophages (MΦ), F4/80hi MΦ, cDC, T cell and B cells in the spleen of naive WT and Spp1−/− mice. (f, g) Evaluating an impact of OPN in fetal liver progenitors. Spp1+/– mice were bred to produce WT, heterozygous (HZ), and Spp1−/− embryos to evaluate population sizes of MPPs, GMPs, CMPs, and CLPs at E13-15 (f). Percentages of embryonic progenitors are shown in (g). Data (c-e, g) are representative of two independent experiments. Data was analyzed by the Student’s t-test (c-e) and ANOVA (g). N.S.; Not significant.
Supplementary Figure 2 Setting up experiments with irradiated mixed-BM chimeras.
In all the experiments described here, donor cells were mixed at the 1:1 ratio and adoptively transferred to lethally irradiated (900 rad) recipients. (a-c) Strategy for the experiment shown in Fig. 1a, b and Supplementary Fig. 8d (a). The composition of donor LSK cells was confirmed to be ~1:1 before transfer (b). Serum OPN levels in the mix BM chimera 4 weeks after BM cell transfer were compared to those in WT and Spp1−/− mice (c). Error bars denote mean ± SD. Data was analyzed by Student’s t-test. (d, e) Generation of mix LSK chimera mice used in experiments shown in Fig. 1e. Flow-Sorted LSK cells from WT and Spp1−/− mouse BM (104 cells each) were transferred with 107 of c-kit− BM cells obtained from WT mice (with the same congenic marker with recipients) used as “rescue cells” (d). The composition of donor LSK cells was confirmed to be ~1:1 before transfer (e). (f, g) Generation of BM chimera mice for experiments shown in Fig. 8a-c (f). LSL-iOPN mice were described in Supplementary Fig. 7. The composition of donor LSK cells was confirmed to be ~1:1 before transfer (g).
Supplementary Figure 3 Cell analysis of mice going through emergency granulopoiesis induced by injection of thioglycollate or systemic infection with C. albicans.
(a) Frequencies of neutrophils in the BM after thioglycollate injection in WT mice at the indicated time-points. (b, c) Population sizes of CMPs, GMPs, and CLPs in the BM at 24 hr after thioglycollate injection (b). Cell proliferation at the same time point was evaluated by Ki-67 staining (c). (d) Decreased neutrophil population sizes in the BM of WT and Spp1−/− mice at 12 after thioglycollate injection. n=3 per group. (e) Numbers of neutrophils in the peritoneal cavity 12 hrs after thioglycollate injection. (f) Comparison of serum OPN levels in WT mice before and 24 hrs after thioglycollate injection. (g) Spp1 levels in BMMs after 16 hr culture with titrated cell densities on flat-bottom plates without stimulation. (h) Relative mRNA levels of Spp1, Tnfa, Il6, Il10 and Cxcl1 in BMMs cultured in flat-bottom wells to round-bottom wells (flat/round). (i) Spp1 mRNA levels at 24-hr in BMMs cultured in supernatants (supe.) obtained from separate BMM culture using either flat- or round-bottom plates for 24 hr. (j) Reduction of Spp1 expression by inhibiting E-cadherin. BMMs were cultured on flat- or round-bottom plates (105 per well) with or without anti-E-cadherin antibody (10 μg/ml) for 16 hr. (k, l) WT mice were treated with i.p.-injection of AMD3100 (CXCR4 antagonist; 5 mg/kg mouse weight). Total BM cell numbers and frequencies of neutrophils in BM (k) and Spp1 mRNA levels in GMPs (l) are shown at indicated time-points. (m) Frequencies of CLPs, CMPs, and GMPs were examined 24 hr after Candida infection. (n) Candida CFU in the spleen 24 hrs after i.v.-injecting Candida spores (106 spores/mouse). Error bars (too short to be observed) denote RQ-Max/Min as described in the Method section (g). Error bars indicate mean ± SEM (a, c, d, f, h-l). Data were analyzed by the Student’s t-test (b, d-f, i, j, l-m) and ANOVA (c). N.S.; Not significant.
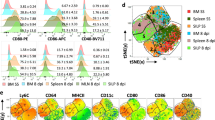
Supplementary Figure 4 Gene-expression profiles of wild-type and Spp1−/− MPPs at the level of single cells and bulk MPPs.
(a-c) t-SNE plots for the WT MPPs (3,312 cells)(a) and Spp1−/− MPPs (4,575 cells)(b). Single-cell RNA-sequencing of MPPs obtained from WT and Spp1−/− mice was performed 20 hrs after intraperitoneal injection of thioglycollate. Data from WT and Spp1−/− MPPs were combined with sequencing depth normalization, shown with the color codes based on UMI (unique molecular identifier) count (c). (d) Heat map of differentially expressed genes in the 10 clusters was indicated. (e) mRNA levels of hematopoiesis-related genes in MPPs determined by qPCR. MPPs were isolated from BM of WT, Spp1−/− or LSL-iOPN-KI (LSL-iOPN) 18 hrs after thioglycollate i.p. injection. Error bars denote RQ-Max/Min as described in the Method section.
Supplementary Figure 5 Apoptosis and proliferation of hematopoietic progenitor cells and myeloid cells during emergency myelopoiesis.
(a, b) 5-bromo-2'-deoxyuridine (BrdU: 30 mg/kg) and 5-fluoro-2'-deoxyuridine (FrdU: 3 mg/kg) were i.p.-injected to WT and Spp1−/− mice at the same time as the thioglycollate i.p.-injection, and BrdU incorporation was assessed at 24 hrs after the injections. Representative histogram images (a) and frequencies of BrdU+ cells in indicated progenitors (b) are shown. (c, d) Evaluating apoptosis in BM progenitors in naive WT and Spp1−/− mice. Representative flow plots (c) and frequencies of apoptotic cells (d) are shown. (e) The strategy of BM transplant used in experiments shown in Fig. 4a, b, i and j. BM cells obtained from WT or Spp1−/− mice (CD45.2) were transferred to lethally irradiated WT (CD45.1) recipients. Three weeks after BM cell transfer, apoptosis of cells in BM was evaluated by flow cytometry. (f, g) Evaluating apoptosis of neutrophils and Ly6C+ monocytes in the BM 24 hrs after i.p. thioglycollate injection. Representative flow images (f) and frequencies of apoptotic cells (g) are shown. (h) Birc5 mRNA levels in Gr-1+ cells and B cells in BM at 15 hr after systemic Candida infection (2x106 spores/mouse). (i) Annexin-V+ cell percentages in splenic T and B cells obtained from naive WT and Spp1−/− mice. (j, k) The proliferation of splenic T and B cells 3 weeks after BM cell (WT vs. Spp1−/−) transfer. BM cells were transferred to WT mice as shown in Supplementary Fig. 5e. Three weeks after BM transplantation, BrdU (30 mg/kg) and FrdU (3 mg/kg) were i.p. injected to the mice and BrdU incorporation was evaluated 24 hrs after BrdU injection. Representative histograms (j) and data with statistical analysis (k) are shown. Each dataset in (b, d, g, h, i, k) is a representative of two independent experiments. Error bars denote mean ± SD. Data was analyzed by the Student’s t-test. N.S.; Not significant. *; p< 0.05.
Supplementary Figure 6 OPN enhances the survival of T cells during cLIP.
(a-d) CD62LhiCD4+ T cells obtained from naive WT and Spp1−/− mice were transferred to Rag1−/− mice, then MLNs were evaluated on day 6. Representative flow cytometry patterns of CD4+ T cells (a). Enumeration of total cells and CD4+ T cells in the MLN (b). Proportions of proliferated CD4+ T cells determined by CFSE dilution (c) and Annexin-V+ cells (d) are shown. Each dataset is a representative of two independent experiments. (e) Serum OPN levels in Rag1−/− or Spp1−/−Rag1−/− recipients 4 weeks after transfer of CD62LhiCD4+ T cells. Error bars denote ± SEM. (f, g) T cell proliferation assay without adding extrinsic antigen. CD62LhiCD4+ T cells obtained from WT or Spp1−/− mice were cultured with or without total MLN cells or splenocytes obtained from Spp1−/−Rag1−/− mice for 7 days (f). T cell proliferation and survival were evaluated by CFSE-dilution and staining with LIVE/DEAD, respectively (g). Each dataset is a representative of three independent experiments. (h, i) Proliferation and survival of T cells obtained from WT and Spp1−/− mice upon TCR stimulation. CellTrace-labeled CD62LhiCD4+ T cells were cultured with CD3/CD28 antibody-coated microbeads for 4 days and proliferation, and the survival was evaluated by CellTrace-dilution and staining with 7-AAD, respectively. Representative flow plots (h), and frequencies of dead cells and live divided cells (i) are shown. (j, k) Numbers and frequencies of helper T cell subsets such as Th1, Th17 and Treg (j) and innate immune cells such as neutrophils, Ly6C+ macrophages (MΦ), and cDCs (k) in MLNs at 4 weeks after WT or Spp1−/− T cell transfer to Rag1−/− or Spp1−/−Rag1−/− mice. (l, m) Cell surface expression of integrins and CCR9 (l) and frequencies of activated CD4+ T cells (CD62LloCD44hi) in MLN at 4 weeks after WT or Spp1−/− T cell transfer to Rag1−/− or Spp1−/−Rag1−/− mice. Each dataset is a representative of two independent experiments. Data was analyzed by the Student’s t-test. N.S.; Not significant. * p< 0.05.
Supplementary Figure 7 Generation of LSL-iOPN knock-in mice.
(a) iOPN knock-in construct. Boxes indicate Spp1 exons (only first 5 exons are shown). The iOPN knock-in BAC vector has a lox-stop-lox (LSL) cassette and a deletion of 45-nt in the exon 2 (gray box). The deletion allows to generate the OPN isoform that is not secreted by the lack of signal sequence. HSK-TK: herpes simplex virus-thymidine kinase. (b) Southern blot of targeted ES cells by using specific probe (blue line in (a)). Genomic DNA was digested with ScaI. The WT allele has the 10.6 kb band and the LSL-iOPN KI allele (before Flp recombination) has the 17 kb band. Clone 1B9 was used to generate the LSLfl/fl-Δ45Spp1 mouse line. (c) Confirmation of successful iOPN expression sequence in LSL-iOPN (LSLfl/fl-Δ45Spp1;Vav1-Cre) mice. The 45-nt deletion in exon 2 was evaluated by PCR using specific primers (gray arrow heads in (a)), which amplify a 344-bp amplicon from the WT Spp1 allele a 299-bp amplicon from mutant Δ45Spp1 allele. (d) Quantitation of iOPN was performed in ELISA in the cytoplasmic fraction obtained by iodixanol density gradient. Amounts of OPN per arbitrary volume of cytoplasmic fraction were shown. Successful separation of the cytoplasmic proteins was confirmed by Western blotting with GAPDH detection, but not with calnexin. (e) Total cell lysates (including ER/Golgi) were quantitatively evaluated for their OPN concentrations by ELISA. (f) sOPN levels in BMM culture supernatants. n=3 mice per group. Error bars denote ± SEM.
Supplementary Figure 8 Irradiated mixed-BM chimeras with wild-type and LSL-iOPN donor cells.
(a-d) BM cells obtained from WT (CD45.1) and LSL-iOPN (CD45.2) mice (a) were mixed at 1:1 ratio (b), and total of 107 mixed BM cells were transferred to lethally irradiated (900 rad) WT C57BL/6 mice (CD45.1/CD45.2). Three weeks after the transfer, cellularity of differentiated donor cells in the BM were analyzed by flow cytometry (c). As a positive control to show increased myeloid populations, Spp1−/− donor-derived cells in mixed BM chimera mice are shown in (d). Data is a representative of two independent experiments. Error bars denote ± SEM.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Tables 1 and 2, and Supplementary Note. (PDF 2318 kb)
Rights and permissions
About this article
Cite this article
Kanayama, M., Xu, S., Danzaki, K. et al. Skewing of the population balance of lymphoid and myeloid cells by secreted and intracellular osteopontin. Nat Immunol 18, 973–984 (2017). https://doi.org/10.1038/ni.3791
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3791
This article is cited by
-
Made to order: emergency myelopoiesis and demand-adapted innate immune cell production
Nature Reviews Immunology (2024)
-
Osteopontin in autoimmune disorders: current knowledge and future perspective
Inflammopharmacology (2022)
-
Hematopoietic progenitor cells as integrative hubs for adaptation to and fine-tuning of inflammation
Nature Immunology (2019)
-
The immunopathology of lung fibrosis: amphiregulin-producing pathogenic memory T helper-2 cells control the airway fibrotic responses by inducing eosinophils to secrete osteopontin
Seminars in Immunopathology (2019)
-
Osteopontin and the immune system: another brick in the wall
Cellular & Molecular Immunology (2018)