Abstract
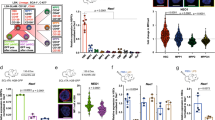
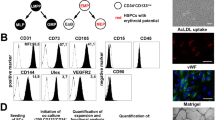
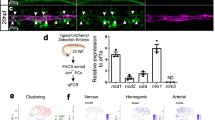
Endothelial cells establish an instructive vascular niche that reconstitutes haematopoietic stem and progenitor cells (HSPCs) through release of specific paracrine growth factors, known as angiocrine factors. However, the mechanism by which endothelial cells balance the rate of proliferation and lineage-specific differentiation of HSPCs is unknown. Here, we demonstrate that Akt activation in endothelial cells, through recruitment of mTOR, but not the FoxO pathway, upregulates specific angiocrine factors that support expansion of CD34−Flt3− KLS HSPCs with long-term haematopoietic stem cell (LT-HSC) repopulation capacity. Conversely, co-activation of Akt-stimulated endothelial cells with p42/44 MAPK shifts the balance towards maintenance and differentiation of the HSPCs. Selective activation of Akt1 in the endothelial cells of adult mice increased the number of colony forming units in the spleen and CD34−Flt3− KLS HSPCs with LT-HSC activity in the bone marrow, accelerating haematopoietic recovery. Therefore, the activation state of endothelial cells modulates reconstitution of HSPCs through the modulation of angiocrine factors, with Akt–mTOR-activated endothelial cells supporting the self-renewal of LT-HSCs and expansion of HSPCs, whereas MAPK co-activation favours maintenance and lineage-specific differentiation of HSPCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003).
Calvi, L. M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003).
Kollet, O. et al. Osteoclasts degrade endosteal components and promote mobilization of hematopoietic progenitor cells. Nat. Med. 12, 657–664 (2006).
Scadden, D. T. The stem-cell niche as an entity of action. Nature 441, 1075–1079 (2006).
Kiel, M. J. & Morrison, S. J. Uncertainty in the niches that maintain haematopoietic stem cells. Nat. Rev. Immunol. 8, 290–301 (2008).
Hooper, A. T. et al. Engraftment and reconstitution of hematopoiesis is dependent on VEGFR2-mediated regeneration of sinusoidal endothelial cells. Cell Stem Cell 4, 263–274 (2009).
Heissig, B. et al. Recruitment of stem and progenitor cells from the bone marrow niche requires MMP-9 mediated release of Kit-ligand. Cell 109, 625–637 (2002).
Lane, S. W., Scadden, D. T. & Gilliland, D. G. The leukemic stem cell niche: current concepts and therapeutic opportunities. Blood 114, 1150–1157 (2009).
Avecilla, S. T. et al. Chemokine-mediated interaction of hematopoietic progenitors with the bone marrow vascular niche is required for thrombopoiesis. Nat. Med. 10, 64–71 (2004).
Butler, J. M., Kobayashi, H. & Rafii, S. Instructive role of the vascular niche in promoting tumour growth and tissue repair by angiocrine factors. Nat. Rev. Cancer 10, 138–146 (2010).
Ding, B. et al. Inductive angiocrine signals from sinusoidal endothelium are required for liver regeneration. Nature doi:10.1038nature09493 (2010).
Butler, J. M. et al. Endothelial cells are essential for the self-renewal and repopulation of Notch-dependent hematopoietic stem cells. Cell Stem Cell 6, 251–264 (2010).
Libby, P., Ridker, P. M. & Maseri, A. Inflammation and atherosclerosis. Circulation 105, 1135–1143 (2002).
Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat. Med. 1, 27–31 (1995).
Fernandez, L. et al. Tumor necrosis factor-alpha and endothelial cells modulate Notch signaling in the bone marrow microenvironment during inflammation. Exp. Hematol. 36, 545–558 (2008).
Yeoh, J.S. et al. Fibroblast growth factor-1 and -2 preserve long-term repopulating ability of hematopoietic stem cells in serum-free cultures. Stem Cells 24, 1564–1572 (2006).
Seandel, M. et al. Generation of a functional and durable vascular niche by the adenoviral E4ORF1 gene. Proc. Natl Acad. Sci. USA 105, 19288–19293 (2008).
Shiojima, I. & Walsh, K. Role of Akt signaling in vascular homeostasis and angiogenesis. Circ. Res. 90, 1243–1250 (2002).
Phung, T. L. et al. Pathological angiogenesis is induced by sustained Akt signalling and inhibited by rapamycin. Cancer Cell 10, 159–170 (2006).
Pages, G. et al. Signaling angiogenesis via p42/p44 MAP kinase cascade. Ann. N. Y. Acad. Sci. 902, 187–200 (2000).
Matsunaga, T., Kato, T., Miyazaki, H. & Ogawa, M. Thrombopoietin promotes the survival of murine hematopoietic long-term reconstituting cells: comparison with the effects of FLT3/FLK-2 ligand and interleukin-6. Blood 92, 452–461 (1998).
Nemeth, M.J., Topol, L., Anderson, S.M., Yang, Y. & Bodine, D.M. Wnt5a inhibits canonical Wnt signaling in hematopoietic stem cells and enhances repopulation. Proc. Natl Acad. Sci. USA 104, 15436–15441 (2007).
Zhang, C. C. & Lodish, H. F. Insulin-like growth factor 2 expressed in a novel fetal liver cell population is a growth factor for hematopoietic stem cells. Blood 103, 2513–2521 (2004).
Humar, R., Kiefer, F.N., Berns, H., Resink, T.J. & Battegay, E.J. Hypoxia enhances vascular cell proliferation and angiogenesis in vitro via rapamycin (mTOR)-dependent signaling. FASEB J. 16, 771–780 (2002).
Potente, M. et al. Involvement of Foxo transcription factors in angiogenesis and postnatal neovascularization. J. Clin. Invest. 115, 2382–2392 (2005).
Sun, J.F. et al. Microvascular patterning is controlled by fine-tuning the Akt signal. Proc. Natl Acad. Sci. USA 102, 128–133 (2005).
Huynh, H. et al. Insulin-like growth factor-binding protein 2 secreted by a tumorigenic cell line supports ex vivo expansion of mouse hematopoietic stem cells. Stem Cells 26, 1628–1635 (2008).
Zhang, C. C., Kaba, M., Iizuka, S., Huynh, H. & Lodish, H. F. Angiopoietin-like 5 and IGFBP2 stimulate ex vivo expansion of human cord blood hematopoietic stem cells as assayed by NOD/SCID transplantation. Blood 111, 3415–3423 (2008).
Han, W., Ye, Q. & Moore, M. A. A soluble form of human Delta-like-1 inhibits differentiation of hematopoietic progenitor cells. Blood 95, 1616–1625 (2000).
Gering, M. & Patient, R. Hedgehog signaling is required for adult blood stem cell formation in zebrafish embryos. Dev. Cell 8, 389–400 (2005).
Goldman, D. C. et al. BMP4 regulates the hematopoietic stem cell niche. Blood 114, 4393–4401 (2009).
Fleming, H. E. et al. Wnt signaling in the niche enforces hematopoietic stem cell quiescence and is necessary to preserve self-renewal in vivo. Cell Stem Cell 2, 274–283 (2008).
Kopp, H.G. et al. Tie2 activation contributes to hemangiogenic regeneration after myelosuppression. Blood 106, 505–513 (2005).
Kopp, H.G. et al. Thrombospondins deployed by thrombopoietic cells determine angiogenic switch and extent of revascularization. J. Clin. Invest. 116, 3277–3291 (2006).
Dimmeler, S. & Zeiher, A. M. Akt takes center stage in angiogenesis signaling. Circ. Res. 86, 4–5 (2000).
Cross, M. J. & Claesson-Welsh, L. FGF and VEGF function in angiogenesis: signalling pathways, biological responses and therapeutic inhibition. Trends Pharmacol. Sci 22, 201–207 (2001).
Rafii, S. et al. Isolation and characterization of human bone marrow microvascular endothelial cells: hematopoietic progenitor cell adhesion. Blood 84, 10–19 (1994).
Tang, E. D., Nunez, G., Barr, F. G. & Guan, K. L. Negative regulation of the forkhead transcription factor FKHR by Akt. J. Biol. Chem. 274, 16741–16746 (1999).
Sarbassov, D. D., Guertin, D. A., Ali, S. M. & Sabatini, D. M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–1101 (2005).
Acknowledgements
S.R. is supported by Howard Hughes Medical Institute; Ansary Stem Cell Institute; National Institute of Health grants HL097797, U01 HL66592-03, RC1 AI080309; Qatar National Priorities Research Program; Anbinder and Newmans Own Foundations; Empire State Stem Cell Board and the New York State Department of Health grant NYS C024180. We thank G. Lam (Weill Cornell Medical College) for HUVEC culture.
Author information
Authors and Affiliations
Contributions
S.R. designed the project. H.K., J.M.B. and S.R. designed experiments and wrote the paper. H.K. and J.M.B. performed most of the data collection and data analysis. M.K. and B.B. performed a significant amount of the experimental work. R.D. and L.B. generated and provided myrAkt1 mouse. B.D., D.N., V.C. and K.S. analysed microarray data and provided advice. The work was carried out in the laboratory of S.R.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 845 kb)
Rights and permissions
About this article
Cite this article
Kobayashi, H., Butler, J., O'Donnell, R. et al. Angiocrine factors from Akt-activated endothelial cells balance self-renewal and differentiation of haematopoietic stem cells. Nat Cell Biol 12, 1046–1056 (2010). https://doi.org/10.1038/ncb2108
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2108
This article is cited by
-
Hydrogel-based microenvironment engineering of haematopoietic stem cells
Cellular and Molecular Life Sciences (2023)
-
Engineering a niche supporting hematopoietic stem cell development using integrated single-cell transcriptomics
Nature Communications (2022)
-
WNT5A from the fetal liver vascular niche supports human fetal liver hematopoiesis
Stem Cell Research & Therapy (2021)
-
Endothelial Jak3 expression enhances pro-hematopoietic angiocrine function in mice
Communications Biology (2021)
-
Perivascular Secretome Influences Hematopoietic Stem Cell Maintenance in a Gelatin Hydrogel
Annals of Biomedical Engineering (2021)