Abstract
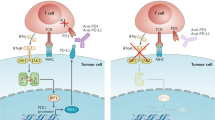
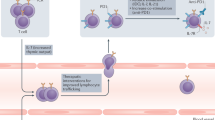
Recent clinical trials using immunotherapy have demonstrated its potential to control cancer by disinhibiting the immune system. Immune checkpoint blocking (ICB) antibodies against cytotoxic-T-lymphocyte-associated protein 4 or programmed cell death protein 1/programmed death-ligand 1 have displayed durable clinical responses in various cancers1. Although these new immunotherapies have had a notable effect on cancer treatment, multiple mechanisms of immune resistance exist in tumours. Among the key mechanisms, myeloid cells have a major role in limiting effective tumour immunity2,3,4. Growing evidence suggests that high infiltration of immune-suppressive myeloid cells correlates with poor prognosis and ICB resistance5,6. These observations suggest a need for a precision medicine approach in which the design of the immunotherapeutic combination is modified on the basis of the tumour immune landscape to overcome such resistance mechanisms. Here we employ a pre-clinical mouse model system and show that resistance to ICB is directly mediated by the suppressive activity of infiltrating myeloid cells in various tumours. Furthermore, selective pharmacologic targeting of the gamma isoform of phosphoinositide 3-kinase (PI3Kγ), highly expressed in myeloid cells, restores sensitivity to ICB. We demonstrate that targeting PI3Kγ with a selective inhibitor, currently being evaluated in a phase 1 clinical trial (NCT02637531), can reshape the tumour immune microenvironment and promote cytotoxic-T-cell-mediated tumour regression without targeting cancer cells directly. Our results introduce opportunities for new combination strategies using a selective small molecule PI3Kγ inhibitor, such as IPI-549, to overcome resistance to ICB in patients with high levels of suppressive myeloid cell infiltration in tumours.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Khalil, D. N., Smith, E. L., Brentjens, R. J. & Wolchok, J. D. The future of cancer treatment: immunomodulation, CARs and combination immunotherapy. Nat. Rev. Clin. Oncol. 13, 273–290 (2016)
Coussens, L. M., Zitvogel, L. & Palucka, A. K. Neutralizing tumor-promoting chronic inflammation: a magic bullet? Science 339, 286–291 (2013)
Gajewski, T. F., Schreiber, H. & Fu, Y.-X. Innate and adaptive immune cells in the tumor microenvironment. Nat. Immunol. 14, 1014–1022 (2013)
Schmid, M. C. & Varner, J. A. Myeloid cells in the tumor microenvironment: modulation of tumor angiogenesis and tumor inflammation. J. Oncol. 2010, 201026 (2010)
Diaz-Montero, C. M., Finke, J. & Montero, A. J. Myeloid-derived suppressor cells in cancer: therapeutic, predictive, and prognostic implications. Semin. Oncol. 41, 174–184 (2014)
Gebhardt, C. et al. Myeloid cells and related chronic inflammatory factors as novel predictive markers in melanoma treatment with Ipilimumab. Clin. Cancer Res. 21, 5453–5459 (2015)
De Vlaeminck, Y., González-Rascón, A., Goyvaerts, C. & Breckpot, K. Cancer-associated myeloid regulatory cells. Front. Immunol. 7, 113 (2016)
Highfill, S. L. et al. Disruption of CXCR2-mediated MDSC tumor trafficking enhances anti-PD1 efficacy. Sci. Transl. Med . 6, 237ra67 (2014)
Meyer, C. et al. Frequencies of circulating MDSC correlate with clinical outcome of melanoma patients treated with ipilimumab. Cancer Immunol. Immunother. 63, 247–257 (2014)
Bjoern, J. et al. Immunological correlates of treatment and response in stage IV malignant melanoma patients treated with Ipilimumab. OncoImmunology 5, e1100788 (2015)
Holmgaard, R. B., Zamarin, D., Lesokhin, A., Merghoub, T. & Wolchok, J. D. Targeting myeloid-derived suppressor cells with colony stimulating factor-1 receptor blockade can reverse immune resistance to immunotherapy in indoleamine 2,3-dioxygenase-expressing tumors. EBioMedicine 6, 50–58 (2016)
Ugel, S., De Sanctis, F., Mandruzzato, S. & Bronte, V. Tumor-induced myeloid deviation: when myeloid-derived suppressor cells meet tumor-associated macrophages. J. Clin. Invest . 125, 3365–3376 (2015)
Gabrilovich, D. Mechanisms and functional significance of tumour-induced dendritic-cell defects. Nat. Rev. Immunol. 4, 941–952 (2004)
Lesokhin, A. M. et al. Monocytic CCR2+ myeloid-derived suppressor cells promote immune escape by limiting activated CD8 T-cell infiltration into the tumor microenvironment. Cancer Res. 72, 876–886 (2012)
Kaneda, M. M. et al. PI3Kγ is a molecular switch that controls immune suppression. Nature http://dx.doi.org/10.1038/nature19834 (2016)
Hirsch, E. et al. Central role for G protein-coupled phosphoinositide 3-kinase γ in inflammation. Science 287, 1049–1053 (2000)
Li, Z. Roles of PLC-β2 and -β3 and PI3Kγ in chemoattractant-mediated signal transduction. Science 287, 1046–1049 (2000)
Schmid, M. C. et al. Receptor tyrosine kinases and TLR/IL1Rs unexpectedly activate myeloid cell PI3kγ, a single convergent point promoting tumor inflammation and progression. Cancer Cell 19, 715–727 (2011)
Evans, C. A. et al. Discovery of a selective phosphoinositide-3-kinase (PI3K)-γ inhibitor (IPI-549) as an immuno-oncology clinical candidate. ACS Med. Chem. Lett . 7, 862–867 (2016)
Laoui, D. et al. Tumor hypoxia does not drive differentiation of tumor-associated macrophages but rather fine-tunes the M2-like macrophage population. Cancer Res. 74, 24–30 (2014)
Movahedi, K. et al. Different tumor microenvironments contain functionally distinct subsets of macrophages derived from Ly6C(high) monocytes. Cancer Res. 70, 5728–5739 (2010)
Larkin, J. et al. Combined Nivolumab and Ipilimumab or monotherapy in untreated melanoma. N. Engl. J. Med. 373, 23–34 (2015)
Bindea, G. et al. Spatiotemporal dynamics of intratumoral immune cells reveal the immune landscape in human cancer. Immunity 39, 782–795 (2013)
Overwijk, W. W. et al. Tumour regression and autoimmunity after reversal of a functionally tolerant state of self-reactive CD8+ T cells. J. Exp. Med. 198, 569–580 (2003)
Winkler, D. G. et al. PI3K-δ and PI3K-γ inhibition by IPI-145 abrogates immune responses and suppresses activity in autoimmune and inflammatory disease models. Chem. Biol. 20, 1364–1374 (2013)
Acknowledgements
We would like to thank the Flow Cytometry and Integrated Genomics Operation Core Facilities at MSKCC. Swim Across America, Ludwig Institute for Cancer Research, Parker Institute for Cancer Immunotherapy, Center for Experimental Therapeutics at MSKCC (ETC), and the Breast Cancer Research Foundation supported this work. The work was also supported in part by the MSKCC Core Grant (P30 CA008748). O.D.H. was supported by J. Houtard foundation, Nuovo Soldati Foundation and Wallonie-Bruxelles International. We would also like to thank Y. Senbabaoglu for his help in bioinformatics data analysis, A. Bossert for his contribution as part of the GME program as well as J. Gladstone and K. Walsh for their contributions while working as co-op students in the laboratory.
Author information
Authors and Affiliations
Contributions
O.D.H., T.M., J.D.W., K.M., J.L.K, V.J.P. and J.A. developed the concepts and discussed experiments. O.D.H., T.M., J.D.W., K.M. and J.L.K. wrote the manuscript. O.D.H., M.R., D.W., L.F.C., D.H.C., S.B., A.G., M.P., J.P. and N.K. performed and analysed animal model experiments, flow cytometry experiments and functional assays. C.L. provided technical assistance; S.S. and K.W. performed assays in human samples. M.D., T.T. and H.S. performed transcriptomic analysis. J.T. and J.S. performed pharmacodynamics and pharmacokinetics studies.
Corresponding authors
Ethics declarations
Competing interests
All authors with affiliation to Infinity Pharmaceuticals, Inc. were employees and shareholders at Infinity Pharmaceuticals, Inc. at the time of the study. All other authors have no competing interests.
Additional information
Reviewer Information Nature thanks F. Balkwill, M. De Palma and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Figure 1 Effect of suppressive myeloid TILs in response to checkpoint blockade.
a, Individual tumour growth of subcutaneous (4T1) or intradermal (B16, B16-GMCSF) implants in anti-PD-1-, anti-CTLA4- or control-treated mice (n = 10). b, In vitro suppressive activity of tumour-infiltrating CD11b+ cells purified from spleen of 4T1, B16, B16-GMCSF tumour-bearing mice. Representative histograms of CD8+ T cell proliferation at corresponding CD11b+ to CD8+ T cell ratio (left panel) and quantification of CD8+ T cell proliferation (right panel) (n = 3), mean ± s.e.m. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 (non-parametric Mann–Whitney U-test).
Extended Data Figure 2 Effect of selective PI3Kγ inhibition on tumour growth and myeloid TILs.
a, Binding affinities (Kd) and cellular IC50 inhibition of pAKT by IPI-549 for class I PI3K isoforms (left table). Percentage of inhibition of expression on bone-marrow-derived macrophages polarized with M-CSF and IL-4, (right panel). b, Percentage of tumour growth inhibition in LLC, MC38, 4T1, CT26, B16-GMCSF tumour-bearing mice treated with IPI-549 (table). c, Quantification of CD11b, CD206, NOS2 and PD-L1 expression in CD11b+ tumour-infiltrating leukocytes from IPI-549- versus vehicle-treated CT26 tumour-bearing mice. d, RNA-seq of co-stimulatory and checkpoint molecules on whole tumours from CT26 tumour-bearing mice treated for 6 or 9 days with IPI-549 compared to vehicle. e, Mean tumour volume of subcutaneous LLC-Brei implants in IPI-549- versus vehicle-treated mice without or after CD11b+ cell depletion. Data represent analysis of 5–10 mice per group, mean ± s.e.m. *P < 0.05, ***P < 0.001 (non-parametric Mann–Whitney U-test).
Extended Data Figure 3 Effect of selective PI3Kγ inhibition on subsets of CD11b myeloid cells.
a, Representative flow cytometry analysis and quantification of Ly6C, MHC class II expression in CD11b+Ly6G− cells infiltrating 4T1 tumours. b, mRNA expression of selected M1 and M2 markers in sorted Ly6ClowMHCIIlow (TAM-M2) compared to Ly6ClowMHCIIhigh (TAM-M1) population from 4T1 tumour, data were relative to GAPDH expression and normalized versus the mean of TAM-M1 population. Mean ± s.e.m. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 (non-parametric Mann–Whitney U-test).
Extended Data Figure 4 Effect of selective PI3Kγ inhibition on suppressive PBMC derived human myeloid cells.
a, Inhibition of CXCL-12 activation of PI3Kγ in monocytes as measured by pAKT (S473) in human whole blood. b, Representative histograms and quantification of human CD8+ T cell proliferation after 72 h of co-culture with or without autologous myeloid-derived suppressor cells generated from the T-cell-depleted PBMCs ± IPI-549.
Extended Data Figure 5 Effect of selective PI3Kγ inhibition on function of tumour specific T cell responses.
a, Quantification of KI67, and CTLA4 expression in CD8+ T cells in TILs of 4T1 or B16-GMCSF tumours at IPI-549 compared to vehicle treatment days 7 and 14. b, Mean tumour volume of subcutaneous 4T1 tumour in IPI-549- versus vehicle-treated BALB/c NU/NU mice (n = 10). c, Mean tumour volume of subcutaneous CT-26 tumour in IPI-549- versus vehicle-treated BALB/c mice with or without CD8+ T cell depletion by anti-CD8 antibody (n = 10). d, Quantification and representative pictures of CT26 tumour-specific immune responses in PBMCs from CT26 tumour-bearing mice treated with IPI-549 in comparison to vehicle by ELISPOT. PBMCs were collected from tumour-bearing animals after 10 days of vehicle or IPI-549 treatment and restimulated overnight with irradiated CT26 or 4T1 stimulator cells.
Extended Data Figure 6 Effect of selective PI3Kγ inhibition on the differentiation of T cells in tumours.
a, Representative flow cytometry analysis and quantification of CD62L and CD44 expression in CD8+ and CD4+ T cell infiltrates in tumour, lymph node (LN) and spleen of 4T1 tumour-bearing mice treated with IPI-549 compared to vehicle. Data represent analyses of five mice per group, mean ± s.e.m. NS, not significant (non-parametric Mann–Whitney U-test).
Extended Data Figure 7 Effect of combination of a selective PI3Kγ inhibitor with checkpoint blockade on various tumours.
a, Survival to 2,000 mm3 tumour volume of LLC Brei tumour in IPI-549- or vehicle-treated mice in combination with or without anti-CTLA4 (vehicle and IPI-549 groups, n = 14; anti-CTLA4, n = 13; IPI-549 and anti-CTLA4 combination, n = 10). b, Mean tumour volume of CT26 tumour in IPI-549- or vehicle-treated mice in combination with or without anti-PD-L1 (n = 15 for all groups except vehicle, n = 13).
Extended Data Figure 8 Effect of combination of a selective PI3Kγ inhibitor with checkpoint blockade on TILs.
a, Mean tumour volume of subcutaneous 4T1 tumour-bearing mice treated with IPI-549, vehicle or anti-PD-1 in combination with IPI-549 or vehicle (n = 10). b, Representative flow cytometry analysis of CD206 and MHCII labelling in CD11b+ F4/80+ cell populations in the different treatment groups of 4T1 tumour-bearing mice. c, Quantification of CD11b+F4/80+, M1/M2 ratio, CD8+/Treg in TILs and granzyme B expression in CD8+ T cells from 4T1 tumours in the different treatment groups. d, Quantification of CD11b+F4/80+, M1/M2 ratio, CD8+/Treg in TILs and granzyme B expression in CD8+ T cells from B16-GMCSF tumours in the different treatment groups, mean ± s.e.m. *P < 0.05, **P < 0.01 (non-parametric Mann–Whitney U-test).
Extended Data Figure 9 Effect of combination of a selective PI3Kγ inhibitor with checkpoint blockade on acquisition of anti-tumour memory.
a, Tumour re-challenge at 100 days (from first tumour implant) following primary tumour complete response in B16-GMCSF tumour-bearing mice treated with vehicle (blue) or IPI-549 (red) in combination with both anti-PD1 and anti-CTLA4. b, CT26 tumour-bearing mice with complete responses in the anti-PD-1 treatment group and the IPI-549 plus anti-PD-1 combination treatment group were re-challenged with CT26 tumour implant. Additional mice with complete responses from the IPI-549 plus anti-PD-1 combination were implanted with 4T1 tumours. There was a low or no tumour take with CT26 re-challenge, while all 4T1 tumours grew, indicating specific anti-tumour memory.
Rights and permissions
About this article
Cite this article
De Henau, O., Rausch, M., Winkler, D. et al. Overcoming resistance to checkpoint blockade therapy by targeting PI3Kγ in myeloid cells. Nature 539, 443–447 (2016). https://doi.org/10.1038/nature20554
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature20554
This article is cited by
-
Prognosis and immunotherapy response prediction based on M2 macrophage-related genes in colon cancer
Journal of Cancer Research and Clinical Oncology (2024)
-
Japanese phase Ib study of the oral PI3K-δ and -γ inhibitor duvelisib in patients with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma
International Journal of Hematology (2024)
-
The Streptococcus virulence protein PepO triggers anti-tumor immune responses by reprograming tumor-associated macrophages in a mouse triple negative breast cancer model
Cell & Bioscience (2023)
-
SIRPα blockade improves the antitumor immunity of radiotherapy in colorectal cancer
Cell Death Discovery (2023)
-
TLR5 agonists enhance anti-tumor immunity and overcome resistance to immune checkpoint therapy
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.