Abstract
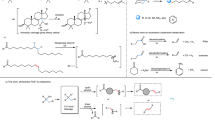
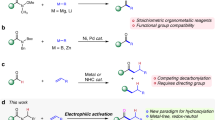
The removal of two vicinal hydrogen atoms from an alkane to produce an alkene is a challenge for synthetic chemists1,2. In nature, desaturases and acetylenases are adept at achieving this essential oxidative functionalization reaction, for example during the biosynthesis of unsaturated fatty acids3, eicosanoids, gibberellins4 and carotenoids5. Alkane-to-alkene conversion almost always involves one or more chemical intermediates in a multistep reaction pathway; these may be either isolable species (such as alcohols or alkyl halides) or reactive intermediates (such as carbocations, alkyl radicals, or σ-alkyl-metal species). Here we report a desaturation reaction of simple, unactivated alkanes that is mechanistically unique. We show that benzynes are capable of the concerted removal of two vicinal hydrogen atoms from a hydrocarbon. The discovery of this exothermic, net redox process was enabled by the simple thermal generation of reactive benzyne intermediates through the hexadehydro-Diels–Alder cycloisomerization reaction of triyne substrates6. We are not aware of any single-step, bimolecular reaction in which two hydrogen atoms are simultaneously transferred from a saturated alkane. Computational studies indicate a preferred geometry with eclipsed vicinal C–H bonds in the alkane donor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Linstead, R. P., Braude, E. A., Mitchell, P. W. D., Wooldridge, K. R. H. & Jackman, L. M. Transfer of hydrogen in organic systems. Nature 169, 100–103 (1952)
Bloomfield, D. K. & Bloch, K. The formation of Δ9-unsaturated fatty acids. J. Biol. Chem. 235, 337–345 (1960)
Buist, P. H. Fatty acid desaturases: selecting the dehydrogenation channel. Nat. Prod. Rep. 21, 249–262 (2004)
Bhattacharya, A. et al. Characterization of the fungal gibberellin desaturase as a 2-oxoglutarate-dependent dioxygenase and its utilization for enhancing plant growth. Plant Physiol. 160, 837–845 (2012)
Moran, N. A. & Jarvik, T. Lateral transfer of genes from fungi underlies carotenoid production in aphids. Science 328, 624–627 (2010)
Hoye, T. R., Baire, B., Niu, D., Willoughby, P. H. & Woods, B. P. The hexadehydro-Diels–Alder reaction. Nature 490, 208–212 (2012)
Hoffmann, R. W. Dehydrobenzene and Cycloalkynes (Organic Chemistry Vol. 11) (Academic, 1967)
Tadross, P. M. & Stoltz, B. M. A comprehensive history of arynes in natural product total synthesis. Chem. Rev. 112, 3550–3577 (2012)
Baire, B., Niu, D., Willoughby, P. H., Woods, B. P. & Hoye, T. R. Synthesis of complex benzenoids via the intermediate generation of o-benzynes through the hexadehydro-Diels–Alder reaction. Nature Protocols 8, 501–508 (2013)
Miyawaki, K., Suzuki, R., Kawano, T. & Ueda, I. Cycloaromatization of a non-conjugated polyenyne system: synthesis of 5H-benzo[d]fluoreno[3,2-b]pyrans via diradicals generated from 1-[2-{4-(2-alkoxymethylphenyl)butan-1,3-diynyl}]phenylpentan-2,4-diyn-l-ols and trapping evidence for the 1,2-didehydrobenzene diradical. Tetrahedr. Lett. 38, 3943–3946 (1997)
Bradley, A. Z. & Johnson, R. P. Thermolysis of 1,3,8-nonatriyne: evidence for intramolecular [2+4] cycloaromatization to a benzyne intermediate. J. Am. Chem. Soc. 119, 9917–9918 (1997)
Ajaz, A. et al. Concerted vs. stepwise mechanisms in dehydro-Diels–Alder reactions. J. Org. Chem. 76, 9320–9328 (2011)
Hoffmann, R. W. & Suzuki, K. A ‘hot, energized’ benzyne. Angew. Chem. Int. Ed. 52, 2–4 (2013)
Yun, S. Y., Wang, K.-P., Lee, N.-K., Mamidipalli, P. & Lee, D. Alkane C–H insertion by aryne intermediates with a silver catalyst. J. Am. Chem. Soc. 135, 4668–4671 (2013)
Voica, A.-F., Mendoza, A., Gutekunst, W. R., Fraga, J. O. & Baran, P. S. Guided desaturation of unactivated aliphatics. Nature Chem. 4, 629–635 (2012)
Tsui, J. A. & Sterenberg, B. T. A metal-templated 4 + 2 cycloaddition reaction of an alkyne and a diyne to form a 1,2-aryne. Organometallics 28, 4906–4908 (2009)
de Almeida, G., Townsend, L. C. & Bertozzi, C. R. Synthesis and reactivity of dibenzoselenacycloheptynes. Org. Lett. 15, 3038–3041 (2013)
Hünig, S., Müller, H. & Thier, W. Reduktionen mit diimid. Tetrahedr. Lett. 2, 353–357 (1961)
Corey, E. J., Pasto, D. J. & Mock, W. L. Chemistry of diimide. II. Stereochemistry of hydrogen transfer to carbon–carbon multiple bonds. J. Am. Chem. Soc. 83, 2957–2958 (1961)
Fernández, I., Cossío, F. P. & Sierra, M. A. Dyotropic reactions: mechanisms and synthetic applications. Chem. Rev. 109, 6687–6711 (2009)
Fernández, I., Sierra, M. A. & Cossío, F. P. In-plane aromaticity in double-group transfer reactions. J. Org. Chem. 72, 1488–1491 (2007)
Hoye, T. R., Eklov, B. M., Ryba, T. D., Voloshin, M. & Yao, L. J. No-D NMR (no deuterium proton NMR) spectroscopy: a simple yet powerful method for analyzing reaction and reagent solutions. Org. Lett. 6, 953–956 (2004)
Himeshima, Y., Sonoda, T. & Kobayashi, H. Fluoride-induced 1,2-elimination of o-trimethylsilylphenyl triflate to benzyne under mild conditions. Chem. Lett. 12, 1211–1214 (1983)
Ma, Z.-X., Feltenberger, J. B. & Hsung, R. P. Total syntheses of chelidonine and norchelidonine via an enamide–benzyne–[2+2] cycloaddition cascade. Org. Lett. 14, 2742–2745 (2012)
Sumida, Y., Kato, T. & Hosoya, T. Generation of arynes via ate complexes of arylboronic esters with an ortho-leaving group. Org. Lett. 15, 2806–2809 (2013)
Davies, H. M. L., Du Bois, J. & Yu, J.-Q. C–H functionalization in organic synthesis. Chem. Soc. Rev. 40, 1855–1856 (2011)
Paul, G. F., Jak, B. & Lankin, D. A. Routine experimental protocol for qHNMR illustrated with taxol. J. Nat. Prod. 70, 589–595 (2007)
Acknowledgements
We thank C. J. Cramer for helpful discussions about the computational studies. D.N. and P.H.W. thank the University of Minnesota Graduate School Doctoral Dissertation Fellowship and National Science Foundation Graduate Research Fellowship program, respectively. Financial support from the National Institute of General Medical Sciences (GM65597) and the National Cancer Institute (CA76497) of the US Department of Health and Human Services is acknowledged. Portions of this work were performed with hardware and software resources available through the University of Minnesota Supercomputing Institute (MSI).
Author information
Authors and Affiliations
Contributions
D.N. made the initial key observations and performed most of the scope studies. P.H.W. performed most of the mechanistic studies. B.P.W. and B.B. also performed aspects of the experimental work. All authors interpreted the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains General Experimental Protocols; Preparation procedures and Characterization data for all new compounds; Computational details for double hydrogen atom transfer reactions; Supplementary References and Copies of 1H and 13C NMR spectra of all new compounds (see Contents list for details). (PDF 4471 kb)
Rights and permissions
About this article
Cite this article
Niu, D., Willoughby, P., Woods, B. et al. Alkane desaturation by concerted double hydrogen atom transfer to benzyne. Nature 501, 531–534 (2013). https://doi.org/10.1038/nature12492
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12492
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.