Abstract
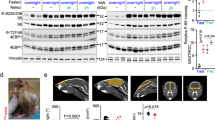
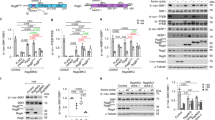
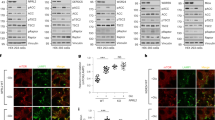
The mechanistic target of rapamycin complex 1 (mTORC1) pathway regulates organismal growth in response to many environmental cues, including nutrients and growth factors1. Cell-based studies showed that mTORC1 senses amino acids through the RagA–D family of GTPases2,3 (also known as RRAGA, B, C and D), but their importance in mammalian physiology is unknown. Here we generate knock-in mice that express a constitutively active form of RagA (RagAGTP) from its endogenous promoter. RagAGTP/GTP mice develop normally, but fail to survive postnatal day 1. When delivered by Caesarean section, fasted RagAGTP/GTP neonates die almost twice as rapidly as wild-type littermates. Within an hour of birth, wild-type neonates strongly inhibit mTORC1, which coincides with profound hypoglycaemia and a decrease in plasma amino-acid concentrations. In contrast, mTORC1 inhibition does not occur in RagAGTP/GTP neonates, despite identical reductions in blood nutrient amounts. With prolonged fasting, wild-type neonates recover their plasma glucose concentrations, but RagAGTP/GTP mice remain hypoglycaemic until death, despite using glycogen at a faster rate. The glucose homeostasis defect correlates with the inability of fasted RagAGTP/GTP neonates to trigger autophagy and produce amino acids for de novo glucose production. Because profound hypoglycaemia does not inhibit mTORC1 in RagAGTP/GTP neonates, we considered the possibility that the Rag pathway signals glucose as well as amino-acid sufficiency to mTORC1. Indeed, mTORC1 is resistant to glucose deprivation in RagAGTP/GTP fibroblasts, and glucose, like amino acids, controls its recruitment to the lysosomal surface, the site of mTORC1 activation. Thus, the Rag GTPases signal glucose and amino-acid concentrations to mTORC1, and have an unexpectedly key role in neonates in autophagy induction and thus nutrient homeostasis and viability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zoncu, R., Efeyan, A. & Sabatini, D. M. mTOR: from growth signal integration to cancer, diabetes and ageing. Nature Rev. Mol. Cell Biol. 12, 21–35 (2010)
Kim, E., Goraksha-Hicks, P., Li, L., Neufeld, T. P. & Guan, K. L. Regulation of TORC1 by Rag GTPases in nutrient response. Nature Cell Biol. 10, 935–945 (2008)
Sancak, Y. et al. The Rag GTPases bind raptor and mediate amino acid signaling to mTORC1. Science 320, 1496–1501 (2008)
Sancak, Y. et al. Ragulator-Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell 141, 290–303 (2010)
Zoncu, R. et al. mTORC1 senses lysosomal amino acids through an inside-out mechanism that requires the vacuolar H-ATPase. Science 334, 678–683 (2011)
Hirose, E., Nakashima, N., Sekiguchi, T. & Nishimoto, T. RagA is a functional homologue of S. cerevisiae Gtr1p involved in the Ran/Gsp1-GTPase pathway. J. Cell Sci. 111, 11–21 (1998)
Kwiatkowski, D. J. et al. A mouse model of TSC1 reveals sex-dependent lethality from liver hemangiomas, and up-regulation of p70S6 kinase activity in Tsc1 null cells. Hum. Mol. Genet. 11, 525–534 (2002)
Zhang, H. et al. Loss of Tsc1/Tsc2 activates mTOR and disrupts PI3K-Akt signaling through downregulation of PDGFR. J. Clin. Invest. 112, 1223–1233 (2003)
Scheuner, D. et al. Translational control is required for the unfolded protein response and in vivo glucose homeostasis. Mol. Cell 7, 1165–1176 (2001)
Birsoy, K. et al. Analysis of gene networks in white adipose tissue development reveals a role for ETS2 in adipogenesis. Development 138, 4709–4719 (2011)
Kuma, A. et al. The role of autophagy during the early neonatal starvation period. Nature 432, 1032–1036 (2004)
Komatsu, M. et al. Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice. J. Cell Biol. 169, 425–434 (2005)
Mizushima, N., Levine, B., Cuervo, A. M. & Klionsky, D. J. Autophagy fights disease through cellular self-digestion. Nature 451, 1069–1075 (2008)
Mortimore, G. E. & Schworer, C. M. Induction of autophagy by amino-acid deprivation in perfused rat liver. Nature 270, 174–176 (1977)
Kroemer, G., Marino, G. & Levine, B. Autophagy and the integrated stress response. Mol. Cell 40, 280–293 (2010)
Roczniak-Ferguson, A. et al. The transcription factor TFEB links mTORC1 signaling to transcriptional control of lysosome homeostasis. Sci. Signal. 5, ra42 (2012)
Settembre, C. et al. A lysosome-to-nucleus signalling mechanism senses and regulates the lysosome via mTOR and TFEB. EMBO J. 31, 1095–1108 (2012)
Kalender, A. et al. Metformin, independent of AMPK, inhibits mTORC1 in a rag GTPase-dependent manner. Cell Metab. 11, 390–401 (2010)
Choo, A. Y. et al. Glucose addiction of TSC null cells is caused by failed mTORC1-dependent balancing of metabolic demand with supply. Mol. Cell 38, 487–499 (2010)
Shaw, R. J. et al. The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc. Natl Acad. Sci. USA 101, 3329–3335 (2004)
Proud, C. G. eIF2 and the control of cell physiology. Semin. Cell Dev. Biol. 16, 3–12 (2005)
Guerra, C. et al. Tumor induction by an endogenous K-ras oncogene is highly dependent on cellular context. Cancer Cell 4, 111–120 (2003)
Lo, S., Russell, J. C. & Taylor, A. W. Determination of glycogen in small tissue samples. J. Appl. Physiol. 28, 234–236 (1970)
Acknowledgements
We thank members of the Sabatini laboratory for suggestions, and A. Hutchins for technical assistance. We thank R. Shaw for providing the AMPK-DKO MEFs, D. Ron for the GCN2-KO MEFs and M. Barbacid for the transcriptional STOP cassette. This work was supported by grants from the National Institutes of Health (R01 CA129105, R01 CA103866 and R37 AI047389) and awards from the American Federation for Aging, Starr Foundation, Koch Institute Frontier Research Program, and the Ellison Medical Foundation to D.M.S., fellowships from the Human Frontiers Science Program to A.E., and the Jane Coffin Childs Memorial Fund for Medical Research and the LAM Foundation to R.Z. D.M.S. in an investigator of Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
A.E. and D.M.S. conceived the project. A.E. designed and performed most experiments with input from D.M.S, and assistance from S.C., R.L.W. and O.K. R.Z. performed experiments and participated in discussion of the results. I.G., H.S. and D.D.S. performed electron microscopy experiments and interpretations. D.D.S. helped with discussion and interpretation of results. A.E. wrote and D.M.S. edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-4. (PDF 2779 kb)
Rights and permissions
About this article
Cite this article
Efeyan, A., Zoncu, R., Chang, S. et al. Regulation of mTORC1 by the Rag GTPases is necessary for neonatal autophagy and survival. Nature 493, 679–683 (2013). https://doi.org/10.1038/nature11745
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11745
This article is cited by
-
AMPK knocks at the gate of GATOR
Nature Metabolism (2023)
-
Multifaceted role of mTOR (mammalian target of rapamycin) signaling pathway in human health and disease
Signal Transduction and Targeted Therapy (2023)
-
Malonyl-CoA is a conserved endogenous ATP-competitive mTORC1 inhibitor
Nature Cell Biology (2023)
-
RAGA prevents tumor immune evasion of LUAD by promoting CD47 lysosome degradation
Communications Biology (2023)
-
Amino acid availability governs mTOR ubiquitination
Cell Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.