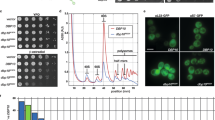
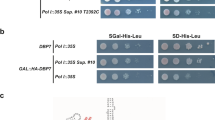
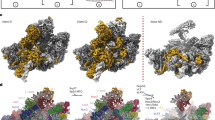
Abstract
Superfamily 1 and superfamily 2 RNA helicases are ubiquitous messenger-RNA–protein complex (mRNP) remodelling enzymes that have critical roles in all aspects of RNA metabolism1,2. The superfamily 2 DEAD-box ATPase Dbp5 (human DDX19) functions in mRNA export and is thought to remodel mRNPs at the nuclear pore complex (NPC)3,4,5,6,7,8. Dbp5 is localized to the NPC via an interaction with Nup159 (NUP214 in vertebrates)3,4,5,8,9 and is locally activated there by Gle1 together with the small-molecule inositol hexakisphosphate (InsP6)10,11. Local activation of Dbp5 at the NPC by Gle1 is essential for mRNA export in vivo10,12; however, the mechanistic role of Dbp5 in mRNP export is poorly understood and it is not known how Gle1InsP6 and Nup159 regulate the activity of Dbp5. Here we report, from yeast, structures of Dbp5 in complex with Gle1InsP6, Nup159/Gle1InsP6 and RNA. These structures reveal that InsP6 functions as a small-molecule tether for the Gle1–Dbp5 interaction. Surprisingly, the Gle1InsP6–Dbp5 complex is structurally similar to another DEAD-box ATPase complex essential for translation initiation, eIF4G–eIF4A, and we demonstrate that Gle1InsP6 and eIF4G both activate their DEAD-box partner by stimulating RNA release. Furthermore, Gle1InsP6 relieves Dbp5 autoregulation and cooperates with Nup159 in stabilizing an open Dbp5 intermediate that precludes RNA binding. These findings explain how Gle1InsP6, Nup159 and Dbp5 collaborate in mRNA export and provide a general mechanism for DEAD-box ATPase regulation by Gle1/eIF4G-like activators.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Primary accessions
Protein Data Bank
Data deposits
Coordinates and structure factors for Dbp5–CTD(L327V)/InsP6/Gle1(H337R), Dbp5–CTD(L327V)/InsP6/Gle1(WT), Δ90Dbp5(L327V)/RNA/ADP•BeF;3, Δ90Dbp5/RNA/ADP•BeF;3, Δ90Dbp5(L327V)/InsP6/Gle1(H337R)/ADP and Δ90Dbp5(L327V)/InsP6/Gle1(H337R)/ADP/Nup159 have been deposited in the protein data bank under accession numbers 3PEU, 3PEV, 3PEW, 3PEY, 3PEX and 3PEZ, respectively.
References
Cordin, O., Banroques, J., Tanner, N. K. & Linder, P. The DEAD-box protein family of RNA helicases. Gene 367, 17–37 (2006)
Jankowsky, E. RNA helicases at work: binding and rearranging. Trends Biochem. Sci. 36, 19–29 (2010)
Schmitt, C. et al. Dbp5, a DEAD-box protein required for mRNA export, is recruited to the cytoplasmic fibrils of nuclear pore complex via a conserved interaction with CAN/Nup159p. EMBO J. 18, 4332–4347 (1999)
Hodge, C. A., Colot, H. V., Stafford, P. & Cole, C. N. Rat8p/Dbp5p is a shuttling transport factor that interacts with Rat7p/Nup159p and Gle1p and suppresses the mRNA export defect of xpo1-1 cells. EMBO J. 18, 5778–5788 (1999)
Snay-Hodge, C. A., Colot, H. V., Goldstein, A. L. & Cole, C. N. Dbp5p/Rat8p is a yeast nuclear pore-associated DEAD-box protein essential for RNA export. EMBO J. 17, 2663–2676 (1998)
Lund, M. K. & Guthrie, C. The DEAD-box protein Dbp5p is required to dissociate Mex67p from exported mRNPs at the nuclear rim. Mol. Cell 20, 645–651 (2005)
Tran, E. J., Zhou, Y., Corbett, A. H. & Wente, S. R. The DEAD-box protein Dbp5 controls mRNA export by triggering specific RNA:protein remodeling events. Mol. Cell 28, 850–859 (2007)
Weirich, C. S., Erzberger, J. P., Berger, J. M. & Weis, K. The N-terminal domain of Nup159 forms a β-propeller that functions in mRNA export by tethering the helicase Dbp5 to the nuclear pore. Mol. Cell 16, 749–760 (2004)
von Moeller, H., Basquin, C. & Conti, E. The mRNA export protein DBP5 binds RNA and the cytoplasmic nucleoporin NUP214 in a mutually exclusive manner. Nature Struct. Mol. Biol. 16, 247–254 (2009)
Weirich, C. S. et al. Activation of the DExD/H-box protein Dbp5 by the nuclear-pore protein Gle1 and its coactivator InsP6 is required for mRNA export. Nature Cell Biol. 8, 668–676 (2006)
Alcázar-Roman, A. R., Tran, E. J., Guo, S. & Wente, S. R. Inositol hexakisphosphate and Gle1 activate the DEAD-box protein Dbp5 for nuclear mRNA export. Nature Cell Biol. 8, 711–716 (2006)
Dossani, Z. Y., Weirich, C. S., Erzberger, J. P., Berger, J. M. & Weis, K. Structure of the C-terminus of the mRNA export factor Dbp5 reveals the interaction surface for the ATPase activator Gle1. Proc. Natl Acad. Sci. USA 106, 16251–16256 (2009)
Holm, L., Kaariainen, S., Rosenstrom, P. & Schenkel, A. Searching protein structure databases with DaliLite v.3. Bioinformatics 24, 2780–2781 (2008)
Oberer, M., Marintchev, A. & Wagner, G. Structural basis for the enhancement of eIF4A helicase activity by eIF4G. Genes Dev. 19, 2212–2223 (2005)
Korneeva, N. L., First, E. A., Benoit, C. A. & Rhoads, R. E. Interaction between the NH2-terminal domain of eIF4A and the central domain of eIF4G modulates RNA-stimulated ATPase activity. J. Biol. Chem. 280, 1872–1881 (2005)
Sonenberg, N. & Dever, T. E. Eukaryotic translation initiation factors and regulators. Curr. Opin. Struct. Biol. 13, 56–63 (2003)
He, F., Brown, A. H. & Jacobson, A. Upf1p, Nmd2p, and Upf3p are interacting components of the yeast nonsense-mediated mRNA decay pathway. Mol. Cell. Biol. 17, 1580–1594 (1997)
Chamieh, H., Ballut, L., Bonneau, F. & Le Hir, H. NMD factors UPF2 and UPF3 bridge UPF1 to the exon junction complex and stimulate its RNA helicase activity. Nature Struct. Mol. Biol. 15, 85–93 (2008)
Kadlec, J., Guilligay, D., Ravelli, R. B. & Cusack, S. Crystal structure of the UPF2-interacting domain of nonsense-mediated mRNA decay factor UPF1. RNA 12, 1817–1824 (2006)
Alcázar-Román, A. R., Bolger, T. A. & Wente, S. R. Control of mRNA export and translation termination by inositol hexakisphosphate requires specific interaction with Gle1. J. Biol. Chem. 285, 16683–16692 (2010)
Schutz, P. et al. Crystal structure of the yeast eIF4A-eIF4G complex: an RNA-helicase controlled by protein–protein interactions. Proc. Natl Acad. Sci. USA 105, 9564–9569 (2008)
Marintchev, A. et al. Topology and regulation of the human eIF4A/4G/4H helicase complex in translation initiation. Cell 136, 447–460 (2009)
Henn, A., Cao, W., Hackney, D. D. & De La Cruz, E. M. The ATPase cycle mechanism of the DEAD-box rRNA helicase, DbpA. J. Mol. Biol. 377, 193–205 (2008)
Henn, A. et al. Pathway of ATP utilization and duplex rRNA unwinding by the DEAD-box helicase, DbpA. Proc. Natl Acad. Sci. USA 107, 4046–4050 (2010)
Collins, R. et al. The DEXD/H-box RNA helicase DDX19 is regulated by an α-helical switch. J. Biol. Chem. 284, 10296–10300 (2009)
Napetschnig, J. et al. Structural and functional analysis of the interaction between the nucleoporin Nup214 and the DEAD-box helicase Ddx19. Proc. Natl Acad. Sci. USA 106, 3089–3094 (2009)
Büttner, K., Nehring, S. & Hopfner, K. P. Structural basis for DNA duplex separation by a superfamily-2 helicase. Nature Struct. Mol. Biol. 14, 647–652 (2007)
Fan, J. S. et al. Solution and crystal structures of mRNA exporter Dbp5p and its interaction with nucleotides. J. Mol. Biol. 388, 1–10 (2009)
Kapust, R. B. & Waugh, D. S. Controlled intracellular processing of fusion proteins by TEV protease. Protein Expr. Purif. 19, 312–318 (2000)
MacDowell, A. A. et al. Suite of three protein crystallography beamlines with single superconducting bend magnet as the source. J. Synchrotron Radiat. 11, 447–455 (2004)
Otwinowski, Z. & Minor, W. in Methods in Enzymology (eds Carter, W. C. & Sweet, R. M.) 307–326 (Academic, 1997)
Holton, J. & Alber, T. Automated protein crystal structure determination using ELVES. Proc. Natl Acad. Sci. USA 101, 1537–1542 (2004)
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Davis, I. W. et al. MolProbity: all-atom contacts and structure validation for proteins and nucleic acids. Nucleic Acids Res. 35, W375–W383 (2007)
Arnold, K., Bordoli, L., Kopp, J. & Schwede, T. The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22, 195–201 (2006)
Baker, N. A., Sept, D., Joseph, S., Holst, M. J. & McCammon, J. A. Electrostatics of nanosystems: application to microtubules and the ribosome. Proc. Natl Acad. Sci. USA 98, 10037–10041 (2001)
Flores, S. et al. The Database of Macromolecular Motions: new features added at the decade mark. Nucleic Acids Res. 34, D296–D301 (2006)
Brünger, A. T. et al. Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Lorsch, J. R. & Herschlag, D. The DEAD box protein eIF4A. 1. A minimal kinetic and thermodynamic framework reveals coupled binding of RNA and nucleotide. Biochemistry 37, 2180–2193 (1998)
Acknowledgements
We thank J. Kirsch, J. Thorner, M. Welch, D. Wemmer, B. Krantz, J. Zorn and all J.M.B. and K.W. laboratory members for discussions and advice. We also thank J. Kuriyan, M. Welch and S. Marqusee for access to equipment and workspace; J. Tanamachi, G. Meigs and J. Holton at ALS beamline 8.3.1; and N. Echols for assistance with programs from the Yale Morph Server. Research was supported by the G Harold and Leila Y Mathers foundation (J.M.B.) and the NIH (K.W., R01GM58065 and RC1GM91533).
Author information
Authors and Affiliations
Contributions
B.M., N.D.T., J.M.B. and K.W. designed the experiments. Protein purifications for crystallization and biochemical assays were performed by B.M. Crystal screening, optimization and data collection were performed by B.M. and N.D.T. Data processing, structure solution and model building was performed by N.D.T. In vivo tests and in vitro RNA binding, ATPase assays and other biochemical assays were performed by B.M. with assistance from M.A.S. and K.J.H. B.M., N.D.T., J.M.B. and K.W. both analysed and interpreted the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-6 with legends, Supplementary Tables 1-5 and additional references. (PDF 7788 kb)
Supplementary Movie 1
This movie shows an energy-minimized, linear interpolation of Dbp5 and Gle1 between the RNA-Δ90Dbp5 and Gle1-Δ90Dbp5 structural states. The movie begins in the RNA-Δ90Dbp5 state, with Gle1 and IP6 separated from the Dbp5 complex. The interpolation then transitions to the Gle1-Δ90Dbp5 state, in which the RNA and BeF3 are moved far from the ligand binding sites in order to mimic their release. The interpolation then plays in reverse to mimic RNA and ATP dependent closing of Dbp5 and release of Gle1. Note the large rigid body movement of the two RecA-like domains. Colouring is consistent with Fig. 1. (MOV 3881 kb)
Supplementary Movie 2
This movie shows the same interpolation as in Supplementary Movie 1, but focusing on the RNA binding site. Gle1-induced separation of the two RecA-like domains causes a significant structural change at the RNA binding site, disrupting the bipartite nucleic acid binding cleft and pulling RNA binding sidechains far apart. Colouring is consistent with Fig. 1. (MOV 2551 kb)
Supplementary Movie 3
This movie shows an energy-minimized, linear interpolation of Dbp5 between the RNA-Δ90Dbp5, Gle1-Δ90Dbp5, and Nup159-Δ90Dbp5-Gle1 structural states. The movie begins with Dbp5 in the RNA-Δ90Dbp5 state, then the interpolation transitions to the Gle1-Δ90Dbp5 state followed the Nup159-Δ90Dbp5-Gle1 state. The interpolation then plays in reverse. Note the large rigid body movement of the two RecA-like domains, which proceed along a common trajectory between the three discrete structural states. Colouring is consistent with Fig. 4. (MOV 1565 kb)
Rights and permissions
About this article
Cite this article
Montpetit, B., Thomsen, N., Helmke, K. et al. A conserved mechanism of DEAD-box ATPase activation by nucleoporins and InsP6 in mRNA export. Nature 472, 238–242 (2011). https://doi.org/10.1038/nature09862
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09862
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.