Abstract
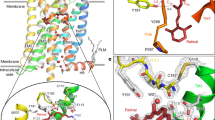
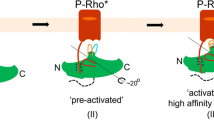
G-protein-coupled receptors (GPCRs) comprise the largest family of membrane proteins in the human genome and mediate cellular responses to an extensive array of hormones, neurotransmitters and sensory stimuli. Although some crystal structures have been determined for GPCRs, most are for modified forms, showing little basal activity, and are bound to inverse agonists or antagonists. Consequently, these structures correspond to receptors in their inactive states. The visual pigment rhodopsin is the only GPCR for which structures exist that are thought to be in the active state1,2. However, these structures are for the apoprotein, or opsin, form that does not contain the agonist all-trans retinal. Here we present a crystal structure at a resolution of 3 Å for the constitutively active rhodopsin mutant Glu 113 Gln3,4,5 in complex with a peptide derived from the carboxy terminus of the α-subunit of the G protein transducin. The protein is in an active conformation that retains retinal in the binding pocket after photoactivation. Comparison with the structure of ground-state rhodopsin6 suggests how translocation of the retinal β-ionone ring leads to a rotation of transmembrane helix 6, which is the critical conformational change on activation7. A key feature of this conformational change is a reorganization of water-mediated hydrogen-bond networks between the retinal-binding pocket and three of the most conserved GPCR sequence motifs. We thus show how an agonist ligand can activate its GPCR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Park, J. H. et al. Crystal structure of the ligand-free G-protein-coupled receptor opsin. Nature 454, 183–187 (2008)
Scheerer, P. et al. Crystal structure of opsin in its G-protein-interacting conformation. Nature 455, 497–502 (2008)
Zhukovsky, E. A. & Oprian, D. D. Effect of carboxylic acid side chains on the absorption maximum of visual pigments. Science 246, 928–930 (1989)
Sakmar, T. P., Franke, R. R. & Khorana, H. G. Glutamic acid-113 serves as the retinylidene Schiff base counterion in bovine rhodopsin. Proc. Natl Acad. Sci. USA 86, 8309–8313 (1989)
Robinson, P. R., Cohen, G. B., Zhukovsky, E. A. & Oprian, D. D. Constitutively active mutants of rhodopsin. Neuron 9, 719–725 (1992)
Li, J. et al. Structure of bovine rhodopsin in a trigonal crystal form. J. Mol. Biol. 343, 1409–1438 (2004)
Altenbach, C. et al. High-resolution distance mapping in rhodopsin reveals the pattern of helix movement due to activation. Proc. Natl Acad. Sci. USA 105, 7439–7444 (2008)
Hofmann, K. P. et al. A G protein-coupled receptor at work: the rhodopsin model. Trends Biochem. Sci. 34, 540–552 (2009)
Nakamichi, H. & Okada, T. Local peptide movement in the photoreaction intermediate of rhodopsin. Proc. Natl Acad. Sci. USA 103, 12729–12734 (2006)
Ruprecht, J. J. et al. Electron crystallography reveals the structure of metarhodopsin I. EMBO J. 23, 3609–3620 (2004)
Salom, D. et al. Crystal structure of a photoactivated deprotonated intermediate of rhodopsin. Proc. Natl Acad. Sci. USA 103, 16123–16128 (2006)
Hamm, H. E. et al. Site of G protein binding to rhodopsin mapped with synthetic peptides from the alpha subunit. Science 241, 832–835 (1988)
Ballesteros, J. & Weinstein, H. Integrated methods for the construction of three dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods Neurosci. 25, 366–428 (1995)
Standfuss, J., Zaitseva, E., Mahalingam, M. & Vogel, R. Structural impact of the E113Q counterion mutation on the activation and deactivation pathways of the G protein-coupled receptor rhodopsin. J. Mol. Biol. 380, 145–157 (2008)
Cohen, G. B., Oprian, D. D. & Robinson, P. R. Mechanism of activation and inactivation of opsin: role of Glu113 and Lys296. Biochemistry 31, 12592–12601 (1992)
Xie, G., Gross, A. K. & Oprian, D. D. An opsin mutant with increased thermal stability. Biochemistry 42, 1995–2001 (2003)
Standfuss, J. et al. Crystal structure of a thermally stable rhodopsin mutant. J. Mol. Biol. 372, 1179–1188 (2007)
Martin, E. L., Rens-Domiano, S., Schatz, P. J. & Hamm, H. E. Potent peptide analogues of a G protein receptor-binding region obtained with a combinatorial library. J. Biol. Chem. 271, 361–366 (1996)
Groenendijk, G. W., Jacobs, C. W., Bonting, S. L. & Daemen, F. J. Dark isomerization of retinals in the presence of phosphatidylethanolamine. Eur. J. Biochem. 106, 119–128 (1980)
Kefalov, V. J., Crouch, R. K. & Cornwall, M. C. Role of noncovalent binding of 11-cis-retinal to opsin in dark adaptation of rod and cone photoreceptors. Neuron 29, 749–755 (2001)
Kono, M., Goletz, P. W. & Crouch, R. K. 11-cis- and all-trans-retinols can activate rod opsin: rational design of the visual cycle. Biochemistry 47, 7567–7571 (2008)
Ahuja, S. et al. Location of the retinal chromophore in the activated state of rhodopsin. J. Biol. Chem. 284, 10190–10201 (2009)
Warne, T. et al. Structure of a β1-adrenergic G-protein-coupled receptor. Nature 454, 486–491 (2008)
Cherezov, V. et al. High-resolution crystal structure of an engineered human β2-adrenergic G protein-coupled receptor. Science 318, 1258–1265 (2007)
Jaakola, V. P. et al. The 2.6 angstrom crystal structure of a human A2A adenosine receptor bound to an antagonist. Science 322, 1211–1217 (2008)
Shi, L. et al. Beta2 adrenergic receptor activation. Modulation of the proline kink in transmembrane 6 by a rotamer toggle switch. J. Biol. Chem. 277, 40989–40996 (2002)
Schwartz, T. W. et al. Molecular mechanism of 7TM receptor activation–a global toggle switch model. Annu. Rev. Pharmacol. Toxicol. 46, 481–519 (2006)
Pardo, L. et al. The role of internal water molecules in the structure and function of the rhodopsin family of G protein-coupled receptors. ChemBioChem 8, 19–24 (2007)
Vogel, R. et al. Functional role of the “ionic lock”–an interhelical hydrogen-bond network in family A heptahelical receptors. J. Mol. Biol. 380, 648–655 (2008)
Reeves, P. J., Callewaert, N., Contreras, R. & Khorana, H. G. Structure and function in rhodopsin: high-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc. Natl Acad. Sci. USA 99, 13419–13424 (2002)
Reeves, P. J., Kim, J. M. & Khorana, H. G. Structure and function in rhodopsin: a tetracycline-inducible system in stable mammalian cell lines for high-level expression of opsin mutants. Proc. Natl Acad. Sci. USA 99, 13413 (2002)
Reeves, P. J., Callewaert, N., Contreras, R. & Khorana, H. G. Structure and function in rhodopsin: high-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc. Natl Acad. Sci. USA 99, 13419 (2002)
Sakmar, T. P., Franke, R. R. & Khorana, H. G. Glutamic acid-113 serves as the retinylidene Schiff base counterion in bovine rhodopsin. Proc. Natl Acad. Sci. USA 86, 8309 (1989)
Zhukovsky, E. A. & Oprian, D. D. Effect of carboxylic acid side chains on the absorption maximum of visual pigments. Science 246, 928 (1989)
Standfuss, J., Zaitseva, E., Mahalingam, M. & Vogel, R. Structural impact of the E113Q counterion mutation on the activation and deactivation pathways of the G protein-coupled receptor rhodopsin. J. Mol. Biol. 380, 145 (2008)
Kabsch, W. Automatic processing of rotation diffraction data from crystals of initially unknown symmetry and cell constants. J. Appl. Cryst. 26, 795 (1993)
Collaborative Computational Project, Number 4 . The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
McCoy, A. J., Grosse-Kunstleve, R. W., Storoni, L. C. & Read, R. J. Likelihood-enhanced fast translation functions. Acta Crystallogr. D 61, 458–464 (2005)
Park, J. H. et al. Crystal structure of the ligand-free G-protein-coupled receptor opsin. Nature 454, 183 (2008)
Scheerer, P. et al. Crystal structure of opsin in its G-protein-interacting conformation. Nature 455, 497 (2008)
Li, J. et al. Structure of bovine rhodopsin in a trigonal crystal form. J. Mol. Biol. 343, 1409 (2004)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948–1954 (2002)
Acknowledgements
We thank X. Deupi and R. Vogel for discussions and reading of the manuscript. We thank R. Crouch for the kind gift of 11-cis retinal. P. J. Reeves we thank for providing the pACMVtetO vector and the HEK293S-GnTI− cells, and for his advice on creating stable cell lines and tetracycline-inducible expression. We also thank the staff at the macromolecular crystallography beamlines at the European Synchrotron Radiation Facility, the Diamond Light Source and the Swiss Light Source. The work was financially supported by NIH grant EY007965 (to D.D.O.), the Human Frontier Science Project programme grant RG/0052 (to D.D.O. and G.F.X.S.), the European Commission FP6 specific targeted research project LSH-2003-1.1.0-1 (to G.F.X.S.), the Marie Curie Intra European Fellowship MEIF-CT-2006-039171 (to J.S.) and the EMBO long-term fellowship ALTF 198-2006 (to J.S.).
Author information
Authors and Affiliations
Contributions
The project was initiated by D.D.O. and G.F.X.S. J.S. performed cloning, initial expression and purification using essential experimental protocols and materials contributed by A.D. and D.D.O. Receptor activation and retinal binding studies were contributed by A.D. and G.X. Initial crystallization was by J.S., who also collected data and refined the structures. P.C.E. optimized expression and crystallization, performed crystal cryo-cooling and coordinated data collection. M.F. investigated the stability of mutant proteins. Manuscript preparation was performed by J.S. and D.D.O. The overall project management was by G.F.X.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-5 with legends, Supplementary Tables 1-2 and additional references. (PDF 503 kb)
Rights and permissions
About this article
Cite this article
Standfuss, J., Edwards, P., D’Antona, A. et al. The structural basis of agonist-induced activation in constitutively active rhodopsin. Nature 471, 656–660 (2011). https://doi.org/10.1038/nature09795
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09795
This article is cited by
-
Analyzing CW EPR Spectra of Nitroxide Labeled Macromolecules
Applied Magnetic Resonance (2024)
-
Single-molecule visualization of human A2A adenosine receptor activation by a G protein and constitutively activating mutations
Communications Biology (2023)
-
Ultrafast structural changes direct the first molecular events of vision
Nature (2023)
-
The rhodopsin-retinochrome system for retinal re-isomerization predates the origin of cephalopod eyes
BMC Ecology and Evolution (2021)
-
Structural aspects of rod opsin and their implication in genetic diseases
Pflügers Archiv - European Journal of Physiology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.