Abstract
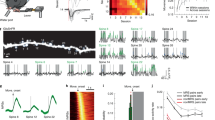
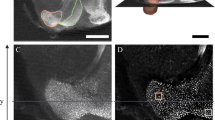
Behavioural learning depends on the brain’s capacity to respond to instructive experience and is often enhanced during a juvenile sensitive period. How instructive experience acts on the juvenile brain to trigger behavioural learning remains unknown. In vitro studies show that forms of synaptic strengthening thought to underlie learning are accompanied by an increase in the stability, number and size of dendritic spines, which are the major sites of excitatory synaptic transmission in the vertebrate brain1,2,3,4,5,6,7. In vivo imaging studies in sensory cortical regions reveal that these structural features can be affected by disrupting sensory experience and that spine turnover increases during sensitive periods for sensory map formation8,9,10,11,12. These observations support two hypotheses: first, the increased capacity for behavioural learning during a sensitive period is associated with enhanced spine dynamics on sensorimotor neurons important for the learned behaviour; second, instructive experience rapidly stabilizes and strengthens these dynamic spines. Here we report a test of these hypotheses using two-photon in vivo imaging to measure spine dynamics in zebra finches, which learn to sing by imitating a tutor song during a juvenile sensitive period13,14. Spine dynamics were measured in the forebrain nucleus HVC, the proximal site where auditory information merges with an explicit song motor representation15,16,17,18,19, immediately before and after juvenile finches first experienced tutor song20. Higher levels of spine turnover before tutoring correlated with a greater capacity for subsequent song imitation. In juveniles with high levels of spine turnover, hearing a tutor song led to the rapid (∼24-h) stabilization, accumulation and enlargement of dendritic spines in HVC. Moreover, in vivo intracellular recordings made immediately before and after the first day of tutoring revealed robust enhancement of synaptic activity in HVC. These findings suggest that behavioural learning results when instructive experience is able to rapidly stabilize and strengthen synapses on sensorimotor neurons important for the control of the learned behaviour.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Matsuzaki, M., Honkura, N., Ellis-Davies, G. C. & Kasai, H. Structural basis of long-term potentiation in single dendritic spines. Nature 429, 761–766 (2004)
De Roo, M., Klauser, P. & Muller, D. LTP promotes a selective long-term stabilization and clustering of dendritic spines. PLoS Biol. 6, e219 (2008)
Chklovskii, D. B., Mel, B. W. & Svoboda, K. Cortical rewiring and information storage. Nature 431, 782–788 (2004)
Engert, F. & Bonhoeffer, T. Dendritic spine changes associated with hippocampal long-term synaptic plasticity. Nature 399, 66–70 (1999)
Alvarez, V. A. & Sabatini, B. L. Anatomical and physiological plasticity of dendritic spines. Annu. Rev. Neurosci. 30, 79–97 (2007)
Maletic-Savatic, M., Malinow, R. & Svoboda, K. Rapid dendritic morphogenesis in CA1 hippocampal dendrites induced by synaptic activity. Science 283, 1923–1927 (1999)
Zito, K., Scheuss, V., Knott, G., Hill, T. & Svoboda, K. Rapid functional maturation of nascent dendritic spines. Neuron 61, 247–258 (2009)
Hofer, S. B., Mrsic-Flogel, T. D., Bonhoeffer, T. & Hübener, M. Experience leaves a lasting structural trace in cortical circuits. Nature 457, 313–317 (2009)
Majewska, A. & Sur, M. Motility of dendritic spines in visual cortex in vivo: changes during the critical period and effects of visual deprivation. Proc. Natl Acad. Sci. USA 100, 16024–16029 (2003)
Zuo, Y., Lin, A., Chang, P. & Gan, W. B. Development of long-term dendritic spine stability in diverse regions of cerebral cortex. Neuron 46, 181–189 (2005)
Zuo, Y., Yang, G., Kwon, E. & Gan, W. B. Long-term sensory deprivation prevents dendritic spine loss in primary somatosensory cortex. Nature 436, 261–265 (2005)
Holtmaat, A., Wilbrecht, L., Knott, G. W., Welker, E. & Svoboda, K. Experience-dependent and cell-type-specific spine growth in the neocortex. Nature 441, 979–983 (2006)
Eales, L. A. Song learning in zebra finches: some effects of song model availability on what is learnt and when. Anim. Behav. 33, 1293–1300 (1985)
Immelmann, K. in Bird Vocalisations (ed. Hinde, R. A.) 61–74 (Cambridge Univ. Press, 1969)
Prather, J. F., Peters, S., Nowicki, S. & Mooney, R. Precise auditory-vocal mirroring in neurons for learned vocal communication. Nature 451, 305–310 (2008)
Bauer, E. E. et al. A synaptic basis for auditory-vocal integration in the songbird. J. Neurosci. 28, 1509–1522 (2008)
McCasland, J. S. & Konishi, M. Interaction between auditory and motor activities in an avian song control nucleus. Proc. Natl Acad. Sci. USA 78, 7815–7819 (1981)
Nottebohm, F., Stokes, T. M. & Leonard, C. M. Central control of song in the canary, Serinus canarius . J. Comp. Neurol. 165, 457–486 (1976)
Hahnloser, R. H. R., Kozhevnikov, A. A. & Fee, M. S. An ultra-sparse code underlies the generation of neural sequences in a songbird. Nature 419, 65–70 (2002)
Tchernichovski, O., Mitra, P. P., Lints, T. & Nottebohm, F. Dynamics of the vocal imitation process: how a zebra finch learns its song. Science 291, 2564–2569 (2001)
Roberts, T. F., Klein, M. E., Kubke, M. F., Wild, J. M. & Mooney, R. Telencephalic neurons monosynaptically link brainstem and forebrain premotor networks necessary for song. J. Neurosci. 28, 3479–3489 (2008)
Dittgen, T. et al. Lentivirus-based genetic manipulations of cortical neurons and their optical and electrophysiological monitoring in vivo . Proc. Natl Acad. Sci. USA 101, 18206–18211 (2004)
Mooney, R. Different subthreshold mechanisms underlie song-selectivity in identified HVc neurons of the zebra finch. J. Neurosci. 20, 5420–5436 (2000)
Scharff, C., Kirn, J. R., Grossman, M., Macklis, J. D. & Nottebohm, F. Targeted neuronal death affects neuronal replacement and vocal behavior in adult songbirds. Neuron 25, 481–492 (2000)
Trachtenberg, J. T. et al. Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420, 788–794 (2002)
Derégnaucourt, S., Mitra, P. P., Feher, O., Pytte, C. & Tchernichovski, O. How sleep affects the developmental learning of bird song. Nature 433, 710–716 (2005)
Kopec, C. D., Li, B., Wei, W., Boehm, J. & Malinow, R. Glutamate receptor exocytosis and spine enlargement during chemically induced long-term potentiation. J. Neurosci. 26, 2000–2009 (2006)
Holtmaat, A. J. et al. Transient and persistent dendritic spines in the neocortex in vivo . Neuron 45, 279–291 (2005)
Shank, S. S. & Margoliash, D. Sleep and sensorimotor integration during early vocal learning in a songbird. Nature 458, 73–77 (2009)
Acknowledgements
We thank M. Ehlers and L. Katz for access to the two-photon microscope and support in making lentivirus, K. Hamaguchi for peak detection and analysis software and D. Kloetzer for animal husbandry and laboratory support. D. Fitzpatrick, D. Purves and M. Ehlers provided comments on the manuscript. This work was supported by grants from the US National Science Foundation (NSF) and the US National Institutes of Health (NIH) (R.M.). T.F.R. was supported by a National Research Service Award from the NIH, K.A.T. was supported by a pre-doctoral award from the NSF and M.E.K. was supported by the Howard Hughes Medical Institute (Investigator, M. Ehlers).
Author Contributions T.F.R. and R.M. designed the study and wrote the manuscript. T.F.R. and K.A.T. collected and analysed the imaging and behavioural data. T.F.R. and M.E.K. designed the lentiviral construct and M.E.K. made the lentivirus. T.F.R and R.M. collected the electrophysiological data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Table 1 and Supplementary Figures 1-4 with Legends. (PDF 915 kb)
Rights and permissions
About this article
Cite this article
Roberts, T., Tschida, K., Klein, M. et al. Rapid spine stabilization and synaptic enhancement at the onset of behavioural learning. Nature 463, 948–952 (2010). https://doi.org/10.1038/nature08759
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08759
This article is cited by
-
Developing a structural-based local learning rule for classification tasks using ionic liquid space-based reservoir
Neural Computing and Applications (2022)
-
Expression of FoxP2 in the basal ganglia regulates vocal motor sequences in the adult songbird
Nature Communications (2021)
-
Ube2b-dependent degradation of DNMT3a relieves a transcriptional brake on opiate-induced synaptic and behavioral plasticity
Molecular Psychiatry (2021)
-
An avian cortical circuit for chunking tutor song syllables into simple vocal-motor units
Nature Communications (2020)
-
Cortex-wide neural interfacing via transparent polymer skulls
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.