Abstract
Arising from: D. Ben-Zvi et al. Nature 453, 1205–1211 (2008)10.1038/nature07059; Francois et al. reply
Metazoan organisms can ‘scale’, that is, maintain similar proportions regardless of size. Ben-Zvi et al.1 use experiments in Xenopus to support a quantitative model that explains morphological scaling as the result of scaling of a gradient of bone morphogenetic protein (BMP) signals. We believe that the evidence for scaling in Xenopus is misinterpreted, and that their model for embryonic patterning disagrees with prior data. The experiments they present supporting their model admit alternative interpretations.
Similar content being viewed by others
Main
The authors’ model (box 1 of ref. 1) is built around the (BMP inhibitor) Chordin-facilitated transport of two members of the BMP family of ligands, BMP (BMP2/4/7), the total amount of which is preserved, and ADMP, which is produced only in the dorsal organizer but concentrates ventrally and scales along the dorsal–ventral axis at blastula stage.
The paper is based on the assumption that dorsal embryonic halves produce well-proportioned (scaled) tadpoles, which is in contrast to existing data. Kageura et al.2 show that removing ventral cells from the eight-cell blastula (series 15, 17) results in normal heads attached to a small body. This is in accordance with standard fate maps that assign most of what is conventionally called ventral in the blastula to posterior tissue in the tadpole3. It is right–left half-embryos that will reproduce correctly proportioned half-size tadpoles2. The quoted paper by Cooke4 examined only mesoderm patterning in transverse sections of tailbud embryos. Therefore, dorsal half-embryos do not scale in the sense defined by the authors. We henceforth focus on molecular evidence contradicting the presented theory construed as a model for embryonic patterning.
In the frog a twofold change in morphogen levels can elicit different cell fates5. Because ligands cannot be directly measured, nuclear Smad1/5/8 transcription factor is the best measure for total BMP signalling6. Experiments in frog7 and fish8 show at most a fourfold variation versus the 102 to 104 range required for scaling in the model.
The model requires that total BMP activity derives predominantly from ADMP, yet BMP depletion (figure 2H in ref. 9) has a stronger phenotype than ADMP-depletion (figure 2H in ref. 10), resulting in embryos with disproportionately large heads8,9 similar to dorsal half-embryos (figure 3C in ref. 10). BMP4 injection significantly ventralizes the embryo (figure 1H in ref. 9), yet the model does not constrain the total amount of BMP or its initial location, because ‘shuttling’ actively concentrates it on the ventral side.
Experiments in figure 3 of ref. 1 were performed to demonstrate Chordin-dependent shuttling. BMP4 is used instead of ADMP, and the protein distribution is shown in mid–late gastrula, although BMP must act before early gastrulation to affect dorsal–ventral patterning significantly11. Labelled BMP4 is localized in endoderm and not ventral mesoderm as in the schematic of figure 3a of ref. 1. Other explanations for the localization of injected BMP4, such as secretion into the blastocoel cavity and ectopic uptake12, need to be addressed. The Chordin-depleted embryos used as controls still show movement of injected BMP4, and the phenotype undercuts the larger message given that such embryos have well-defined axes13. The complete model that addresses these questions (supplementary information 6a–h of ref. 1) contains over 30 free constants to explain essentially qualitative data; a number so large as to render the predictions questionable.
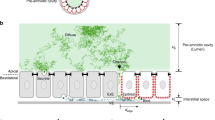
Axis duplication experiments (figure 4 of ref. 1) are taken as evidence for shuttling: the authors assert that the (well-known) expression of ventral markers between the two axes is evidence for their mode of transport. However, there must be a maximum in BMP signalling between the two axes because it is suppressed in each. Reaction diffusion models14,15 show that ordinary diffusion, as opposed to facilitated diffusion through shuttling, can generate patterns consistent with the qualitative data presented (our Fig. 1).
The gene network is adapted from figure 3L in ref. 10 and involves self-sustained ventral and dorsal centres. The concentration of the ventral BMP marker in the lateral region between the duplicated axes is clearly reproduced. (The inhibitors, ADMP and Sizzled, diffuse rapidly, the other species slowly. Activation and repression are modelled as Hill functions and summarize more complex biochemistry. The equations and parameters that were solved to produce Fig. 1 are available from the authors.
In summary, we feel it is incorrect to appeal to qualitative data beyond the onset of gastrulation to support a model for blastula patterning, because other layers of regulation may intervene.
References
Ben-Zvi, D., Shilo, B., Fainsod, A. & Barkai, N. Scaling of the BMP activation gradient in Xenopus embryos. Nature 453, 1205–1211 (2008)
Kageura, H. & Yamana, K. Pattern regulation in defect embryos of Xenopus laevis. Dev. Biol. 101, 410–415 (1984)
Gerhart, J. Changing the axis changes the perspective. Dev. Dyn. 225, 380–383 (2002)
Cooke, J. Scale of body pattern adjusts to available cell number in amphibian embryos. Nature 290, 775–778 (1981)
Kinoshita, T., Jullien, J. & Gurdon, J. B. Two-dimensional morphogen gradient in Xenopus: boundary formation and real-time transduction response. Dev. Dyn. 235, 3189–3198 (2006)
Faure, S., Lee, M. A., Keller, T., ten Dijke, P. & Whitman, M. Endogenous patterns of TGFbeta superfamily signaling during early Xenopus development. Development 127, 2917–2931 (2000)
Schohl, A. & Fagotto, F. Beta-catenin, MAPK and Smad signaling during early Xenopus development. Development 129, 37–52 (2002)
Tucker, J. A., Mintzer, K. A. & Mullins, M. C. The BMP signaling gradient patterns dorsoventral tissues in a temporally progressive manner along the anteroposterior axis. Dev. Cell 14, 108–119 (2008)
Reversade, B., Kuroda, H., Lee, H., Mays, A. & De Robertis, E. M. Depletion of Bmp2, Bmp4, Bmp7 and Spemann organizer signals induces massive brain formation in Xenopus embryos. Development 132, 3381–3392 (2005)
Reversade, B. & De Robertis, E. Regulation of ADMP and BMP2/4/7 at opposite embryonic poles generates a self-regulating morphogenetic field. Cell 123, 1147–1160 (2005)
Marom, K., Levy, V., Pillemer, G. & Fainsod, A. Temporal analysis of the early BMP functions identifies distinct anti-organizer and mesoderm patterning phases. Dev. Biol. 282, 442–454 (2005)
Williams, P. H., Hagemann, A., Gonzalez-Gaitan, M. & Smith, J. C. Visualizing long-range movement of the morphogen Xnr2 in the Xenopus embryo. Curr. Biol. 14, 1916–1923 (2004)
Oelgeschlager, M., Kuroda, H., Reversade, B. & De Robertis, E. M. Chordin is required for the Spemann organizer transplantation phenomenon in Xenopus embryos. Dev. Cell 4, 219–230 (2003)
Meinhardt, H. Organizer and axes formation as a self-organizing process. Int. J. Dev. Biol. 45, 177–188 (2001)
Solnica-Krezel, L. Vertebrate development: taming the nodal waves. Curr. Biol. 13, R7–R9 (2003)
Author information
Authors and Affiliations
PowerPoint slides
Rights and permissions
About this article
Cite this article
Francois, P., Vonica, A., Brivanlou, A. et al. Scaling of BMP gradients in Xenopus embryos. Nature 461, E1 (2009). https://doi.org/10.1038/nature08305
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08305
This article is cited by
-
Scale-invariant patterning by size-dependent inhibition of Nodal signalling
Nature Cell Biology (2018)
-
Dynamic scaling of morphogen gradients on growing domains
Nature Communications (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.