Abstract
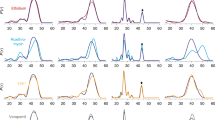
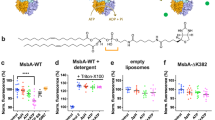
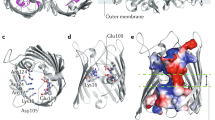
Membrane proteins that transport hydrophobic compounds have important roles in multi-drug resistance1,2,3 and can cause a number of diseases4,5, underscoring the importance of protein-mediated transport of hydrophobic compounds. Hydrophobic compounds readily partition into regular membrane lipid bilayers6, and their transport through an aqueous protein channel is energetically unfavourable3. Alternative transport models involving acquisition from the lipid bilayer by lateral diffusion have been proposed for hydrophobic substrates3,4,7,8,9,10,11,12. So far, all transport proteins for which a lateral diffusion mechanism has been proposed function as efflux pumps. Here we present the first example of a lateral diffusion mechanism for the uptake of hydrophobic substrates by the Escherichia coli outer membrane long-chain fatty acid transporter FadL. A FadL mutant in which a lateral opening in the barrel wall is constricted, but which is otherwise structurally identical to wild-type FadL, does not transport substrates. A crystal structure of FadL from Pseudomonas aeruginosa shows that the opening in the wall of the β-barrel is conserved and delineates a long, hydrophobic tunnel that could mediate substrate passage from the extracellular environment, through the polar lipopolysaccharide layer and, by means of the lateral opening in the barrel wall, into the lipid bilayer from where the substrate can diffuse into the periplasm. Because FadL homologues are found in pathogenic and biodegrading bacteria, our results have implications for combating bacterial infections and bioremediating xenobiotics in the environment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Saier, M. H. & Paulsen, I. T. Phylogeny of multidrug transporters. Semin. Cell Dev. Biol. 12, 205–213 (2001)
Poole, K. Efflux pumps as antimicrobial resistance mechanisms. Ann. Med. 39, 162–176 (2007)
Sharom, F. J. Shedding light on drug transport: structure and function of the P-glycoprotein multidrug transporter (ABCB1). Biochem. Cell Biol. 84, 979–992 (2006)
van Meer, G., Halter, D., Sprong, H., Somerharju, P. & Egmond, M. R. ABC lipid transporters: extruders, flippases or flopless activators? FEBS Lett. 580, 1171–1177 (2006)
Borst, P., Zelcer, N. & van Helvoort, A. ABC transporters in lipid transport. Biochim. Biophys. Acta 1486, 128–144 (2000)
Siarheyeva, A., Lopez, J. J. & Glaubitz, C. Localization of multidrug transporter substrates within model membranes. Biochemistry 45, 6203–6211 (2006)
Higgins, C. F. & Gottesman, M. M. Is the multidrug transporter a flippase? Trends Biochem. Sci. 17, 18–21 (1992)
Higgins, C. F. Multiple molecular mechanisms for multidrug resistance transporters. Nature 446, 749–757 (2007)
van Veen, H. W., Putman, M., Margolles, A., Sakamoto, K. & Konings, W. N. Structure–function analysis of multidrug transporters in Lactococcus lactis. Biochim. Biophys. Acta 1461, 201–206 (1999)
Bolhuis, H. et al. Multidrug resistance in Lactococcus lactis: evidence for ATP-dependent drug extrusion from the inner leaflet of the cytoplasmic membrane. EMBO J. 15, 4239–4245 (1996)
Shapiro, A. B. & Ling, V. Extraction of Hoechst 33342 from the cytoplasmic leaflet of the plasma membrane by P-glycoprotein. Eur. J. Biochem. 250, 122–129 (1997)
Shapiro, A. B. & Ling, V. Transport of LDS-751 from the cytoplasmic leaflet of the plasma membrane by the rhodamine-123-selective site of P-glycoprotein. Eur. J. Biochem. 254, 181–188 (1998)
Nunn, W. D. & Simons, R. W. Transport of long-chain fatty acids by Escherichia coli: mapping and characterization of mutants in the fadL gene. Proc. Natl Acad. Sci. USA 75, 3377–3381 (1978)
Black, P. N., Said, B., Ghosn, C. R., Beach, J. V. & Nunn, W. D. Purification and characterization of an outer membrane-bound protein involved in long-chain fatty acid transport in Escherichia coli. J. Biol. Chem. 262, 1412–1419 (1987)
van den Berg, B., Black, P. N., Clemons, W. M. & Rapoport, T. M. Crystal structure of the long-chain fatty acid transporter FadL. Science 304, 1506–1509 (2004)
DiRusso, C. C. & Black, P. N. Bacterial long-chain fatty acid transport: gateway to a fatty acid-responsive signalling system. J. Biol. Chem. 279, 49563–49566 (2004)
Hearn, E. M., Patel, D. R. & van den Berg, B. Outer-membrane transport of aromatic hydrocarbons as a first step in biodegradation. Proc. Natl Acad. Sci. USA 105, 8601–8606 (2008)
Kumar, G. B. & Black, P. N. Linker mutagenesis of a bacterial fatty acid transport protein: identification of domains with functional importance. J. Biol. Chem. 266, 1348–1353 (1991)
Vorum, H., Brodersen, R., Kragh-Hansen, U. & Pedersen, A. O. Solubility of long-chain fatty acids in phosphate buffer at pH 7.4. Biochim. Biophys. Acta 1126, 135–142 (1992)
Black, P. N. Characterization of FadL-specific fatty acid binding in Escherichia coli. Biochim. Biophys. Acta 1046, 97–105 (1990)
Wang, Y. et al. Identification of a membrane protein and a truncated LysR-type regulator associated with the toluene degradation pathway in Pseudomonas putida F1. Mol. Gen. Genet. 246, 570–579 (1995)
Kahng, H. Y., Byrne, A. M., Olsen, R. H. & Kukor, J. J. Characterization and role of tbuX in utilization of toluene in Ralstonia pickettii PKO1. J. Bacteriol. 182, 1232–1242 (2000)
Eastcott, L., Shiu, W. Y. & Mackay, D. Environmentally relevant physical-chemical properties of hydrocarbons: a review of data and development of simple correlations. Oil Chem. Pollut. 4, 191–216 (1988)
Ahn, V. E. et al. A hydrocarbon ruler measures palmitate in the enzymatic acylation of endotoxin. EMBO J. 23, 2931–2941 (2004)
Hong, H., Patel, D. R., Tamm, L. K. & van den Berg, B. The outer membrane protein OmpW forms an eight-stranded beta-barrel with a hydrophobic channel. J. Biol. Chem. 281, 7568–7577 (2006)
Eaton, R. W. Organization and evolution of naphthalene catabolic pathways: sequence of the DNA encoding 2-hydroxychromene-2-carboxylate isomerase and trans-o-hydroxybenzylidenepyruvate hydratase-aldolase from the NAH7 plasmid. J. Bacteriol. 176, 7757–7762 (1994)
van Beilen, J. B., Eggink, G., Enequist, H., Bos, R. & Witholt, B. DNA sequence determination and functional characterization of the OCT-plasmid-encoded alkJKL genes of Pseudomonas oleovorans. Mol. Microbiol. 6, 3121–3136 (1992)
Sun, P. D., Radaev, S. & Kattah, M. Generating isomorphous heavy-atom derivatives by a quick-soak method. Part I: test cases. Acta Crystallogr. D 58, 1092–1098 (2002)
Guzman, L. M., Belin, D., Carson, M. J. & Beckwith, J. Tight regulation, modulation and high-level expression by vectors containing the arabinose PBAD promoter. J. Bacteriol. 177, 4121–4130 (1995)
Ginsburgh, C. L., Black, P. N. & Nunn, W. D. Transport of long-chain fatty acids in Escherichia coli: identification of a membrane protein associated with the fadL gene. J. Biol. Chem. 259, 8437–8443 (1984)
Ye, J. & van den Berg, B. Crystal structure of the bacterial nucleoside transporter Tsx. EMBO J. 23, 3187–3195 (2004)
Abramoff, M. D., Magelhaes, P. J. & Ram, S. J. Image processing with ImageJ. Biophotonics Int. 11, 36–42 (2004)
Miroux, B. & Walker, J. E. Over-production of proteins in Escherichia coli: mutant hosts that allow synthesis of some membrane proteins and globular proteins at high levels. J. Mol. Biol. 260, 289–298 (1996)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
McCoy, A. J., Grosse-Kunstleve, R. W., Storoni, L. C. & Read, R. J. Likelihood-enhanced fast translation functions. Acta Crystallogr. D 61, 458–464 (2005)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Brunger, A. T. et al. Crystallography and NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Bendtsen, J. D., Nielsen, H., von Heijne, G. & Brunak, S. Improved prediction of signal peptides: SignalP 3.0. J. Mol. Biol. 340, 783–795 (2004)
Matthews, B. W. Solvent content of protein crystals. J. Mol. Biol. 33, 491–497 (1968)
Terwilliger, T. C. & Berendzen, J. Automated MAD and MIR structure solution. Acta Crystallogr. D 55, 849–861 (1999)
de La Fortelle, E. & Bricogne, G. Maximum-likelihood heavy-atom parameter refinement for multiple isomorphous replacement and multi-wavelength anomalous diffraction methods. Methods Enzymol. 276, 472–494 (1997)
Acknowledgements
We thank the personnel of the National Synchrotron Light Source (NSLS) beamlines X6A and X29 for beam time and beamline support. We are grateful to P. Black and C. Petteys for the strain D10 and for their technical advice on the fatty acid transport assays. This work was supported by a training grant from the National Insitutes of Health (to E.M.H.) and by a NIH research grant (1R01GM074824 to B.v.d.B.).
Author Contributions E.M.H. cloned, purified and crystallized FadL mutants, performed activity assays, and wrote the paper; D.R.P. cloned, purified and crystallized FadL mutants; B.W.L. performed activity assays; M.I. purified and crystallized PaFadL; and B.v.d.B. determined crystal structures, designed research and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-2 and Supplementary Figure 1-5 with Legends (PDF 845 kb)
Rights and permissions
About this article
Cite this article
Hearn, E., Patel, D., Lepore, B. et al. Transmembrane passage of hydrophobic compounds through a protein channel wall. Nature 458, 367–370 (2009). https://doi.org/10.1038/nature07678
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07678
This article is cited by
-
Uptake of monoaromatic hydrocarbons during biodegradation by FadL channel-mediated lateral diffusion
Nature Communications (2020)
-
Improving Escherichia coli membrane integrity and fatty acid production by expression tuning of FadL and OmpF
Microbial Cell Factories (2017)
-
Lipopolysaccharide transport and assembly at the outer membrane: the PEZ model
Nature Reviews Microbiology (2016)
-
A Review on the Genetics of Aliphatic and Aromatic Hydrocarbon Degradation
Applied Biochemistry and Biotechnology (2016)
-
Crystal structure of a COG4313 outer membrane channel
Scientific Reports (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.