Abstract
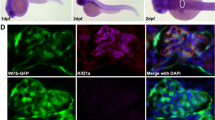
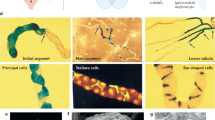
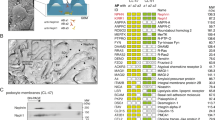
The nephron is the basic structural and functional unit of the vertebrate kidney. It is composed of a glomerulus, the site of ultrafiltration, and a renal tubule, along which the filtrate is modified. Although widely regarded as a vertebrate adaptation1, ‘nephron-like’ features can be found in the excretory systems of many invertebrates, raising the possibility that components of the vertebrate excretory system were inherited from their invertebrate ancestors2. Here we show that the insect nephrocyte has remarkable anatomical, molecular and functional similarity to the glomerular podocyte, a cell in the vertebrate kidney that forms the main size-selective barrier as blood is ultrafiltered to make urine. In particular, both cell types possess a specialized filtration diaphragm, known as the slit diaphragm in podocytes or the nephrocyte diaphragm in nephrocytes. We find that fly (Drosophila melanogaster) orthologues of the major constituents of the slit diaphragm, including nephrin, NEPH1 (also known as KIRREL), CD2AP, ZO-1 (TJP1) and podocin, are expressed in the nephrocyte and form a complex of interacting proteins that closely mirrors the vertebrate slit diaphragm complex. Furthermore, we find that the nephrocyte diaphragm is completely lost in flies lacking the orthologues of nephrin or NEPH1—a phenotype resembling loss of the slit diaphragm in the absence of either nephrin (as in human congenital nephrotic syndrome of the Finnish type, NPHS1) or NEPH1. These changes markedly impair filtration function in the nephrocyte. The similarities we describe between invertebrate nephrocytes and vertebrate podocytes provide evidence suggesting that the two cell types are evolutionarily related, and establish the nephrocyte as a simple model in which to study podocyte biology and podocyte-associated diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Smith, H. W. From Fish to Philosopher (Little, Brown, 1953)
Ruppert, E. E. Evolutionary origin of the vertebrate nephron. Am. Zool. 34, 542–533 (1994)
Rodewald, R. & Karnovsky, M. J. Porous substructure of the glomerular slit diaphragm in the rat and mouse. J. Cell Biol. 60, 423–433 (1974)
Wartiovaara, J. et al. Nephrin strands contribute to a porous slit diaphragm scaffold as revealed by electron tomography. J. Clin. Invest. 114, 1475–1483 (2004)
Patrakka, J. et al. Congenital nephrotic syndrome (NPHS1): features resulting from different mutations in Finnish patients. Kidney Int. 58, 972–980 (2000)
Berridge, M. J. & Oschman, J. L. Transporting Epithelia 11–15 (Academic, 1972)
Crossley, A. C. in Comprehensive Insect Physiology, Biochemistry and Pharmacology (eds Kerkut, G. A. & Gilbert, L. I.) 487–515 (Pergamon, 1985)
Kowalevsky, A. Ein Beitrag zur Kenntnis der Excretions-organe. Biol. Centralbl. 9, 74–79 (1889)
Locke, M. & Russell, V. W. in Microscopic Anatomy of Invertebrates (eds Harrison, F. W. & Locke, M.) 687–709 (Wiley, 1998)
Kestila, M. et al. Positionally cloned gene for a novel glomerular protein—nephrin—is mutated in congenital nephrotic syndrome. Mol. Cell 1, 575–582 (1998)
Sellin, L. et al. NEPH1 defines a novel family of podocin interacting proteins. FASEB J. 17, 115–117 (2003)
Ruotsalainen, V. et al. Nephrin is specifically located at the slit diaphragm of glomerular podocytes. Proc. Natl Acad. Sci. USA 96, 7962–7967 (1999)
Gerke, P., Huber, T. B., Sellin, L., Benzing, T. & Walz, G. Homodimerization and heterodimerization of the glomerular podocyte proteins nephrin and NEPH1. J. Am. Soc. Nephrol. 14, 918–926 (2003)
Barletta, G. M., Kovari, I. A., Verma, R. K., Kerjaschki, D. & Holzman, L. B. Nephrin and Neph1 co-localize at the podocyte foot process intercellular junction and form cis hetero-oligomers. J. Biol. Chem. 278, 19266–19271 (2003)
Khoshnoodi, J. et al. Nephrin promotes cell–cell adhesion through homophilic interactions. Am. J. Pathol. 163, 2337–2346 (2003)
Liu, G. et al. Neph1 and nephrin interaction in the slit diaphragm is an important determinant of glomerular permeability. J. Clin. Invest. 112, 209–221 (2003)
Donoviel, D. B. et al. Proteinuria and perinatal lethality in mice lacking NEPH1, a novel protein with homology to NEPHRIN. Mol. Cell. Biol. 21, 4829–4836 (2001)
Tepass, U. & Hartenstein, V. The development of cellular junctions in the Drosophila embryo. Dev. Biol. 161, 563–596 (1994)
Rugendorff, A., Younossi-Hartenstein, A. & Hartenstein, V. Embryonic origin and differentiation of the Drosophila heart. Rouxs Arch. Dev. Biol. 203, 266–280 (1994)
Huber, T. B. et al. The carboxyl terminus of Neph family members binds to the PDZ domain protein zonula occludens-1. J. Biol. Chem. 278, 13417–13421 (2003)
Shih, N. Y. et al. Congenital nephrotic syndrome in mice lacking CD2-associated protein. Science 286, 312–315 (1999)
Shih, N. Y. et al. CD2AP localizes to the slit diaphragm and binds to nephrin via a novel C-terminal domain. Am. J. Pathol. 159, 2303–2308 (2001)
Boute, N. et al. NPHS2, encoding the glomerular protein podocin, is mutated in autosomal recessive steroid-resistant nephrotic syndrome. Nature Genet. 24, 349–354 (2000)
Kim, J. M. et al. CD2-associated protein haploinsufficiency is linked to glomerular disease susceptibility. Science 300, 1298–1300 (2003)
Schwarz, K. et al. Podocin, a raft-associated component of the glomerular slit diaphragm, interacts with CD2AP and nephrin. J. Clin. Invest. 108, 1621–1629 (2001)
Galletta, B. J., Chakravarti, M., Banerjee, R. & Abmayr, S. M. SNS: Adhesive properties, localization requirements and ectodomain dependence in S2 cells and embryonic myoblasts. Mech. Dev. 121, 1455–1468 (2004)
Kramer-Zucker, A. G., Wiessner, S., Jensen, A. M. & Drummond, I. A. Organization of the pronephric filtration apparatus in zebrafish requires Nephrin, Podocin and the FERM domain protein Mosaic eyes. Dev. Biol. 285, 316–329 (2005)
Akilesh, S. et al. Podocytes use FcRn to clear IgG from the glomerular basement membrane. Proc. Natl Acad. Sci. USA 105, 967–972 (2008)
Das, D., Aradhya, R., Ashoka, D. & Inamdar, M. Post-embryonic pericardial cells of Drosophila are required for overcoming toxic stress but not for cardiac function or adult development. Cell Tissue Res. 331, 565–570 (2008)
Huber, T. B. & Benzing, T. The slit diaphragm: a signaling platform to regulate podocyte function. Curr. Opin. Nephrol. Hypertens. 14, 211–216 (2005)
Nagel, A. C., Maier, D. & Preiss, A. Green fluorescent protein as a convenient and versatile marker for studies on functional genomics in Drosophila . Dev. Genes Evol. 212, 93–98 (2002)
Strunkelnberg, M. et al. rst and its paralogue kirre act redundantly during embryonic muscle development in Drosophila . Development 128, 4229–4239 (2001)
Acknowledgements
We thank F. Evers, Z. Cseresnyes, M. Guerra and E. Salvador for technical assistance, and S. Abmayr, L. Cooley, C. Doe, M. Affolter, K. Fischbach and K. Tryggvason for reagents. We thank V. Hartenstein, M. Inamdar, A. Woolf, I. Miguel-Aliaga, F. Evers, M. Landgraf and members of the Skaer laboratory for discussions, and E. Knust and W. B. Huttner for their support. This work was supported by Wellcome Trust grants awarded to H.S. (072441 and 079221; H.W., B.D. and H.S.); Deutsche Forschungsgemeinschaft SFB 590 awarded to E. Knust (F.G.) and ARC 1242 (H.W., B.D., H.S. and F.G.); an MEC grant awarded to M.R.-G. (BFU2007-62201; S.P.-S. and M.R.-G.); a Fundación Ramón Areces grant to the CBMSO (M.R.-G.); EC grant LSHG-CT-2004-511978 to MYORES (M.R.-G.); and an FPU fellowship from the MEC awarded to A.G.-L.
Author Contributions B.D., H.S. and M.R.-G. designed and directed the project. B.D., H.W., M.R.-G. and S.P.-S. performed the experiments. F.G. and M.W.-B. provided technical assistance. A.G.-L. and R.A. provided materials. B.D. and H.S. wrote the paper. All authors discussed results and commented on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-4 with Legends, Supplementary Table 1 and Supplementary Notes (PDF 7650 kb)
Rights and permissions
About this article
Cite this article
Weavers, H., Prieto-Sánchez, S., Grawe, F. et al. The insect nephrocyte is a podocyte-like cell with a filtration slit diaphragm. Nature 457, 322–326 (2009). https://doi.org/10.1038/nature07526
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07526
This article is cited by
-
A SNARE protective pool antagonizes APOL1 renal toxicity in Drosophila nephrocytes
Cell & Bioscience (2023)
-
A multiscale approach reveals elaborate circulatory system and intermittent heartbeat in velvet worms (Onychophora)
Communications Biology (2023)
-
Functional-metabolic coupling in distinct renal cell types coordinates organ-wide physiology and delays premature ageing
Nature Communications (2023)
-
Insights into human kidney function from the study of Drosophila
Pediatric Nephrology (2023)
-
Drosophila melanogaster: a simple genetic model of kidney structure, function and disease
Nature Reviews Nephrology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.