Abstract
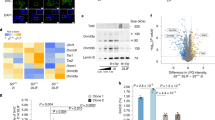
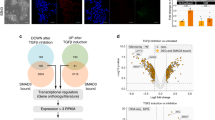
Recently, we identified in adult tissues a population of Oct4+SSEA-1+Sca-1+lin−CD45− very small embryonic-like stem cells (VSELs). First, to address recent controversies on Oct4 expression in cells isolated from adult organs, we show here evidence that Oct4 promoter in bone marrow (BM)-derived VSELs has an open chromatin structure and is actively transcribed. Next, to explain VSELs quiescence and lack of teratoma formation, we demonstrate a unique DNA methylation pattern at some developmentally crucial imprinted genes, showing hypomethylation/erasure of imprints in paternally methylated and hypermethylation of imprints in maternally methylated ones. These epigenetic characteristics leading to upregulation in VSELs of H19 and p57KIP2 (also known as Cdkn1c) and repression of Igf2 and Rasgrf1 explain VSEL's quiescent status. Interestingly, this unique pattern in imprinted gene methylation is reverted in cocultures with a C2C12 supportive cell-line when VSELs are induced to form VSEL-derived spheres (VSEL-DSs) enriched for stem cells able to differentiate into all three germ layers. Therefore, we suggest that the proliferative/developmental potential of Oct4+ VSELs is epigenetically regulated by expression of Oct4 and some imprinted genes, and postulate that restoring the proper methylation pattern of imprinted genes will be a crucial step for using these cells in regenerative medicine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Evans MJ, Kaufman MH . Establishment in culture of pluripotential cells from mouse embryos. Nature 1981; 292: 154–156.
Tesar PJ, Chenoweth JG, Brook FA, Davies TJ, Evans EP, Mack DL et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature 2007; 448: 196–199.
Matsui Y, Zsebo K, Hogan BLM . Derivation of pluripotential embryonic stem cells from murine primordial germ cells in culture. Cell 1992; 70: 841–847.
Takahashi K, Yamanaka S . Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006; 126: 663–676.
Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 2007; 448: 318–324.
Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 2002; 418: 41–49.
D'Ippolito G, Diabira S, Howard GA, Menei P, Roos BA, Schiller PC . Marrow-isolated adult multilineage inducible (MIAMI) cells, a unique population of postnatal young and old human cells with extensive expansion and differentiation potential. J Cell Sci 2004; 117: 2971–2981.
Liedtke S, Enczmann Jg, Waclawczyk S, Wernet P, Kogler G . Oct4 and its pseudogenes confuse stem cell research. Cell Stem Cell 2007; 1: 364–366.
Lengner CJ, Camargo FD, Hochedlinger K, Welstead GG, Zaidi S, Gokhale S et al. Oct4 expression is not required for mouse somatic stem cell self-renewal. Cell Stem Cell 2007; 1: 403–415.
Kucia M, Reca R, Campbell FR, Zuba-Surma E, Majka M, Ratajczak J et al. A population of very small embryonic-like (VSEL) CXCR4+SSEA-1+Oct-4+ stem cells identified in adult bone marrow. Leukemia 2006; 20: 857–869.
Kucia M, Halasa M, Wysoczynski M, Baskiewicz-Masiuk M, Moldenhawer S, Zuba-Surma E et al. Morphological and molecular characterization of novel population of CXCR4+ SSEA-4+ Oct-4+ very small embryonic-like cells purified from human cord blood - preliminary report. Leukemia 2006; 21: 297–303.
Kucia M, Zhang YP, Reca R, Wysoczynski M, Machalinski B, Majka M et al. Cells enriched in markers of neural tissue-committed stem cells reside in the bone marrow and are mobilized into the peripheral blood following stroke. Leukemia 2005; 20: 18–28.
Wojakowski W, Tendera M, Kucia M, Zuba-Surma E, Paczkowska E, Ciosek J et al. Mobilization of bone marrow-derived Oct-4+ SSEA-4+ very small embryonic-like stem cells in patients with acute myocardial infarction. J Am Coll Cardio 2009; 53: 1–9.
Paczkowska E, Kucia M, Koziarska D, Halasa M, Safranow K, Masiuk M et al. Clinical evidence that very small embryonic-like stem cells are mobilized into peripheral blood in patients after stroke. Stroke 2009; 40: 1237–1244.
Ratajczak MZ, Machalinski B, Wojakowski W, Ratajczak J, Kucia M . A hypothesis for an embryonic origin of pluripotent Oct-4+ stem cells in adult bone marrow and other tissues. Leukemia 2007; 21: 860–867.
Reik W, Walter J . Genomic imprinting: parental influence on the genome. Nat Rev Genet 2001; 2: 21–32.
Yamazaki Y, Mann MR, Lee SS, Marh J, McCarrey JR, Yanagimachi R et al. Reprogramming of primordial germ cells begins before migration into the genital ridge, making these cells inadequate donors for reproductive cloning. Proc Natl Acad Sci USA 2003; 100: 12207–12212.
Horii T, Kimura M, Morita S, Nagao Y, Hatada I . Loss of genomic imprinting in mouse parthenogenetic embryonic stem cells. Stem Cells 2008; 26: 79–88.
Pannetier Ml, Feil R . Epigenetic stability of embryonic stem cells and developmental potential. Trends Biotechnol 2007; 25: 556–562.
Delaval K, Feil R . Epigenetic regulation of mammalian genomic imprinting. Curr Opin Genet Dev 2004; 14: 188–195.
Lopes S, Lewis A, Hajkova P, Dean W, Oswald J, Forne T et al. Epigenetic modifications in an imprinting cluster are controlled by a hierarchy of DMRs suggesting long-range chromatin interactions. Hum Mol Genet 2003; 12: 295–305.
Kobayashi H, Suda C, Abe T, Kohara Y, Ikemura T, Sasaki H . Bisulfite sequencing and dinucleotide content analysis of 15 imprinted mouse differentially methylated regions (DMRs): paternally methylated DMRs contain less CpGs than maternally methylated DMRs. Cytogenet Genome Res 2006; 113: 130–137.
Mager J, Montgomery ND, de Villena FP-M, Magnuson T . Genome imprinting regulated by the mouse Polycomb group protein Eed. Nat Genet 2003; 33: 502–507.
Fournier C, Goto Y, Ballestar E, Delaval K, Hever AM, Esteller M et al. Allele-specific histone lysine methylation marks regulatory regions at imprinted mouse genes. EMBO J 2002; 21: 6560–6570.
O'Neill LP, VerMilyea MD, Turner BM . Epigenetic characterization of the early embryo with a chromatin immunoprecipitation protocol applicable to small cell populations. Nat Genet 2006; 38: 835–841.
Carr IM, Valleley EMA, Cordery SF, Markham AF, Bonthron DT . Sequence analysis and editing for bisulphite genomic sequencing projects. Nucl Acids Res 2007; 35: e79.
Feldman N, Gerson A, Fang J, Li E, Zhang Y, Shinkai Y et al. G9a-mediated irreversible epigenetic inactivation of Oct-3/4 during early embryogenesis. Nat Cell Biol 2006; 8: 188–194.
Margueron R, Trojer P, Reinberg D . The key to development: interpreting the histone code? Curr Opin Genet Dev 2005; 15: 163–176.
Kono T, Obata Y, Wu Q, Niwa K, Ono Y, Yamamoto Y et al. Birth of parthenogenetic mice that can develop to adulthood. Nature 2004; 428: 860–864.
Hajkova P, Erhardt S, Lane N, Haaf T, El-Maarri O, Reik W et al. Epigenetic reprogramming in mouse primordial germ cells. Mech Dev 2002; 117: 15–23.
Pant V, Mariano P, Kanduri C, Mattsson A, Lobanenkov V, Heuchel R et al. The nucleotides responsible for the direct physical contact between the chromatin insulator protein CTCF and the H19 imprinting control region manifest parent of origin-specific long-distance insulation and methylation-free domains. Genes Dev 2003; 17: 586–590.
Yoon B, Herman H, Hu B, Park YJ, Lindroth A, Bell A et al. Rasgrf1 imprinting is regulated by a CTCF-dependent methylation-sensitive enhancer blocker. Mol Cell Biol 2005; 25: 11184–11190.
Shovlin TC, Durcova-Hills G, Surani A, McLaren A . Heterogeneity in imprinted methylation patterns of pluripotent embryonic germ cells derived from pre-migratory mouse germ cells. Dev Biol 2008; 313: 674–681.
Zhu T-N, He H-J, Kole S, D′Souza T, Agarwal R, Morin PJ et al. Filamin A-mediated down-regulation of the exchange factor Ras-GRF1 correlates with decreased matrix metalloproteinase-9 expression in human melanoma cells. J Biol Chem 2007; 282: 14816–14826.
Lefebvre L, Viville S, Barton SC, Ishino F, Keverne EB, Surani MA . Abnormal maternal behaviour and growth retardation associated with loss of the imprinted gene Mest. Nat Genet 1998; 20: 163–169.
Chen T, Ueda Y, Dodge JE, Wang Z, Li E . Establishment and maintenance of genomic methylation patterns in mouse embryonic stem cells by Dnmt3a and Dnmt3b. Mol Cell Biol 2003; 23: 5594–5605.
Bourc'his D, Xu G-L, Lin C-S, Bollman B, Bestor TH . Dnmt3 L and the establishment of maternal genomic imprints. Science 2001; 294: 2536–2539.
Durcova-Hills G, Tang F, Doody G, Tooze R, Surani MA . Reprogramming primordial germ cells into pluripotent stem cells. PLoS ONE 2008; 3: e3531.
Eggenschwiler J, Ludwig T, Fisher P, Leighton PA, Tilghman SM, Efstratiadis A . Mouse mutant embryos overexpressing IGF-II exhibit phenotypic features of the Beckwith-Wiedemann and Simpson-Golabi-Behmel syndromes. Genes Dev 1997; 11: 3128–3142.
Hao Y, Crenshaw T, Moulton T, Newcomb E, Tycko B . Tumour-suppressor activity of H19 RNA. Nature 1993; 365: 764–767.
Ludwig T, Eggenschwiler J, Fisher P, D'Ercole AJ, Davenport ML, Efstratiadis A . Mouse mutants lacking the type 2 IGF receptor (IGF2R) are rescued from perinatal lethality inIgf2 and Igf1r null backgrounds. Dev Biol 1996; 177: 517–535.
Font de Mora J, Esteban LM, Burks DJ, Nunez A, Garces C, Garcia-Barrado MJ et al. Ras-GRF1 signaling is required for normal beta-cell development and glucose homeostasis. EMBO J 2003; 22: 3039–3049.
Scandura JM, Boccuni P, Massague J, Nimer SD . Transforming growth factor β-induced cell cycle arrest of human hematopoietic cells requires p57KIP2 up-regulation. Proc Natl Acad Sci USA 2004; 101: 15231–15236.
Umemoto T, Yamato M, Nishida K, Yang J, Tano Y, Okano T . p57Kip2 is expressed in quiescent mouse bone marrow side population cells. Biochem Biophys Res Commun 2005; 337: 14–21.
Acknowledgements
We thank Chris Worth, Rui Liu and Izabela Klich for technical support; and Dr Hal Broxmeyer, Dr Jan Nolta, Dr Miodrag Stojkovic and Dr Mervin Yoder for critical comments. This work was supported by NIH P20RR018733 from the National Center for Research Resources to MK and by NIH R01 CA106281-01, NIH R01 DK074720 and Stella and Henry Endowment to MZR.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu)
Supplementary information
Rights and permissions
About this article
Cite this article
Shin, D., Zuba-Surma, E., Wu, W. et al. Novel epigenetic mechanisms that control pluripotency and quiescence of adult bone marrow-derived Oct4+ very small embryonic-like stem cells. Leukemia 23, 2042–2051 (2009). https://doi.org/10.1038/leu.2009.153
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2009.153
Keywords
This article is cited by
-
Initiation of Cancer: The Journey From Mutations in Somatic Cells to Epigenetic Changes in Tissue-resident VSELs
Stem Cell Reviews and Reports (2024)
-
Proteomic Analysis of Murine Bone Marrow Very Small Embryonic-like Stem Cells at Steady-State Conditions and after In Vivo Stimulation by Nicotinamide and Follicle-Stimulating Factor Reflects their Germ-Lineage Origin and Multi Germ Layer Differentiation Potential
Stem Cell Reviews and Reports (2023)
-
Preventing/Reversing Adverse Effects of Endocrine Disruption on Mouse Testes by Normalizing Tissue Resident VSELs
Stem Cell Reviews and Reports (2023)
-
In Vitro Simulated Neuronal Environmental Conditions Qualify Umbilical Cord Derived Highly Potent Stem Cells for Neuronal Differentiation
Stem Cell Reviews and Reports (2023)
-
Testicular cancer in mice: interplay between stem cells and endocrine insults
Stem Cell Research & Therapy (2022)