Abstract
Objective:
Studies in adults indicate that ampicillin, in a dose-dependent manner, impairs platelet function and moderately prolongs the bleeding time (generally by 60 to 90 s). Unlike aspirin, the inhibition induced by ampicillin involves both reversible and irreversible mechanisms and is not observed immediately after initial dosing (generally requiring approximately 24 h). Ampicillin is administered commonly to neonatal intensive care unit (NICU) patients, but its effect on bleeding time in this population has not been reported earlier.
Study Design:
We performed neonatal template bleeding times and platelet function analyzer (PFA)-100 tests on 15 NICU patients before and at various intervals after intravenous ampicillin dosing.
Result:
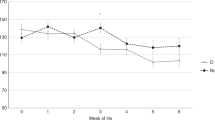
Neonates were only studied if no beta-lactam antibiotics were administered to their mother during labor, and if they had ampicillin ordered by the clinician at a dose of 50 to 100 mg kg–1 every 12 h. Subjects ranged from 33 to 41 weeks gestation and weighed 1760 to 3835 g. Bleeding times before the first ampicillin dose (n=15) averaged 134 s (95% confidence interval (CI), 120 to 148 s) and PFA-100 times averaged 123 s (95% CI, 96 to 149 s). After the first dose of ampicillin (n=5), bleeding times and PFA-100 times did not increase, but after the third (n=5) and fourth doses (n=4) bleeding times lengthened by an average of 60 s (95% CI, 37 to 83 s, P<0.001) and PFA-100 times lengthened by an average of 20 s (95% CI, −20 to 60 s, P=0.15).
Conclusion:
Ampicillin administered intravenously to NICU patients prolongs the bleeding time, with a magnitude-of-effect and time-to-effect similar to that shown earlier in adults.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Sheffield MS, Schmutz N, Lambert DK, Henry E, Christensen RD . Ibuprofen lysine administration to neonates with a patent ductus arteriosus: effect on platelet plug formation assessed by in vivo and in vitro measurements. J Perinatol 2009; 29: 39–43.
Barradas MA, Jeremy JY, Mikhailidis DP, Dandona P . The effect of tiaprofenic acid and indomethacin administration of human platelet function. Inflammation Res 1989; 26: 386–390.
Gibbs NM, Weightman WM, Thackray NM, Michalopoulos N, Weidmann C . The effects of recent aspirin ingestion on platelet function in cardiac surgical patients. J Cardiothorac Vasc Anesth 2001; 15: 55–59.
Brown III CH, Bradshaw MJ, Natelson EA, Alfrey Jr CP, Williams Jr TW . Defective platelet function following the administration of penicillin compounds. Blood 1976; 47 (6): 949–956.
Wisløff F, Larsen JP, Dahle A, Lie M, Godal HC . Effect of prophylactic high-dose treatment with ampicillin and cloxacillin on bleeding time and bleeding in patients undergoing elective vascular surgery. Scand J Haematol 1983; 31 (2): 97–101.
Pillgram-Larsen J, Wisløff F, Jørgensen JJ, Godal HC, Semb G . Effect of high-dose ampicillin and cloxacillin on bleeding time and bleeding in open-heart surgery. Scand J Thorac Cardiovasc Surg 1985; 19 (1): 45–48.
Fass RJ, Copelan EA, Brandt JT, Moeschberger ML, Ashton JJ . Platelet-mediated bleeding caused by broad-spectrum penicillins. J Infect Dis 1987; 155: 1242–1248.
Burroughs SF, Johnson GJ . Beta-lactam antibiotic-induced platelet dysfunction: evidence for irreversible inhibition of platelet activation in vitro and in vivo after prolonged exposure to penicillin. Blood 1990; 75 (7): 1473–1480.
Burroughs SF, Johnson GJ . Beta-lactam antibiotics inhibit agonist-stimulated platelet calcium influx. Thromb Haemost 1993; 69 (5): 503–508.
Del Vecchio A, Sola MC . Performing and interpreting the bleeding time in the neonatal intensive care unit. Clin Perinatol 2000; 27: 643–654.
Del Vecchio A . Use of the bleeding time in the neonatal intensive care unit. Acta Paediatr Suppl 2002; 91: 82–86.
Del Vecchio A, Latini G, Henry E, Christensen RD . Template bleeding times of 240 neonates born at 24 to 41weeks gestation. J Perinatol 2008; 28: 427–431.
Andrew M, Castle V, Mitchell L, Paes B . Modified bleeding time in the infant. Am J Hematol 1989; 30: 190–191.
Borzini P, Laxxaro A, Mazzucco L, Papili F . The in-vitro bleeding time for the screening of platelet function in the newborn: assessment of a normal reference range. Eur J Pediatr 2001; 160: 199–200.
Israels SJ, Cheang T, McMillan-Ward EM, Cheang M . Evaluation of primary hemostasis in neonates with a new in vitro platelet function analyzer. J Pediatr 2001; 138: 116–119.
Boudewijns M, Raes M, Peeters V, Mewis A, Cartuvvels R, Magerman K et al. Evaluation of platelet function on cord blood in 80 healthy term neonates using the Platelet Function Analyser (PFA-100); shorter in vitro bleeding times in neonates than adults. Eur J Pediatr 2003; 162: 212–213.
Shah U, Ma AD . Tests of platelet function. Curr Opin Hematol 2007; 14: 432–437.
Roschitz B, Sudi K, Köstenberger M, Muntean W . Shorter PFA-100 closure times in neonates than in adults: role of red cells, white cells, platelets and von Willebrand factor. Acta Paediatr 2001; 90: 664–670.
Perneby C, Wallén NH, Hofman-Bang C, Tornvall P, Ivert T, Li N et al. Effect of clopidogrel treatment on stress-induced platelet activation and myocardial ischemia in aspirin-treated patients with stable coronary artery disease. Thromb Haemost 2007; 98: 1316–1322.
Knijff-Dutmer EA, Kalsbeek-Batenburg EM, Koerts J, van de Laar MA . Platelet function is inhibited by non-selective non-steroidal anti-inflammatory drugs but not by cyclo-oxygenase-2-selective inhibitors in patients with rheumatoid arthritis. Rheumatol (Oxford) 2002; 41: 458–461.
Bolisetty S, Patole S, Koh G, Stalewski H, Whitehall J . Neonatal acute haemorrhagic gastritis and antenatal exposure to indomethacin for tocolysis. ANZ J Surg 2001; 71: 122–123.
Chirico G, Gasparoni MC, Rondini G . Exchange transfusion for indomethacin-induced hemorrhagic complication in the neonate. J Pediatr 1985; 107: 312.
Peskine F, Despaux E, Rodiere M, Balmayer B, Kassis J, Fuseau E et al. Pharmacokinetic and clinical investigation of amoxicillin administered intravenously in the newborn. Pathol Biol (Paris) 1982; 30 (6 Pt 2): 476–480.
Kaplan JM, McCracken Jr GH, Horton LJ, Thomas ML, Davis N . Pharmacologic studies in neonates given large dosages of ampicillin. J Pediatr 1974; 84 (4): 571–577.
George TN, Johnson KJ, Bates JN, Segar JL . The effect of inhaled nitric oxide therapy on bleeding time and platelet aggregation in neonates. J Pediatr 1998; 132 (4): 731–734.
Keh D, Kürer I, Dudenhausen JW, Woltmann W, Falke KJ, Gerlach H . Response of neonatal platelets to nitric oxide in vitro. Intensive Care Med 2001; 27 (1): 283–286.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Sheffield, M., Lambert, D., Henry, E. et al. Effect of ampicillin on the bleeding time of neonatal intensive care unit patients. J Perinatol 30, 527–530 (2010). https://doi.org/10.1038/jp.2009.192
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2009.192
Keywords
This article is cited by
-
Ampicillin dosing in premature infants for early-onset sepsis: exposure-driven efficacy, safety, and stewardship
Journal of Perinatology (2022)
-
Results of a two-center, before and after study of piperacillin–tazobactam versus ampicillin and gentamicin as empiric therapy for suspected sepsis at birth in neonates ⩽1500 g
Journal of Perinatology (2013)
-
Effect of ampicillin on bleeding time in very low birth-weight neonates during the first week after birth
Journal of Perinatology (2011)