Abstract
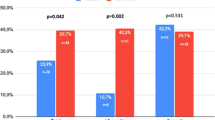
The aim of this post-hoc pooled-data analysis was to evaluate the effects of once-daily tadalafil in men with mild or moderate ED and aged <50 or ⩾50 years. Data from three randomized controlled trials were analyzed. Analysis of covariance models included a term for ED severity and age group. The three coprimary outcome measures in the base studies were changes from baseline to end point in the mean International Index of Erectile Function Erectile Function (IIEF-EF) domain score and the mean per-patient percentage successful vaginal penetration and intercourse attempts. Irrespective of baseline severity, once-daily tadalafil 5 mg for 12 weeks compared with placebo significantly increased the mean: (1) IIEF-EF by 6.8; (2) percent successful penetration attempts from 70.1 to 91.3%; and (3) percent successful intercourse attempts from 33.4 to 76.8% (each P<0.001). Treatment-by-age-group interaction P-values for all three coprimary efficacy end points exceeded 0.10, indicating that tadalafil treatment effects did not differ by age <50 vs ⩾50 years. Tadalafil was generally well tolerated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Grover SA, Lowensteyn I, Kaouache M, Marchand S, Coupal L, DeCarolis E et al. The prevalence of erectile dysfunction in the primary care setting: importance of risk factors for diabetes and vascular disease. Arch Intern Med 2006; 166: 213–219.
Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ, McKinlay JB . Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol 1994; 151: 54–61.
Kubin M, Wagner G, Fugl-Meyer AR . Epidemiology of erectile dysfunction. Int J Impot Res 2003; 15: 63–71.
Nicolosi A, Glasser DB, Moreira ED, Villa M . Prevalence of erectile dysfunction and associated factors among men without concomitant diseases: a population study. Int J Impot Res 2003; 15: 253–257.
Wu CJ, Hsieh JT, Lin JS, Hwang TI, Jiann BP, Huang ST et al. Comparison of prevalence between self-reported erectile dysfunction and erectile dysfunction as defined by five-item International Index of Erectile Function in Taiwanese men older than 40 years. Urology 2007; 69: 743–747.
Laumann EO, West S, Glasser D, Carson C, Rosen R, Kang JH . Prevalence and correlates of erectile dysfunction by race and ethnicity among men aged 40 or older in the United States: from the male attitudes regarding sexual health survey. J Sex Med 2007; 4: 57–65.
Heruti R, Shochat T, Tekes-Manova D, Ashkenazi I, Justo D . Prevalence of erectile dysfunction among young adults: results of a large-scale survey. J Sex Med 2004; 1: 284–291.
Laumann EO, Paik A, Rosen RC . Sexual dysfunction in the United States: prevalence and predictors. JAMA 1999; 281: 537–544.
Shabsigh R, Perelman MA, Laumann EO, Lockhart DC . Drivers and barriers to seeking treatment for erectile dysfunction: a comparison of six countries. BJU Int 2004; 94: 1055–1065.
Rosen RC, Fisher WA, Eardley I, Niederberger C, Nadel A, Sand M . The multinational Men’s Attitudes to Life Events and Sexuality (MALES) study: I. Prevalence of erectile dysfunction and related health concerns in the general population. Curr Med Res Opin 2004; 20: 607–617.
Braun M, Wassmer G, Klotz T, Reifenrath B, Mathers M, Engelmann U . Epidemiology of erectile dysfunction: results of the ‘Cologne Male Survey’. Int J Impot Res 2000; 12: 305–311.
Price D, Hackett G . Management of erectile dysfunction in diabetes: an update for 2008. Curr Diab Rep 2008; 8: 437–443.
Bacon CG, Mittleman MA, Kawachi I, Giovannucci E, Glasser DB, Rimm EB . Sexual function in men older than 50 years of age: results from the health professionals follow-up study. Ann Intern Med 2003; 139: 161–168.
Heidler S, Temml C, Broessner C, Mock K, Rauchenwald M, Madersbacher S et al. Is the metabolic syndrome an independent risk factor for erectile dysfunction? J Urol 2007; 177: 651–654.
Carson CC, Rajfer J, Eardley I, Carrier S, Denne JS, Walker DJ et al. The efficacy and safety of tadalafil: an update. BJU Int 2004; 93: 1276–1281.
Hellstrom WJ, Gittelman M, Karlin G, Segerson T, Thibonnier M, Taylor T et al. Vardenafil for treatment of men with erectile dysfunction: efficacy and safety in a randomized, double-blind, placebo-controlled trial. J Androl 2002; 23: 763–771.
Goldstein I, Lue TF, Padma-Nathan H, Rosen RC, Steers WD, Wicker PA . Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. N Engl J Med 1998; 338: 1397–1404.
Rowland DL, Incrocci L, Slob AK . Aging and sexual response in the laboratory in patients with erectile dysfunction. J Sex Marital Ther 2005; 31: 399–407.
Rowland DL, Greenleaf W, Mas M, Myers L, Davidson JM . Penile and finger sensory thresholds in young, aging, and diabetic males. Arch Sex Behav 1989; 18: 1–12.
Forgue ST, Patterson BE, Bedding AW, Payne CD, Phillips DL, Wrishko RE et al. Tadalafil pharmacokinetics in healthy subjects. Br J Clin Pharmacol 2006; 61: 280–288.
Costa P, Grivel T, Gehchan N . Tadalafil once daily in the management of erectile dysfunction: patient and partner perspectives. Patient Prefer Adherence 2009; 3: 105–111.
Seftel AD, Buvat J, Althof SE, McMurray JG, Zeigler HL, Burns PR et al. Improvements in confidence, sexual relationship and satisfaction measures: results of a randomized trial of tadalafil 5 mg taken once daily. Int J Impot Res 2009; 21: 240–248.
Rubio-Aurioles E, Kim ED, Rosen RC, Porst H, Burns P, Zeigler H et al. Impact on erectile function and sexual quality of life of couples: a double-blind, randomized, placebo-controlled trial of tadalafil taken once daily. J Sex Med 2009; 6: 1314–1323.
Wrishko R, Sorsaburu S, Wong D, Strawbridge A, McGill J . Safety, efficacy, and pharmacokinetic overview of low-dose daily administration of tadalafil. J Sex Med 2009; 6: 2039–2048.
Donatucci CF, Wong DG, Giuliano F, Glina S, Dowsett SA, Watts S et al. Efficacy and safety of tadalafil once daily: considerations for the practical application of a daily dosing option. Curr Med Res Opin 2008; 24: 3383–3392.
Shabsigh R, Broderick GA, Althof S, Natanegara F, Wong DG . Daily patterns of sexual intercourse attempts by men with erectile dysfunction treated with tadalafil: influence of age and marital status. Int J Impot Res 2009; 21: 285–291.
Porst H, Giuliano F, Glina S, Ralph D, Casabé AR, Elion-Mboussa A et al. Evaluation of the efficacy and safety of once-a-day dosing of tadalafil 5mg and 10mg in the treatment of erectile dysfunction: results of a multicenter, randomized, double-blind, placebo-controlled trial. Eur Urol 2006; 50: 351–359.
Rajfer J, Aliotta PJ, Steidle CP, Fitch WP III, Zhao Y, Yu A . Tadalafil dosed once a day in men with erectile dysfunction: a randomized, double-blind, placebo-controlled study in the US. Int J Impot Res 2007; 19: 95–103.
Eli Lilly Cialis (tadalafil) tablets, film coated for oral use (US package insert). Available at http://pi.lilly.com/us/cialis-pi.pdf2011 (accessed 19 November 2012).
Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A . The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997; 49: 822–830.
Cappelleri JC, Rosen RC, Smith MD, Mishra A, Osterloh IH . Diagnostic evaluation of the erectile function domain of the International Index of Erectile Function. Urology 1999; 54: 346–351.
Giuliano F, Donatucci C, Montorsi F, Auerbach S, Karlin G, Norenberg C et al. Vardenafil is effective and well-tolerated for treating erectile dysfunction in a broad population of men, irrespective of age. BJU Int 2005; 95: 110–116.
Fink HA, Mac DR, Rutks IR, Nelson DB, Wilt TJ . Sildenafil for male erectile dysfunction: a systematic review and meta-analysis. Arch Intern Med 2002; 162: 1349–1360.
Rosen RC, Allen KR, Ni X, Araujo AB . Minimal clinically important differences in the erectile function domain of the International Index of Erectile Function scale. Eur Urol 2011; 60: 1010–1016.
Sharlip ID, Shumaker BP, Hakim LS, Goldfischer E, Natanegara F, Wong DG . Tadalafil is efficacious and well tolerated in the treatment of erectile dysfunction (ED) in men over 65 years of age: results from multiple observations in men with ED in National Tadalafil Study in the United States. J Sex Med 2008; 5: 716–725.
Morgentaler A, Barada J, Niederberger C, Donatucci C, Garcia CS, Natanegara F et al. Efficacy and safety of tadalafil across ethnic groups and various risk factors in men with erectile dysfunction: Use of a novel noninferiority study design. J Sex Med 2006; 3: 492–503.
Acknowledgements
Assistance in manuscript preparation was provided by Stephen W Gutkin, Rete Biomedical Communications Corp. (Wyckoff, NJ, USA), with support from Eli Lilly and Company (Indianapolis, IN, USA), which had a role in the designs of both the base studies and the present analysis; data acquisition and interpretation; drafting of the manuscript; and the decision to report the findings. Statistical assistance was provided by Sunny Li, MS, inVentiv Clinical (Somerset, NJ, USA), with support from Eli Lilly, as well as from Cheolho Park, PhD, of Eli Lilly. We also thank Anne Esler, PhD, Senior Biostatistician, PharmaNet/i3 inVentiv Clinical (Indianapolis, IN, USA), for reviewing and commenting on the manuscript.
Author contributions
Conception and design: all authors. Acquisition of data: Drs Seftel, Shinghal, Kim and Samuels, along with the clinical investigators in the base studies. Analysis and interpretation of data: Dr Ni conducted statistical analyses (with assistance from Ms Li and Dr Park), and all authors interpreted data. Drafting the article: Drs Seftel, Shinghal and Kim with assistance from Mr Gutkin. Revising it for intellectual content: all authors. Final approval of the completed article: all authors. Dr Seftel had access to all data analyzed and takes responsibility for the analysis and this report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Seftel is a paid consultant to Abbott, Auxilium, Eli Lilly, Endo and Pfizer, and has received research grants from Eli Lilly and Auxilium. Dr Shinghal is an advisor to Eli Lilly and PercSys, a clinical investigator for GlaxoSmithKline, and a speaker for Astellas. Dr Kim is an advisor, recipient of honoraria, and a paid consultant/investigator for Eli Lilly. He is also a speaker for Astellas, Auxilium and Watson. Dr Samuels holds stocks (or options) in Eli Lilly (minor). Drs Ni and Burns are employees of, and stock (or option) holders in, Eli Lilly (minor).
Appendix
Appendix
Eligibility criteria for subjects enrolled in the base studies,23, 27, 28
Inclusion criteria
-
Age ⩾18 years.
-
Minimum 3-month history of ED.
-
ED of any severity or etiology.
Exclusion criteria
-
ED secondary to other primary sexual disorders (e.g., premature ejaculation) or to untreated endocrine disorder (e.g., hypogonadism, hypothyroidism).
-
History of (non-nerve-sparing) radical prostatectomy or other pelvic surgery with subsequent failure to achieve erection
-
Clinically significant penile deformity or penile implant (prosthesis).
-
Clinically significant renal or hepatobiliary disease.
-
Significant spinal-cord or other central-nervous-system injury (e.g., stroke) within 6 months before screening
-
History of human immunodeficiency virus infection.
-
Current treatment with nitrates, cancer chemotherapy,23, 28 or anti-androgens23, 28 (finasteride and dutasteride were permitted in Study H6D-MC-LVGH23).
-
Recent histories of unstable or poorly controlled cardiovascular disorders, including:
-
Unstable angina within the past 6 months.
-
Congestive heart failure (New York Heart Association Class II or higher) within prior 6 months.
-
Myocardial infarction, coronary artery bypass grafting, percutaneous coronary intervention within the prior 90 days.
-
-
Supraventricular arrhythmia with uncontrolled ventricular response at rest, or history of spontaneous or induced sustained ventricular tachycardia, despite medical or device therapy; or the presence of an automatic internal cardioverter-defibrillator.
-
Extremes of blood pressure at screening: 90 mm Hg>systolic blood pressure>170 mm Hg or 50 mm Hg>diastolic blood pressure>100 mm Hg or malignant hypertension.
-
Hemoglobin A1c>13%.
-
History of alcohol, drug, or substance abuse within the prior 6 months.
Rights and permissions
About this article
Cite this article
Seftel, A., Shinghal, R., Kim, E. et al. Retrospective analysis of the efficacy and safety of once-daily tadalafil in patient subgroups: men with mild vs moderate ED and aged <50 vs ⩾50 years. Int J Impot Res 25, 91–98 (2013). https://doi.org/10.1038/ijir.2012.40
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijir.2012.40