Abstract
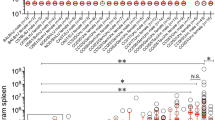
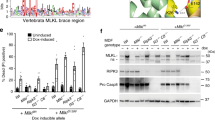
The outcome of infection with Salmonella Typhimurium in mouse models of human typhoid fever is dependent upon a coordinated complex immune response. A panel of recombinant congenic strains (RCS) derived from reciprocal backcross of A/J and C57BL/6J mice was screened for their susceptibility to Salmonella infection and two susceptibility loci, Ity4 (Immunity to Typhimurium locus 4) and Ity5, were identified. We validated Ity5 in a genetic environment free of the impact of Ity4 using a cross between A/J and 129S6. Using a time-series analysis of genome-wide transcription during infection, comparing A/J with AcB60 mice having a C57BL/6J-derived Ity5 interval, we have identified the differential expression of the positional candidate gene Cd40, Cd40-associated signaling pathways, and the differential expression of numerous genes expressed in neutrophils. CD40 is known to coordinate T cell-dependent B-cell responses and myeloid cell activation. In fact, CD40 signaling is altered in A/J mice as seen by impaired IgM upregulation during infection, decreased Ig class switching, neutropenia, reduced granulocyte recruitment in response to infection and inflammation, and decreased ERK1/2 activity. These results suggest that altered CD40 signaling and granulocyte recruitment in response to infection are responsible for the Ity5-associated Salmonella susceptibility of A/J mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Accession codes
References
Pegues DA, Hohmann EL, Miller SI . Salmonella Including S. typhi. In: Blaser MJ, Smith PD, Ravdin JI, Greenberg HB, Guerrant RL (eds). Infections of the gastrointestinal tract. Raven Press: New York, NY, USA, 1995, pp 785–809.
Vidal S, Tremblay ML, Govoni G, Gauthier S, Sebastiani G, Malo D et al. The Ity/Lsh/Bcg locus: natural resistance to infection with intracellular parasites is abrogated by disruption of the Nramp1 gene. J Exp Med 1995; 182: 655–666.
Qureshi ST, Skamene E, Malo D . Comparative genomics and host resistance against infectious diseases. Emerg Infect Dis 1999; 5: 36–47.
Qureshi ST, Larivière L, Sebastiani G, Clermont S, Skamene E, Gros P et al. A high-resolution map in the chromosomal region surrounding the Lps locus. Genomics 1996; 31: 283–294.
Sancho-Shimizu V, Khan R, Mostowy S, Larivière L, Wilkinson R, Riendeau N et al. Molecular genetic analysis of two loci (Ity2 and Ity3) involved in the host response to infection with Salmonella typhimurium using congenic mice and expression profiling. Genetics 2007; 177: 1125–1139.
Richer E, Prendergast C, Zhang D-E, Qureshi ST, Vidal SM, Malo D . N-ethyl-N-nitrosourea-induced mutation in ubiquitin-specific peptidase 18 causes hyperactivation of IFN-αβ signaling and suppresses STAT4-induced IFN-γ production, resulting in increased susceptibility to Salmonella typhimurium. J Immunol 2010; 185: 3593–3601.
Eva MM, Yuki KE, Dauphinee SM, Schwartzentruber JA, Pyzik M, Paquet M et al. Altered IFN-γ-mediated immunity and transcriptional expression patterns in N-Ethyl-N-nitrosourea-induced STAT4 mutants confer susceptibility to acute typhoid-like disease. J Immunol 2014; 192: 259–270.
Khan R, Sancho-Shimizu V, Prendergast C, Roy MF, Loredo-Osti JC, Malo D . Refinement of the genetics of the host response to Salmonella infection in MOLF/Ei: regulation of type 1 IFN and TRP3 pathways by Ity2. Genes Immun 2012; 13: 175–183.
Roy MF, Riendeau N, Bédard C, Hélie P, Min-Oo G, Turcotte K et al. Pyruvate kinase deficiency confers susceptibility to Salmonella typhimurium infection in mice. J Exp Med 2007; 204: 2949–2961.
Yuki KE, Eva MM, Richer E, Chung D, Paquet M, Cellier M et al. Suppression of hepcidin expression and iron overload mediate Salmonella susceptibility in Ankyrin 1 ENU-induced mutant. PLoS ONE 2013; 8: e55331.
Roy MF, Riendeau N, Loredo-Osti JC, Malo D . Complexity in the host response to Salmonella typhimurium infection in AcB and BcA recombinant congenic strains. Genes Immun 2006; 7: 655–666.
Hageman RS, Leduc MS, Caputo CR, Tsaih S-W, Churchill GA, Korstanje R . Uncovering genes and regulatory pathways related to urinary albumin excretion. J Am Soc Nephrol 2011; 22: 73–81.
Peatman E, Terhune J, Baoprasertkul P, Xu P, Nandi S, Wang S et al. Microarray analysis of gene expression in the blue catfish liver reveals early activation of the MHC class I pathway after infection with Edwardsiella ictaluri. Mol Immunol 2008; 45: 553–566.
Storey JD, Xiao W, Leek JT, Tompkins RG, Davis RW . Significance analysis of time course microarray experiments. Proc Natl Acad Sci USA 2005; 102: 12837–12842.
Banchereau J, Bazan F, Blanchard D, Briè F, Galizzi JP, van Kooten C et al. The CD40 antigen and its ligand. Annu Rev Immunol 1994; 12: 881–926.
Kawabe T, Naka T, Yoshida K, Tanaka T, Fujiwara H, Suematsu S et al. The immune responses in CD40-deficient mice: impaired immunoglobulin class switching and germinal center formation. Immunity 1994; 1: 167–178.
Thienel U, Loike J, Yellin MJ . CD154 (CD40L) induces human endothelial cell chemokine production and migration of leukocyte subsets. Cell Immunol 1999; 198: 87–95.
Song Z, Jin R, Yu S, Rivet JJ, Smyth SS, Nanda A et al. CD40 is essential in the upregulation of TRAF proteins and NF-KappaB-dependent proinflammatory gene expression after arterial injury. PLoS ONE 2011; 6: e23239.
Mukundan L, Bishop GA, Head KZ, Zhang L, Wahl LM, Suttles J . TNF receptor-associated factor 6 is an essential mediator of CD40-activated proinflammatory pathways in monocytes and macrophages. J Immunol 2005; 174: 1081–1090.
Fernandez-Cabezudo MJ, Ullah A, Flavell RA, Al-Ramadi BK . Evidence for the requirement for CD40—CD154 interactions in resistance to infections with attenuated Salmonella. In: J Endotoxin Res 2005; 11 p 395.
Ferrari S, Giliani S, Insalaco A, Al-Ghonaium A, Soresina AR, Loubser M et al. Mutations of CD40 gene cause an autosomal recessive form of immunodeficiency with hyper IgM. Proc Natl Acad Sci 2001; 98: 12614–12619.
Kawaguchi H, Kobayashi M, Nakamura K, Konishi N, Miyagawa S-i, Sato T et al. Dysregulation of transcriptions in primary granule constituents during myeloid proliferation and differentiation in patients with severe congenital neutropenia. J Leukoc Biol 2003; 73: 225–234.
Jackson L, Cady CT, Cambier JC . TLR4-mediated signaling induces MMP9-dependent cleavage of B cell surface CD23. J Immunol 2009; 183: 2585–2592.
Bjerregaard MD, Jurlander J, Klausen P, Borregaard N, Cowland JB . The in vivo profile of transcription factors during neutrophil differentiation in human bone marrow. Blood 2003; 101: 4322–4332.
Garwicz D, Lennartsson A, Jacobsen SE, Gullberg U, Lindmark A . Biosynthetic profiles of neutrophil serine proteases in a human bone marrow-derived cellular myeloid differentiation model. Haematologica 2005; 90: 38–44.
Wu H, Zhang G, Minton JE, Ross CR, Blecha F . Regulation of cathelicidin gene expression: induction by lipopolysaccharide, interleukin-6, retinoic acid, and Salmonella enterica Serovar Typhimurium infection. Infect Immun 2000; 68: 5552–5558.
Mavroudi I, Papadaki V, Pyrovolaki K, Katonis P, Eliopoulos AG, Papadaki HA . The CD40/CD40 ligand interactions exert pleiotropic effects on bone marrow granulopoiesis. J Leukoc Biol 2011; 89: 771–783.
Pullen SS, Miller HG, Everdeen DS, Dang TTA, Crute JJ, Kehry MR . CD40−tumor necrosis factor receptor-associated factor (TRAF) interactions: regulation of CD40 signaling through multiple TRAF binding sites and TRAF hetero-oligomerization. Biochemistry 1998; 37: 11836–11845.
Peters AL, Plenge RM, Graham RR, Altshuler DM, Moser KL, Gaffney PM et al. A novel polymorphism of the human CD40 receptor with enhanced function. Blood 2008; 112: 1863–1871.
Fortin A, Diez E, Rochefort D, Laroche L, Malo D, Rouleau GA et al. Recombinant congenic strains derived from A/J and C57BL/6J: a tool for genetic dissection of complex traits. Genomics 2001; 74: 21–35.
Ullman-Culleré MH, Foltz CJ . Body condition scoring: a rapid and accurate method for assessing health status in mice. Lab Anim Sci 1999; 49: 319–323.
Verdugo RA, Deschepper CF, Muñoz G, Pomp D, Churchill GA . Importance of randomization in microarray experimental designs with Illumina platforms. Nucleic Acids Res 2009; 37: 5610–5618.
R Core Team R: A Language and Environment for Statistical Computing. In: 3.00 edn R Foundation for Statistical Computing: Vienna, Austria, 2013.
Baldi P, Long AD . A Bayesian framework for the analysis of microarray expression data: regularized t -test and statistical inferences of gene changes. Bioinformatics 2001; 17: 509–519.
Murie C, Woody O, Lee AY, Nadon R . Comparison of small n statistical tests of differential expression applied to microarrays. BMC Bioinformatics 2009; 10: 45.
Baldi P, Hatfield GW . DNA Microarrays and Gene Expression: From Experiments to Data Analysis and Modeling. Cambridge University Press: New York, NY, USA, 2002.
Benjamini Y, Hochberg Y . Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Series B Stat Methodol 1995; 57: 289–300.
Rocke DM . Design and analysis of experiments with high throughput biological assay data. Semin Cell Dev Biol 2004; 15: 703–713.
Allison DB, Cui X, Page GP, Sabripour M . Microarray data analysis: from disarray to consolidation and consensus. Nat Rev Genet 2006; 7: 55–65.
Edgar R, Domrachev M, Lash AE . Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 2002; 30: 207–210.
Rose S, Misharin A, Perlman H . A novel Ly6C/Ly6G-based strategy to analyze the mouse splenic myeloid compartment. Cytometry A 2012; 81A: 343–350.
Schneider CA, Rasband WS, Eliceiri KW . NIH Image to ImageJ: 25 years of image analysis. Nat Methods 2012; 9: 671–675.
Acknowledgements
We are very grateful for the technical support of Nadia Prud’homme, Catherine Paré, Stuart Foster, Leïla Rached-D’Astous and for the assistance with data analysis provided by Robert Nadon. This work was supported by Canadian Institutes of Health Research (CIHR) Grants to DM (MOP-15461) and to SMV (MOP-89821).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Genes and Immunity website
Supplementary information
Rights and permissions
About this article
Cite this article
Beatty, S., Yuki, K., Eva, M. et al. Survival analysis and microarray profiling identify Cd40 as a candidate for the Salmonella susceptibility locus, Ity5. Genes Immun 17, 19–29 (2016). https://doi.org/10.1038/gene.2015.41
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2015.41
This article is cited by
-
Complex genetics architecture contributes to Salmonella resistance in AcB60 mice
Mammalian Genome (2017)