Abstract
Background
Surgically induced scleral necrosis (SINS) is a severe form of scleritis threatening both vision and integrity of the eye. SINS is a rare sequel of ocular surgery and has been described after cataract extraction, trabeculectomy, strabismus, retinal detachment surgery including parsplana vitrectomy, penetrating keratoplasty, pterygium excision, and diode cyclophotocoagulation.
Materials and methods
To report on the application of autologous fascia lata as a readily available natural biomaterial for ocular tectonic support in SINS, we performed this retrospective chart review including two eyes of two patients; one case following both repaired rupture globe, parsplana vitrectomy, and diode laser transscleral cyclophotocoagulation and one case following pterygium surgical excision.
Results
Successful coverage of the area of scleral thinning with autologous fascia lata was achieved in both cases with overlying healthy vascularized conjunctiva and resolution of the ocular inflammation.
Conclusion
The fascia lata transplant combined with systemic immunosuppression was successful in providing adequate tectonic support and controlling the progression of scleral melt for two cases with SINS.
Similar content being viewed by others
Introduction
Surgically induced scleral necrosis (SINS) is a severe form of scleritis threatening both vision and integrity of the eye.1, 2, 3, 4, 5 SINS is a rare sequel of ocular surgery and has been described after cataract extraction, trabeculectomy, strabismus, retinal detachment surgery including parsplana vitrectomy, penetrating keratoplasty, pterygium excision, and diode cyclophotocoagulation.6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16
Materials and methods
The study was conducted in accordance with the hospital ethical committee and IRB at Cairo University hospitals. This is a retrospective chart review including two eyes of two patients; one case following repaired rupture globe, parsplana vitrectomy, and diode laser transscleral cyclophotocoagulation and one case following pterygium surgery. The indication for surgical intervention was postsurgical progressive scleral melting with underlying uveal exposure with progressive ectasia.
Results
Case report 1
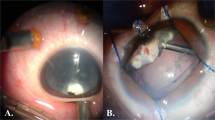
A 37-year-old female patient with a 6-month history of repaired rupture globe following blunt trauma followed by traumatic non resolving vitreous hemorrhage, which required a parsplana vitrectomy 2 months following her primary repair. She developed angle recession glaucoma with gradual scleral thinning and ectasia of the underling uvea, causing a progressive staphyloma at the site of prior scleral repair and progressive scleral melting in the region of the sclerotomies performed during the vitrectomy procedure (Figure 1a and b). Slit-lamp examination revealed areas of localized scleral melt corresponding to the site of transscleral cyclophotocoagulation laser application that soon became confluent areas of scleral melt (Figure 1c). An Ahmed valve in the lower temporal quadrant and 360° (four quadrants) fascia lata placement aiming to cover the entire scleral melt was performed (Figure 1d–f) with successful coverage of areas of melt being achieved with only limited retraction of the fascia lata covering the temporal sclerotomy. Systemic immunosuppression was used preoperatively with oral prednisone 2 mg/kg for 3 weeks with gradual taper and azathioprine 2.5 mg/kg, which was substituted postoperatively with oral prednisone 15 mg/day and oral cyclophosphamide 100 mg/day for 6 months.
(a) Preoperative color photo demonstrating scleral thinning and ectasia of the underling uvea causing a progressive staphyloma at the site of prior scleral repair (blue arrow). (b) Preoperative color photo demonstrating scleral melting in the region of the sclerotomies performed during the vitrectomy procedure (red arrow). (c) Preoperative color photo showing confluent areas of scleral melt corresponding to the site of transscleral cyclophotocoagulation (yellow arrow). (d) Post operative, following fascia lata placement with coverage of areas of melt in the area of prior repaired rupture (green arrow). (e) Post operative, following fascia lata placement covering area of melt in the region of the sclerotomy with only limited retraction of the fascia lata and minimal exposure of the underlying melt. (f) Post operative, following fascia lata placement covering the area of prior diode laser induced scleral melt.
Case report 2
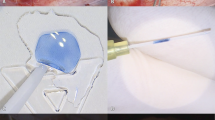
A 55-year-old male who presented with ocular pain 2 weeks following uneventful primary pterygium excision using bare sclera technique without the use of mitomycin C (MMC). Slit-lamp examination 2 weeks post-pterygium excision showed the sclera bed was markedly thinned, avascular, and ectatic with blue coloration of the underlying uvea and hyperemia of the surrounding conjunctiva (Figure 2a). The necrotic tissue was excised and autologous fascia lata (8 mm × 4 mm) was used to cover the area of scleral melt (Figure 2b and c). The sutured fascia was covered with a rotational conjunctival flap (Figure 2d). One week later, the fascia started to retract near the limbus revealing the underlying progression of the scleral melt (Figure 2e). Oral prednisone 80 mg/day with gradual taper to 15 mg/day and Azathioprine 150 mg/day were prescribed for 9 months postoperatively. The fascia lata was successfully incorporated into the underlying scleral bed with adequate vascularization of the overlying conjunctiva (Figure 2f). The excised tissue was histologically examined for evidence of vasculitis (Figure 3a and b).
(a) Photo demonstrating sclera bed markedly thinned, avascular, and ectatic with dark coloration of the underlying uvea in the region of prior pterygium excision. (b) Photo demonstrating the intraoperative harvested fascia lata (8 × 4) mm in size. (c) Photo demonstrating the intraoperative sutured fascia lata with interrupted 7/0 vicryl sutures. (d) Photo demonstrating the rotational conjunctival flap used to cover the sutured fascia lata. (e) Photo demonstrating the fascia starting to retract one week later near the limbus revealing the underlying progression of the scleral melt, which required the addition of immunesuppression. (f) The fascia lata was successfully incorporated into the underlying scleral bed with adequate vascularization of the overlying conjunctiva. The full color version of this figure is available at the Eye Journal online.
Discussion
Necrotizing anterior scleritis is the most severe form of scleritis and is a serious threat to vision and the integrity of the eye.17, 18 Various tissues have been used for patch grafting, including processed pericardium19, 20 sclera,21, 22 and fascia lata.23 In our small series, treatment was initiated as early as possible with immunosuppressive treatment with high-dose corticosteroids and cytotoxic drugs as necessary, together with surgical replacement of damaged tissue.
Our first case is very unique in ophthalmic literature, as it combines three different predisposing surgical interventions with a chronic progressive course following the initial repaired trauma with exacerbation in multiple scleral quadrants with each proceeding surgical intervention; in this case, the fascia was used to cover the site of prior surgical repair, the sites of entry during parsplana vitrectomy, and sites of prior diode laser application, as well as the tube of the Ahmed valve; thus, the four scleral quadrants were covered in this case. Our second case demonstrates surgically induced necrotizing scleritis following excessive dissection and cautery during pterygium removal, causing ischemia of the scleral bed.
Concomitant use of systemic immunosuppressive drugs increases the chance of survival of the patch graft in patients with active scleritis, as melting of the patch grafts in eyes with necrotizing scleritis is not unusual and has been reported with different materials.21 Both cases received systemic immunosuppression as in these eyes, the inflammatory milieu arising from the vasculitis may delay epithelialization of the graft; with the conjunctiva being severely affected by the vasculitic process, which may not provide sufficient vascular supply to the graft.15, 23
We primarily preferred to use autogenous fascia lata as it is relatively acellular, durable, and readily available with no extra expenses incurred with the use of other alternative banked materials, which is advantageous in developing countries, there is no tissue reaction or rejection and it withstands tension being very strong and lasts longer when compared with preserved banked fascia lata with an increased number of late failures with banked fascia and no potential of disease transmission as seen with banked tissue.
Conclusion
The fascia lata transplant combined with systemic immunosuppression was successful in providing adequate tectonic support for cases with SINS.

References
Sevel D . Necrogranulomatous scleritis, a clinical, pathological, and experimental study. MD Thesis. University of London, London, UK, 1966.
Watson PG, Hazleman BL . The sclera and systemic disorders, Chapter 6. Saunders: London, 1976; 171–205.
Arentsen JJ, Christiansen JM, Maumenee AE . Marginal ulceration after intracapsular cataract extraction. Am J Ophthalmol 1976; 81: 194–197.
Grindle CF, Marshall J, McLeod D, Clarke E, Fison LG . Complications of explants used in retinal detachment surgery. Mod Probl Ophthalmol 1979; 20: 219–220.
Lyne AJ, Lloyd-Jones D . Necrotising scleritis after ocular surgery. Trans Ophthalmol Soc UK 1979; 99: 146–149.
Bloomfield SE, Becker CG, Christian CL, Nauheim JS . Bilateral necrotising scleritis with marginal corneal ulceration after cataract surgery in a patient with vasculitis. Br J Ophthalmol 1980; 64: 170–174.
Salamon SM, Mondino BJ, Zaidman GW . Peripheral corneal ulcers, conjunctival ulcers, and scleritis after cataract surgery. Am J Ophthalmol 1982; 93: 334–337.
Alsagoff Z, Tan DT, Chee SP . Necrotising scleritis after bare sclera excision of pterygium. Br J Ophthalmol 2000; 84: 1050–1052.
Vagefi MR, Hollander DA, Seitzman GD, Margolis TP . Bilateral surgically induced necrotising scleritis with secondary superinfection. Br J Ophthalmol 2005; 89: 124–125.
Kaufman LM, Folk ER, Miller MT, Tessler HH . Necrotising scleritis following strabismus surgery for thyroid ophthalmopathy. J Pediatr Ophthalmol Strabismus 1989; 26: 236–238.
Shen SY, Lai JS, Lam DS . Necrotizing scleritis following diode laser transscleral cyclophotocoagulation. Ophthalmic Surg Lasers Imaging 2004; 35 (3): 251–253.
O’Donoghue E, Lightman S, Tuft S, Watson P . Surgically induced necrotizing sclerokeratitis-precipitating factors and response to treatment. Br J Ophthalmol 1992; 76: 17–21.
Bloomfield SE, Becker CG, Christian CL, Nauheim JS . Bilateral necrotizing scleritis with marginal ulceration after cataract surgery in a patient with systemic vascularisation. Br J Ophthalmol 1980; 64: 170–174.
Morley AM, Pavesio C . Surgically induced necrotising scleritis following three-port pars plana vitrectomy without scleral buckling: a series of three cases. Eye 2008; 22 (1): 162–164.
Scott JA, Clearkin LG . Surgically induced diffuse scleritis following cataract surgery. Eye 1994; 8: 292–297.
Okhravi N, Odufuwa B, McCluskey P, Lightman S . Scleritis. Surv Ophthalmol 2005; 50: 351–363.
Sainz de la Maza M, Jabbur NS, Foster CS . Severity of scleritis and episcleritis. Ophthalmology 1994; 101: 389–396.
Tuft SJ, Watson PG . Progression of scleral disease. Ophthalmology 1991; 98: 467–471.
Lazzaro DR . Repair of necrotizing scleritis in ulcerative colitis with processed pericardium and a Prokera amniotic membrane graft. Eye Contact Lens 2010; 36 (1): 60–61.
Schein OD . The use of processed pericardial tissue in anterior ocular segment reconstruction. Am J Ophthalmol 1998; 125: 549–552.
Lin CP, Tsai MC, Wu YH, Shih MH . Repair of a giant scleral ulcer with preserved sclera and tissue adhesive. Ophthalmic Surg Lasers 1996; 27: 995–999.
Rodriguez-Ares MT, Tourino R, Capeans C, Sánchez-Salorio M . Repair of scleral perforation with preserved sclera and amniotic membrane in Marfan’s syndrome. Ophthalmic Surg Lasers 1999; 30: 485–487.
Kachmaryk M, Bouchard CS, Duffner LA . Bilateral fascia lata patch grafts in a patient with progressive scleromalacia perforans. Ophthalmic Surg Lasers 1996; 27: 397–400.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no conflict of interest.
Additional information
This work was partly presented at the ASCRS Boston 2010
Rights and permissions
About this article
Cite this article
Kobtan, H. Use of autologous fascia lata as a natural biomaterial for tectonic support in surgically induced necrotizing scleritis. Eye 29, 580–584 (2015). https://doi.org/10.1038/eye.2014.327
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2014.327
This article is cited by
-
Giant anterior scleral staphyloma caused by blunt ocular trauma: a case report
BMC Ophthalmology (2023)
-
Management of Surgically-Induced Necrotizing Scleritis: Case Series and Review of the Literature
Current Ophthalmology Reports (2023)
-
The use of donor scleral patch in ophthalmic surgery
Cell and Tissue Banking (2017)