Abstract
The tumour-suppressor protein BRCA1 mediates its biological functions by interacting with cellular factors1,2 such as the CtIP polypeptide3,4, a substrate for the ATM (for 'ataxia telangiectasia mutated') protein kinase5. Li et al.6 report that the BRCA1–CtIP interaction is disrupted by ionizing radiation and by other genotoxic stresses that induce phosphorylation of CtIP by ATM kinase, and that this dissociation of the BRCA1–CtIP complex in turn modulates the transcription of DNA-damage-response genes6. We have shown that the BRCA1-binding domain of CtIP (amino-acid residues 133–369) is distal to the sites that are phosphorylated by ATM kinase (residues S664 and S745)7. We now show that the BRCA1–CtIP complex is stable in irradiated cells, and that the phosphorylated isoforms of CtIP that are induced by ionizing radiation still interact in vivo with BRCA1. We conclude that disruption of the BRCA1–CtIP complex cannot account for induction of DNA-damage-response genes in the way proposed by Li et al.6.
Similar content being viewed by others
Main
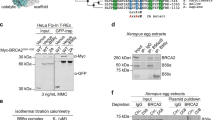
To investigate the effect of genotoxic stress on CtIP, we treated human T24 carcinoma cells with ionizing radiation or ultraviolet light, and immunoblotted the cell lysates with a CtIP-specific monoclonal antibody7. As expected, ionizing radiation induced the formation of the phosphorylated CtIP isoforms6 (Fig. 1a, top, lane 3). These species migrate more slowly than CtIP polypeptides from untreated cells (lane 1) and are converted to a faster-migrating form after incubation with λ-phosphatase (lane 4). As expected, BRCA1 was also hyperphosphorylated in cells exposed to ionizing radiation (Fig. 1a, bottom)8,9.
a, Induction of CtIP phosphorylation. Lysates from T24 carcinoma cells (lanes 1, 2, untreated; lanes 3, 4, treated with 40 Gy ionizing radiation (IR) were immunoblotted with CtIP-specific (top) or BRCA1-specific (bottom) monoclonal antibodies7; the indicated lysates were pretreated with λ-phosphatase (PPase). b, Co-immunoprecipitation of CtIP with BRCA1. T24 cells (top) and GM000637H fibroblasts (bottom) were irradiated with 40 Gy IR or 10 J m −2 ultraviolet (UV) light, and lysates were immunoblotted for CtIP (lanes 1–3). Alternatively, lysates were immunoprecipitated with BRCA1-specific antiserum7 (I) or pre-immune serum (Pr) and then immunoblotted for CtIP (lanes 4–9). c, Co-immunoprecipitation of BRCA1 with CtIP. Lysates of HBL100 epithelial cells were either immunoblotted for BRCA1 (lanes 1, 2) or immunoprecipitated with CtIP-specific 210 antiserum7 and then immunoblotted for BRCA1 (lanes 3–6).
To determine the effect of ionizing radiation on the BRCA1–CtIP complex, we immunoprecipitated T24-cell lysates with a BRCA1-specific polyclonal antiserum, and monitored each precipitate for CtIP by immunoblotting with a CtIP-specific monoclonal antibody. As expected, CtIP was detected in BRCA1 immunoprecipitates from lysates of untreated and ultraviolet-irradiated cells (Fig. 1b, top , lanes 5, 9), which is consistent with our previous findings7 but not with those of Li et al.10. Moreover, we recovered all CtIP species, including the hyperphosphorylated forms, in BRCA1 immunoprecipitates prepared from cells exposed to ionizing radiation (Fig. 1b, lane 7). The same results were obtained in all cell lines tested, including the two lines examined by Li et al.6: T24 carcinoma cells (Fig. 1b, top) and SV40-transformed GM000637H fibroblasts (Fig. 1b, bottom).
The stability of the CtIP–BRCA1 complex was also evident in reciprocal co-immunoprecipitation experiments, in which comparable amounts of BRCA1 polypeptides were present in CtIP immunoprecipitates from both untreated cells and cells exposed to ionizing radiation (Fig. 1c, lanes 4, 6). Our results indicate that, contrary to the findings of Li et al., the CtIP–BRCA1 complex is stable to genotoxic stress such as ultraviolet or ionizing radiation.
References
Scully, R. & Livingston, D. M. Nature 408, 429–432 (2000).
Deng, C.-X. & Brodie, S. G. BioEssays 22, 728–737 (2000).
Yu, X., Wu, L. C., Bowcock, A. M., Aronheim, A. & Baer, R. J. Biol. Chem. 273, 25388–25392 (1998).
Wong, A. K. et al. Oncogene 17, 2279–2285 (1998).
Kim, S. T., Lim, D. S., Canman, C. E. & Kastan, M. B. J. Biol. Chem. 274, 37538–37543 (1999).
Li, S. et al. Nature 406, 210–215 (2000).
Yu, X. & Baer, R. J. Biol. Chem. 275, 18541–18549 (2000).
Scully, R. et al. Cell 90, 425–435 (1997).
Thomas, J. E., Smith, M., Tonkinson, J. L., Rubinfeld, B. & Polakis, P. Cell Growth Differ. 8, 801–809 (1997).
Li, S. et al. J. Biol. Chem. 274, 11334–11338 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu-Baer, F., Baer, R. Effect of DNA damage on a BRCA1 complex. Nature 414, 36 (2001). https://doi.org/10.1038/35102118
Issue Date:
DOI: https://doi.org/10.1038/35102118
This article is cited by
-
Epigenetic inactivation of DNA repair genes as promising prognostic and predictive biomarkers in urothelial bladder carcinoma patients
Molecular Genetics and Genomics (2022)
-
C-terminal-binding protein interacting protein binds directly to adenovirus early region 1A through its N-terminal region and conserved region 3
Oncogene (2007)
-
The role of BRCA1 in transcriptional regulation and cell cycle control
Oncogene (2006)
-
C-terminal binding proteins: Emerging roles in cell survival and tumorigenesis
Apoptosis (2006)
-
SIAH-1 interacts with CtIP and promotes its degradation by the proteasome pathway
Oncogene (2003)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.