Abstract
Design:
Cross-sectional.
Objectives:
(1) To determine the effects of the level of spinal cord injury (SCI) on skeletal muscle, intramuscular fat (IMF) cross-sectional areas (CSAs) and relative IMF; (2) to determine the relation, if any, of spasticity to each of these variables after incomplete SCI.
Settings:
In-patient study at the Shepherd Center, Atlanta, GA, USA.
Methods:
Thirteen individuals with incomplete SCI were classified according to their level of injury into a high level of injury group (HLI, C5–C7, n=8) and a low level of injury group (LLI, T12–L2, n=5). Spasticity was determined for thigh muscles using a modified Ashworth scale at 6 weeks post-injury. T1-weighted magnetic resonance (MR) images were taken 6 weeks post-injury to measure thigh skeletal muscle and IMF CSAs.
Results:
Spasticity was significantly evident in the HLI group compared to the LLI group (P=0.023). Six weeks post-injury, muscle CSA was 103±18 cm2 in the HLI group and 80±20 cm2 in the LLI group (P=0.042). Relative IMF was 3.6±2.0% in HLI and 7.5±4.0% in LLI (P=0.021). Additionally, spasticity accounted for 54% of the variability in muscle CSA for all subjects (r2=0.54, P=0.006).
Conclusions:
Spasticity may be an important factor in defending skeletal muscle size and indirectly preventing IMF accumulation early after incomplete SCI.
Similar content being viewed by others
Introduction
Skeletal muscle atrophy is a common adaptation following spinal cord injury (SCI) resulting from the loss of central activation and subsequent unloading.1, 2, 3, 4, 5 The process of muscle atrophy proceeds as early as 6 weeks post-injury after both complete and incomplete SCI.2, 3 For example, skeletal muscle cross-sectional area (CSA) has been reported to be as much as 45% smaller than that of able-bodied (AB) controls just 6 weeks after a clinically complete SCI.2 We have recently reported that the magnitude of muscle atrophy exceeds 30% 6 weeks after incomplete SCI.3 Previous reports demonstrated that the process of skeletal muscle atrophy has adverse consequences on cardiovascular, metabolic and musculoskeletal profiles in individuals with SCI.6, 7
Skeletal muscle atrophy has been shown to be related to the accumulation of intramuscular fat (IMF) in hemiparetic individuals and those with complete and incomplete SCI.3, 4, 8 IMF can be defined as the infiltrated fat within individual muscle and between muscle groups. Six weeks post-injury, individuals with incomplete SCI were shown to have a greater IMF accumulation compared to their matched AB controls, even after controlling for the difference in muscle size between the two groups.3 Goodpaster et al.9 also showed that IMF is negatively correlated with insulin sensitivity in obese and non-obese subjects with and without type II diabetes. Furthermore, it has been shown that IMF accounts for a 70% reduction in glucose tolerance in individuals with complete SCI.4 Additionally, increasing IMF content negatively affects performance as it influences the force-generating capacity of the lower extremities.10, 11
Individuals with incomplete SCI represent over half of the SCI population.12, 13, 14 However, there are few reports describing their muscle adaptations compared to individuals with complete SCI. Although skeletal muscle atrophy can adversely affect individuals with both complete and incomplete SCI,2, 3 the degree of atrophy may lead to more serious implications among those with incomplete injury because it can limit their functional performance and their ability to attain independent ambulation. Additionally, muscle atrophy is associated with poor performance and decreasing strength that is strongly correlated to gait kinematics and increasing incidence of fracture.15, 16
The factors that control the extent of skeletal muscle atrophy early after injury and the variability from one individual to another are poorly understood. If the magnitude of atrophy after 6 weeks exceeds what occurs after 3 months post-injury,3 then defending against muscle atrophy in early stage is easier than treatment in late stage. Additionally, understanding these factors could contribute to proper medical interventions and early rehabilitation programs that attenuate muscle atrophy. One of these factors could be the level of injury. Previously, the level of injury has been shown to influence the degree of ambulation and cardiovascular response after SCI.17, 18 Furthermore, the level of injury determines the degree of spasticity after incomplete SCI. For example, the prevalence of spasticity has been shown to occur more commonly with incomplete cervical SCI compared to a more distal injury.19, 20 Spasticity has been shown to have beneficial effects such as improved ambulation and peripheral circulation.20, 21 Moreover, spasticity in the tail muscle of the rats preserves the slow properties of skeletal muscle and attenuates the slow-to-fast transformation in myosin expression commonly occurring after SCI,22, 23 whereas in humans, the effect of spasticity on muscle size and composition is yet to be determined. Therefore, the purpose of this study was to determine the effect of spasticity, measured by a modified Ashworth scale (MAS), on thigh skeletal muscle and IMF CSAs after 6 weeks of incomplete SCI in high (C5–C7) versus low (T12–L3) levels of injury. We also investigated whether any relationship exists between each of these variables and spasticity. We hypothesized that spasticity will attenuate skeletal muscle atrophy and prevent IMF accumulation in individuals with high versus low injury.
Methods
Participants
Thirteen, non-recreational and non-exercise trained, individuals with incomplete SCI participated in this study. Participants were classified within a few days of admission to the hospital by the American Spinal Injury Association (ASIA) Neurological Classification system as B or C. They were recruited from the Shepherd Center (Atlanta, GA, USA) approximately 6 weeks after injury when they were medically stable. T1-weighted magnetic resonance (MR) images were taken of both thighs 6 weeks post-injury, accompanied by an MAS evaluation. The study was approved by the Institutional Review Boards of the University of Georgia and the Shepherd Center before the subjects gave consent.
Participants were classified according to their level of injury into two groups: the high level of injury (HLI) group was C5–C7 (n=8, 26±14 years, 82±14 kg and 181±6 cm) and the low level of injury (LLI) group was T12–L2 (n=5, 36±14 years, 77±14 kg and 180±13 cm). Their physical and clinical characteristics are presented in Table 1. All participants were ‘incomplete SCI’ with sacral sparing as confirmed by ASIA and they were wheelchair-dependent.
Magnetic resonance imaging
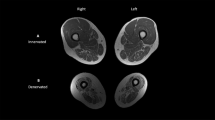
Skeletal muscle and IMF CSAs were determined using MR imaging as described previously.1, 2, 3, 4 Images of both thighs were acquired using a 1.5-T magnet and a whole body coil. T1-weighted transaxial images, 1 cm thick and 0.5 cm apart, were taken from the hip to the knee joints (TR/TE 500/14, 40 cm field of view, 1 repetition and 256 × 256 matrix). Eight slices were analyzed starting with the first slice distal to the gluteus maximus and the rest towards the knee joint.2, 3, 4 MR images were downloaded to a disk and analyzed with modified X-Vessel software. The images were segmented into fat, skeletal muscle and background/bone based on their signal intensity. This technique has been described previously in more detail.24, 25, 26 Skeletal muscle CSAs of the rectus femoris, vasti, hamstring and adductors muscles were determined (Figure 1 and Table 2). Manual selection of a pixel of skeletal muscle highlighted all skeletal muscle pixels and provided the total number of skeletal muscle pixels exclusive of fat or low-intensity pixels. The individual collecting the data performed two trials to determine the test–retest reliability (r=0.99).
Representative magnetic resonance (MR) images of the mid-thigh of (a) high level of injury (HLI) and (b) low level of injury (LLI) individuals with incomplete spinal cord injury (SCI) 6 weeks post-injury. Note the white material (intramuscular fat (IMF)) starts to infiltrate in the thigh skeletal muscle groups of LLI individuals. RF, VL, VI, VM, Hams and Add refer to rectus femoris, vastus lateralis, vastus intermedius, vastus medialis, hamstrings and adductors muscles, respectively.
Skeletal muscle and intramuscular fat calculations
The procedure for calculating skeletal muscle and IMF CSAs has been previously described in detail.3, 4 Briefly, the outer perimeter of the thigh muscle groups was traced and the femur bone was excluded. The region of interest was then highlighted and the signal intensity within this region was automatically determined (see above). After the pixel signal intensity was determined, a bimodal histogram of two peaks was plotted. The first peak represents the muscle peak while the second represents the fat peak. A midpoint between these two peaks separates muscle from fat pixels. The CSAs were calculated using the following equations: Muscle CSA (cm2)=total number of pixels of muscle*(FOV/matrix size)2 and IMF CSA (cm2)=total number of pixels of IMF*(FOV/matrix size)2. To control for the difference in skeletal muscle size between the two groups, IMF CSA was expressed relative to skeletal muscle CSA. Relative IMF=((IMF CSA/muscle CSA)*100).
Although skeletal muscle CSA for both thighs was measured, the involved thigh was only chosen for statistical analysis and it was defined as the thigh with apparent muscle atrophy as classified by MRI after 6 weeks post-injury. We have previously shown that there was no difference in muscle size between the involved and the less-involved side either 6 weeks or 4.5 months post-injury.3 Therefore, we chose to present the data of the involved thigh in both groups.
Modified Ashworth scale
Muscle spasticity was evaluated with the MAS and was conducted for the same muscles that were imaged for CSA and IMF. An MAS was administered by a physical therapist while the patient was lying in the supine position; the therapist was completely blinded to the study design. The room temperature was held constant between 21 and 24°C. The lower leg was moved passively from extension to full flexion at the knee joint using the same technique previously applied to examine spasticity in knee extensors and flexors.19, 20 A single score of the involved side was reported for each participant. Few of the participants in the HLI group were on anti-spastic medications (doses are not documented), but it was not possible to stop the medications owing to medical restrictions. The MAS constitutes six grades, each of which represents the degree and the location of the resistance to passive movement throughout the range of motion (ROM): 0, no increase in skeletal muscle tone; 1, slight increase in skeletal muscle tone manifested by a catch and release at the end of the ROM; 2, slight increase in skeletal muscle tone, manifested by a catch, followed by minimal resistance throughout the ROM; 3, more marked increase in skeletal muscle tone throughout the ROM; 4, considerable increase in skeletal muscle tone; and 5, rigid in flexion or extension. Testing was performed in the morning for all participants within the same time range to avoid the diurnal fluctuation in spasticity.20
Functional independence measure (FIM) was used as an index to determine the level of independence of each patient using the seven-point scale, where 1 indicates total assistance and 7 complete independence.27 The motor component score for each participant is presented in Table 1. FIM was indirectly used to infer the extent of the activity at 6 weeks post-injury.
Statistical analysis
Independent t-tests were used to compare differences in physical characteristics, skeletal muscle CSA, IMF CSA and relative IMF between the HLI and LLI groups. Mann–Whitney U-test was used to test the difference in spasticity scores between the HLI and LLI groups. A two-way analysis of variance (ANOVA; MAS × muscle CSA) was performed to determine the effect of spasticity on the mid-eight slices of the involved thigh after stratifying participants based on their MAS. A Bonferroni correction for multiple comparisons was used when evaluating differences in means. Gamma statistic (nonparametric statistic, which is equivalent to Spearman R) was used to test the relationship between the two ordinal variables (level of injury and MAS). A simple linear regression was used to test for the relationship between spasticity and skeletal muscle CSA, IMF CSA or relative IMF. Data are presented as mean±s.d. and significance level was set at α=0.05.
Results
The physical characteristics of the individuals with HLI and LLI are presented in Table 1. There was no difference in the physical characteristics between the two groups (age (P=0.23), height (P=0.71) and weight (P=0.52)). The duration of injury for both groups was 6±1 weeks, range=3–8 weeks post-injury.
Skeletal muscle CSA
Six weeks post-injury, skeletal muscle CSA of the involved thigh was 22.5% larger in HLI compared to LLI (103±18 versus 80±20 cm2; P=0.042). The participants with high MAS had evidently greater muscle size along their mid-eight slices than those with low MAS (F (3, 64)=14.97; P<0.0001). Post hoc comparisons using Bonferroni demonstrated significant differences in muscle CSA across slices between those with different MAS scores (4 versus 0; P<0.0001), (4 versus 1; P=0.003) and (3 versus 0; P<0.0001). Five HLI individuals with MAS scores 4 and 3 showed a remarkable difference in muscle size across slices compared to those with MAS 0 and 1 (Figure 2). Skeletal muscle CSAs of rectus femoris, vasti, hamstring and adductors muscles were larger in the HLI group compared to the LLI group (Figure 1 and Table 2).
Muscle cross-sectional areas (CSAs; mean±s.d.) of mid-eight slices of the 13 participants classified based on their modified Ashworth scale (MAS). Scores 0 and 1 represent the average of four individuals for each score, score 3 represents the average of three individuals and score 4 represents the average of two individuals. *, significant differences between those with MAS scores (4 versus 0; P<0.0001), (4 versus 1; P=0.003) and (3 versus 0; P<0.0001). Error bars are presented to one side for the purpose of clarity.
Intramuscular fat
IMF CSA of HLI and LLI groups did not differ (6.0±3.0 and 7.1±2.1 cm2, P=0.11). However, relative IMF (calculated as (IMF CSA/muscle CSA)*100) was 40% lower in the HLI group than in the LLI group (7.2±4.0 versus 3.2±2.0%; P=0.021).
Spasticity
Mann–Whitney U-test showed that spasticity was six times higher in the HLI versus the LLI group (P=0.023; Table 3). Gamma statistic showed that there was a strong relationship between the level of injury and spasticity as measured by MAS (γ=0.87; P=0.0001), suggesting that the degree of spasticity after SCI is highly determined by the level of injury. However, three individuals in the HLI group showed MAS scores of 0 and 1 (Table 3). Simple linear regression revealed a significant relationship between skeletal muscle CSA and spasticity scores after covariate for age between both groups (r2=0.54; P⩽0.006; Figure 3). The relationship between absolute or relative IMF and spasticity scores was not evident (r2=0.17; P⩽0.11).
Discussion
The current study used the level of injury as a model to determine the influence of spasticity on skeletal muscle size and composition. It was found that spasticity after incomplete SCI attenuates skeletal muscle atrophy and may indirectly prevent further accumulation of IMF at 6 weeks post-injury. Relative IMF was higher in those with incomplete LLI compared to HLI. Furthermore, the variance in spasticity explains more than 50% of the variance in muscle size that exists between individuals with HLI compared to LLI.
Skeletal muscle atrophy is a health-related problem that leads to costly secondary complications such as cardiovascular diseases and type II diabetes.6, 7 The greatest magnitude of muscle atrophy can be determined as early as 6 weeks post-injury in complete and incomplete SCI.2, 3 In healthy individuals, a 5% loss of muscle mass can impair organ function and a loss of 20% or more can cause organ failure and death.28 We have recently shown that skeletal muscle size after incomplete SCI could be as low as 33% compared to matched AB controls.3 The current results further extend our previous findings by showing a difference in muscle size of more than 20% between individuals with HLI and those with LLI. This degree of atrophy could have serious implications for those with incomplete injury because it can limit their functional performance and their ability to attain independent ambulation.15, 16 Furthermore, skeletal muscle atrophy is associated with an increase in IMF, which might possibly explain the increased prevalence of insulin resistance after SCI.4, 7 These changes demonstrate the importance of understanding the factors that can attenuate muscle atrophy and prevent fat accumulation. Moreover, understanding the factors that could result in variations of muscle size from one injury to another may benefit the outcome of rehabilitation by designing protocols specific to different levels of injury.
An interesting finding in the current study was the greater percentage of atrophy in the rectus femoris muscle compared to the three vasti muscles. Unilateral suspension or bed rest studies showed that the rectus femoris muscle is the least affected muscle compared to the other three vasti muscles owing to its two-joint anatomical advantage.29, 30 The current findings suggest that the three vasti muscles experience a great extent of atrophy in the HLI and the LLI groups, and therefore the percentage difference is smaller compared to the rectus femoris muscle. Another suggestion is that spasticity might play a more protective role with a two-joint muscle than with one-joint muscle.
The idea that spasticity might exert a protective role was recently investigated on the geometry of the radius bone in stroke patients. The authors suggest that tonic activity associated with spasticity might have a protective effect.31 It is difficult to speculate how short-term exposure to spasticity could defend muscle size, especially after considering the flaccid stage. It would seem reasonable to suggest that spasticity could mimic isometric contraction or low-tension resistance training. Franzoi et al.32 measured spasticity in traumatic SCI using an isokinetic dynamometer in response to passive stretching. The results showed that individuals with Ashworth 3 developed more torque output compared to those with Ashworth 1, 2 and AB controls. In the animal model, isometric contraction attenuates skeletal muscle atrophy in response to unloading after a short period of training.33 For instance, isometric contraction for 20 days attenuates hind-limb-induced muscle atrophy in rats.33 This is plausible owing to expression of endogenous growth hormone. In the spinal cord model, acute bout of isometric contraction induced insulin growth factors that play a definite role in attenuating muscle atrophy in individuals with chronic SCI.34 Recently, it has been found that ischemia coupled with low-tension resistance training acutely increases growth hormone levels.35 Therefore, a periodical tension in the muscle for a short period could attenuate skeletal muscle atrophy after SCI.
Furthermore, spasticity may indirectly prevent IMF accumulation. Recently, we found that individuals with incomplete SCI accumulate greater IMF compared to their matched AB controls.3 In the present study, the LLI group showed a twofold increase in IMF compared to the HLI group. It is known that skeletal muscle could utilize up to 70% of total blood glucose. Therefore, skeletal muscle atrophy could result in decreased utilization of glucose with conversion of glucose to fat.36 Another possible mechanism is that the glucose uptake would stay normal after SCI, thus inhibiting fatty acid oxidation and resulting in fat accumulation within the affected skeletal muscle.37, 38 IMF is a better predictor of insulin resistance in non-disabled, obese diabetics and non-diabetics compared to subcutaneous and subfascial fat.9 Additionally, IMF explains 70% of glucose intolerance after chronic SCI.4 The mechanism relating IMF and insulin resistance is not clear; however, the release of intracellular fatty acid intermediates (for example, diacylglycerol, ceramide, long-chain acyl-CoA) from IMF could impair insulin signaling.39 Although we could not find a direct relationship between spasticity and IMF, spasticity may indirectly influence the accumulation of IMF. In the tail muscles of SCI rats, spasticity attenuates the process of slow-to-fast transformation in myosin heavy-chain expression.23 There is also a suggestion that spasticity contributes to the predominance of slow fibers in individuals with SCI.40 Slow fibers are characterized by greater mitochondrial density compared to fast fibers. This could have resulted in partitioning of IMF towards oxidation in the HLI group rather than storage in the LLI group.41 Additionally, spasticity could indirectly influence IMF accumulation by periodically increasing the activity of the thigh skeletal muscle; this rationale has been supported by showing that increasing IMF is negatively correlated to the degree of activity.11
Our results agree with previous findings that individuals with HLI tend to have more spasticity compared to LLI.19, 20 The low-level SCI may be associated with damage to α-motor neuron pools; therefore, neural activation of the affected skeletal muscle fibers cannot be accomplished.42, 43 This observation is evident during electrical stimulation application to those with level of injury below T11 where muscle contraction is barely noticeable and special stimulation parameters have to be considered to attain contraction.42 Denervation of skeletal muscle causes rapid loss of both muscle mass and contractile force and further infiltration of fat into muscle.42, 43 This was confirmed in the present study by the reduction in muscle size and increased IMF in the LLI group few weeks post-injury. Therefore, it is possible that lower motor neuron damage would cause reduction in spasticity and muscle atrophy, at the same time, but without the former causing the latter. In other words, it may be the lower motor neurons that preserve muscle size and spasticity, and not the spasticity that preserves muscle size per se. However, LLI individuals with minimal insult (MAS=1) to their α-motor neuron pools still experience more muscle atrophy than in the HLI group. Further, controlled studies are warranted to determine whether spasticity or α-motor neuron pools preserve muscle size.
Although negative aspects of spasticity on ROM, mobility, pain and functional outcome are clearly documented,19 the current study demonstrates that spasticity may defend muscle size and composition early after injury. This observation is in agreement with the fact that spasticity could positively impact 40% of individuals with SCI in activities such as dressing.19 These findings emphasize the need to accurately measure and record spasticity and to exercise caution about initiating treatment with anti-spastic medications that could decrease spasticity but adversely affect muscle size. It could be also of clinical significance to those who experienced a flaccid stage longer than 6 weeks. Therefore, it is important that clinicians understand the importance of shortening the flaccid phase or provide the proper stimulation protocols to defend muscle size early after injury.
The current results should be interpreted with caution. It was not our intention to state that the individuals with HLI have better functional outcome compared to those with LLI, but rather to set a model to determine the effect of spasticity on muscle size and composition. The small sample size and the use of a subjective tool to measure spasticity are limitations to the current study and suggest the need for further studies that address the relationship between spasticity, muscle size and type II diabetes in SCI population. MAS was used in the current study because it is the most clinically acceptable, reliable and valid measure of spasticity.19, 20 MAS has been shown to be positively correlated to the myoelectrical signals determined by electromyography; increasing spasticity grades correspond to increased electrical activity.44 Additionally, spasticity score may have changed from day to day and even from hour to hour. Thus, a single score may be considered misleading and not a representative measure of daily spasticity. This limitation, however, may be addressed by evidences that suggest that variability in motor neuron firing following acute SCI is small owing to loss of brain stem neuromodulators such as serotonin and norepinepherine that account for such variability.45 Moreover, failure to document the exact dose of the anti-spastic medication administered to the HLI group might have underestimated the actual relationship between spasticity and muscle size by further inhibiting muscle tone. However, the administered dose was very minimal to influence the relationship especially if we considered the flaccid stage period.
In conclusion, individuals with low level of incomplete SCI experienced greater skeletal muscle atrophy and larger relative IMF accumulation compared to individuals with HLI, probably by having less spasticity. Spasticity may help maintain affected skeletal muscle size after SCI by periodic increases in tension. Therefore, our findings suggest that early intervention in individuals with incomplete SCI (especially in those with LLI) may be important in attempting to reduce skeletal muscle atrophy and to prevent factors that could lead to insulin resistance and type II diabetes. Additionally, there is a need for special rehabilitation protocols to shorten the flaccid stage after injury and counteract the effect of hypotonicity in LLI individuals. Treatment of spasticity using anti-spastic medications should proceed with caution because it could have adverse effects on muscle size and composition. Finally, further longitudinal studies are warranted to examine the effect of spasticity on skeletal muscle adaptation over the first year of injury and in chronic SCI.
References
Dudley GA, Castro MJ, Rogers S, Apple Jr DF . A simple means of increasing muscle size after spinal cord injury: a pilot study. Eur J Appl Physiol 1999; 80: 394–396.
Castro MJ, Apple Jr DF, Hillegass EA, Dudley GA . Influence of complete spinal cord injury on skeletal muscle cross-sectional area within the first 6 months of injury. Eur J Appl Physiol 1999; 80: 373–378.
Gorgey AS, Dudley GA . Skeletal muscle atrophy and increased intramuscular fat after incomplete spinal cord injury. Spinal Cord 2007; 45: 304–309.
Elder CP, Apple DF, Bickel CS, Meyer RA, Dudley GA . Intramuscular fat and glucose tolerance after spinal cord injury – a cross-sectional study. Spinal Cord 2004; 42: 711–716.
Gordon T, Mao J . Muscle atrophy and procedures of training after spinal cord injury. Phys Ther 1994; 74: 50–60.
Kocina P . Body composition of spinal cord injured adults. Sports Med 1997; 23: 48–60.
Bauman WA, Spungen AM . Carbohydrate and lipid metabolism in chronic spinal cord injury. J Spinal Cord Med 2001; 24: 266–277.
Ryan AS, Dobrovolony CL, Smith GV, Silver KH, Macko RF . Hemiparetic muscle atrophy and increased intramuscular fat in stroke patients. Arch Phys Med Rehabil 2002; 83: 1703–1707.
Goodpaster BH, Thaete FL, Kelley DE . Thigh adipose tissue distribution is associated with insulin resistance in obesity and in type 2 diabetes mellitus. Am J Clin Nutr 2000; 71: 885–892.
Visser M, Kritchevsky SB, Goodpaster BH, Newman AB, Nevitt M, Stamm E et al. Leg muscle mass and composition in relation to lower extremity performance in men and women aged 70 to 79: the health, aging and body composition study. J Am Geriatr Soc 2002; 50: 897–904.
Kent-Braun JA, Ng AV, Young K . Skeletal muscle contractile and non-contractile composition in younger and older women and men. J Appl Physiol 2000; 88: 662–668.
Hakery HL, White IV EA, Tibbs Jr RE, Haines DE . A clinician's view of spinal cord injury. Anat Rec 2003; 271B: 41–48.
Lapointe R, Lajoie Y, Serrese O, Barbeau H . Functional community ambulation requirements in incomplete spinal cord injured subjects. Spinal Cord 2001; 39: 327–335.
Sekon L, Fehlings M . Epidemiology, demographics and pathophysiology of acute spinal cord injury. Spine 2001; 26: S2–S12.
Hopman MT, Dueck C, Monroe M, Philips WT, Skinner JS . Limits to maximal performance in individuals with spinal cord injury. Int J Sports Med 1998; 19: 98–103.
Jayaraman A, Gregory CM, Bowden M, Stevens JE, Shah P, Behrman AL et al. Lower extremity skeletal muscle function in persons with incomplete spinal cord injury. Spinal Cord 2006; 44: 680–687.
Taylor PN, Ewins DJ, Fox B, Grundy D, Swain ID . Limb blood flow, cardiac output and quadriceps muscle bulk following spinal cord injury and the effect of training for the Odstock functional electrical stimulation standing system. Paraplegia 1991; 31: 303–310.
Bhambhani YN, Holland LJ, Eriksson P, Steadward RD . Physiological responses during wheelchair racing in quadriplegics and paraplegics. Paraplegia 1994; 32: 253–260.
Skold C, Levi R, Seiger A . Spasticity after traumatic spinal cord injury: nature, severity, and location. Arch Phys Med Rehabil 1999; 80: 1548–1557.
Skold C . Spasticity in spinal cord injury: self- and clinically rated intrinsic fluctuations and intervention-induced changes. Arch Phys Med Rehabil 2000; 81: 144–149.
Young RR, Woolsey RM (eds) Diagnosis and Management of Disorders of the Spinal Cord. WB Saunders Company: Philadelphia, 1995.
Talmadge RJ, Roy RR, Bodine-Fowler SC, Pierotti DJ, Edgerton VR . Adaptations in myosin heavy chain profile in chronically unloaded muscles. Basic Appl Myol 1995; 5: 117–137.
Harris RLW, Bobet J, Sanelli L, Bennett DJ . Tail muscles become slow but fatigable in chronic spinal rats with spasticity. J Neurophsiol 2006; 95: 1124–1133.
Gonzalex RC, Wintz P . Digital Image Processing. Addison-Wesley: Reading, MA, 1987.
Cohen MS, Dubois RM, Zeineh NM . Rapid and effective correction of RF inhomogeneity for high field magnetic resonance imaging. Hum Brain Mapp 1987; 10: 204–211.
Suckling J, Siggmundsson T, Greenwood K, Bullmore ET . A modified fuzzy clustering algorithm for operator independent brain tissue classification of dual echo MR Images. Magn Reson Imaging 1999; 17: 1065–1076.
Ottenbacher KJ, Hsu Y, Granger CV, Fiedler RC . The reliability of the functional independence measure: a quantitative review. Arch Phys Med Rehabil 1996; 77: 1226–1232.
Demling RH, DeSanti L . Involuntary weight loss and the nonhealing wound: the role of anabolic agents. Adv Wound Care 1999; 12: 1–14.
Berg HE, Larsson L, Tesch PA . Lower limb skeletal muscle function after 6 wk of bed rest. J Appl Physiol 1997; 82: 182–188.
Hather BM, Adams GR, Tesch PA, Dudley GA . Skeletal muscle responses to lower limb suspension in humans. J Appl Physiol 1992; 7: 1493–1498.
Pang MY, Ashe MC, Eng JJ . Muscle weakness, spasticity and disuse contribute to demineralization and geometric changes in the radius following chronic stroke. Osteoporosis Int, (in press).
Franzoi AC, Castro C, Cardone C . Isokinetic assessment of spasticity in subjects with traumatic spinal cord injury (ASIA A). Spinal Cord 1999; 37: 416–420.
Adams GR, Cheng DC, Haddad F, Baldwin KM . Skeletal muscle hypertrophy in response to isometric, lengthening, and shortening training bouts of equivalent duration. J Appl Physiol 2004; 96: 1613–1618.
Bickel CS, Slade JM, Haddad F, Adams GR, Dudley GA . Acute molecular responses of skeletal muscle to resistance exercise in able-bodied and spinal cord-injured subjects. J Appl Physiol 2003; 94: 2255–2262.
Pierce JR, Clark BC, Ploutz-Snyder LL, Kanaley JA . Growth hormone and muscle function responses to skeletal muscle ischemia. J Appl Physiol 2006; 101: 1588–1595.
Aksnes AK, Hjeltnes N, Wahlstrom EO, Katz A, Zierath JR, Wallberg-Henriksson H . Intact glucose transport in morphologically altered denervated skeletal muscle from quadriplegic patients. Am J Physiol 1996; 271: E593–E600.
Jeukendrup AE . Regulation of fat metabolism in skeletal muscle. Ann NY Acad Sci 2002; 967: 217–235.
Sidossis LS, Stuart CA, Shulman GL, Lopaschuk GD, Robert RW . Glucose plus insulin regulate fat oxidation by controlling the rate of fatty acid entry in the mitochondria. J Clin Invest 1996; 98: 2244–2250.
Yu C, Chen Y, Cline GW, Zhang D, Zong H, Wang Y et al. Mechanism by which fatty acids inhibit insulin activation of insulin receptor substrate-1 (IRS-1)-associated phosphatidylinositol 3-kinase activity in muscle. J Biol Chem 2002; 277: 50230–50236.
Ditor DS, Hamilton S, Tarnopolsky MA, Green HJ, Craven BC, Parise G et al. Na+, K+-ATPase concentration and fiber type distribution after spinal cord injury. Muscle Nerve 2004; 29: 38–45.
Schenk S, Cook JN, Kaufman AE, Horowitz JF . Postexercise insulin sensitivity is not impaired after an overnight lipid infusion. Am J Physiol Endocrinol Metab 2005; 288: E519–E525.
Modlin M, Forstner C, Hofer C, Mayr W, Richter W, Carraro U et al. Electrical stimulation of denervated muscles: first results of a clinical study. Artif Organs 2005; 29: 203–206.
Kern H, Hofer C, Modlin M, Forstner C, Raschka-Hogler D, Mayr W et al. Denervated muscles in humans: limitations and problems of currently used functional electrical stimulation training protocols. Artif Organs 2002; 26: 216–218.
Sköld C, Harms-Ringdahl K, Hultling C, Levi R, Seiger A . Simultaneous Ashworth measurements and electromyographic recordings in tetraplegic patients. Arch Phys Med Rehabil 1998; 79: 959–965.
Harvey PJ, Li X, Li Y, Bennett DJ . Endogenous monoamine receptor activation is essential for enabling persistent sodium currents and repetitive firing in rat spinal motoneurons. J Neurophysiol 2006; 96: 1171–1186.
Acknowledgements
We thank all the participants, the physical therapists at the Shepherd Center and Dr Scott Bickel for their cooperation. We also thank Dr Ronald Meyer for providing the software used to analyze the images for this study. This study was supported by NIH Grants HD37439-S (GAD), HD39676 (GAD) and HD 39676S2 (GAD).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gorgey, A., Dudley, G. Spasticity may defend skeletal muscle size and composition after incomplete spinal cord injury. Spinal Cord 46, 96–102 (2008). https://doi.org/10.1038/sj.sc.3102087
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3102087
Keywords
This article is cited by
-
Denervation impacts muscle quality and knee bone mineral density after spinal cord injury
Spinal Cord (2023)
-
Role of exercise on visceral adiposity after spinal cord injury: a cardiometabolic risk factor
European Journal of Applied Physiology (2021)
-
Influence of mid and low paraplegia on cardiorespiratory fitness and energy expenditure
Spinal Cord Series and Cases (2020)
-
Spasticity Management After Spinal Cord Injury
Current Physical Medicine and Rehabilitation Reports (2020)
-
Spasticity and preservation of skeletal muscle mass in people with spinal cord injury
Spinal Cord (2019)