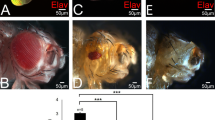
Abstract
An early event in Drosophila eye development is the division of the eye disc into dorsoventral domains. The dorsoventral pattern is displayed in the adult compound eye as a distinct mirror symmetry across the dorsoventral midline or equator1,2. The dorsoventral axis is also implicated in organizing early development of the eye, as retinal differentiation is initiated at the posterior dorsoventral midline3. Here we show that Fringe is expressed specifically in the ventral half of the undifferentiated eye disc, thus creating a dorsoventral boundary. Ectopic Fringe borders that are generated by clones of fringe − cells can reverse the planar polarity of photoreceptor clusters, indicating that the Fringe boundary is crucial for the induction of mirror symmetry. Lack of a Fringe boundary disrupts equatorial expression of Notch signalling proteins and causes a complete failure of eye development. Our results indicate that the formation of the Fringe boundary and subsequent Notch signalling at the equator are essential for organizing mirror symmetry and eye morphogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Dietrich, W. Die Facettenaugen der Dipteren. Z. Wiss. Zool. 92, 465–539 (1909).
Ready, D. F., Hanson, T. E. & Benzer, S. Development of the Drosophila retina, a neurocrystalline lattice. Dev. Biol. 53, 217–240 (1976).
Wolff, T. & Ready, D. F. The beginning of pattern formation in the Drosophila compound eye: the morphogenetic furrow and the second mitotic wave. Development 113, 841–850 (1991).
Yuan, Y. P., Schultz, J., Mlodzik, M. & Bork, P. Secreted Fringe-like signaling molecules may be glycosyltransferases. Cell 88, 9–11 (1997).
Irvine, K. & Wieschaus, E. fringe, a boundary-specific signaling molecule, mediates interactions between dorsal and ventral cells during Drosophila wing development. Cell 79, 595–606 (1994).
Kim, J., Irvine, K. D. & Carroll, S. B. Cell recognition, signal induction, and symmetrical gene activation at the dorsal-ventral boundary of the developing Drosophila wing. Cell 82, 795–802 (1995).
Blair, S. S. Limb development: marginal fringe benefits. Curr. Biol. 7, R686–690 (1997).
Fleming, R. J., Gu, Y. & Hukriede, N. A. Serrate -mediated activation of Notch is specifically blocked by the products of the gene fringe in the dorsal compartment of the Drosophila wing imaginal discs. Development 124, 2973–2981 (1997).
Irvine, K. D. & Vogt, T. F. Dorsal-ventral signaling in limb development. Curr. Opin. Cell Biol. 9, 867–876 (1997).
Panin, V. M., Papayannopoulos, V., Wilson, R. & Irvine, K. D. Fringe modulates Notch-ligand interactions. Nature 387, 908–912 (1997).
Ma, C. & Moses, K. wingless and patched are negative regulators of the morphogenetic furrow and canaffect tissue polarity in the developing Drosophila compound eye. Development 121, 2279–2289 (1995).
Xu, T. & Rubin, G. M. Analysis of genetic mosaics in developing and adult Drosophila tissues. Development 117, 1223–1237 (1993).
Pignoni, F. & Zipursky, S. L. Induction of Drosophila eye development by Decapentaplegic. Development 124, 271–278 (1997).
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993).
McNeil, H., Yang, C.-H., Brodsky, M., Ungos, J. & Simon, M. A. mirror encodes a novel PBX-class homeoprotein that functions in the definition of the dorsal-ventral border in the Drosophila eye. Genes Dev. 11, 1073–1082 (1997).
Kehl, B. T., Cho, K.-O. & Choi, K.-W. mirror, a Drosophila homeobox gene in the iroquois complex, is required for sensory organ and alula formation. Development 125, 1217–1227 (1998).
Netter, S., Fauvarque, M.-O., del Corral, R. D., Dura, J.-M. & Coen, D. white+ transgene insertions presenting a dorsal/ventral pattern define a single cluster of homeobox genes that is silenced by the Polycomb -group proteins in Drosophila melanogaster. Genetics 149, 257–275 (1998).
Heberlein, U., Borod, E. R. & Chanut, F. A. Dorsoventral patterning in the Drosophila retina by wingless. Development 125, 567–577 (1998).
Wiersdorff, V., Lecuit, T., Cohen, S. M. & Mlodzik, M. Mad acts downstream of Dpp receptors, revealing a differential requirement for dpp signaling in initiation and propagation of morphogenesis in the Drosophila eye. Development 122, 2153–2162 (1996).
Chanut, F. & Heberlein, U. Role of decapentaplegic in initiation and progression of the morphogenetic furrow in the developing Drosophila retina. Development 124, 559–567 (1997).
Dominguez, M. & Hafen, E. Hedgehog directly controls initiation and propagation of retinal differentiation in the Drosophila eye. Genes Dev. 11, 3254–3264 (1997).
Royet, J. & Finkelstein, R. Establishing primordia in the Drosophila eye-antennal imaginal disc: the roles of decapentaplegic, wingless and hedgehog. Development 124, 4793–4800 (1997).
Borod, E. R. & Heberelin, U. Mutual regulation of decapentaplegic and hedgehog during the initiation of differentiation in the Drosophila retina. Dev. Biol. 197, 187–197 (1998).
Sun, Y. H. et al. white as a reporter gene to detect transcriptional silencers specifying position-specific gene expression during Drosophila melanogaster eye development. Genetics 141, 1075–1086 (1995).
Carthew, R. W. & Rubin, G. M. seven in absentia, a gene required for specification of R7 cell fate in the Drosophila eye. Cell 63, 561–577 (1990).
Choi, K.-W. & Benzer, S. Rotation of photoreceptor clusters in the developing Drosophila eye requires the nemo gene. Cell 78, 125–136 (1994).
Carroll, S. B. & Whyte, J. S. The role of the hairy gene during Drosophila morphogenesis-stripes in imaginal discs. Genes Dev. 3, 905–916 (1989).
Higashijima, S.-I. et al. Dual Bar homeo box genes of Drosophila required in two photoreceptor cells, R1 and R6, and primary pigment cells for normal eye development. Genes Dev. 6, 50–60 (1992).
Treisman, J. E. & Rubin, G. M. wingless inhibits morphogenetic furrow movement in the Drosophila eye disc. Development 121, 3519–3527 (1995).
Wehrli, M. & Tomlinson, A. Independent regulation of anterior/posterior and equatorial/polar polarity in the Drosophila eye; evidence for the involvement of Wnt signaling in the equatorial/polar axis. Development 125, 1421–1432 (1998).
Acknowledgements
We thank H. Bellen, S. Izaddoost, B. Kehl and G. Mardon for comments; J. Kim for fng cDNA, fng mutants and UAS-fng ; K. Kozopas for flip-out flies; E. Knust for Ser antibody; K. Saigo for Bar antibody; Q. Sun for Eq-3 strain; Artavanis-Tsakonas for Notch and Dl antibodies; the Bloomington Stock Center for providing many of the stocks; H. Bellen for use of the confocal microscope; S. Mehta for preliminary work; and B. Kehl for excellent technical assistance. This work was supported by a grant from the NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, KO., Choi, KW. Fringe is essential for mirror symmetry and morphogenesis in the Drosophila eye. Nature 396, 272–276 (1998). https://doi.org/10.1038/24394
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/24394
This article is cited by
-
Synthesis and biological roles of O-glycans in insects
Glycoconjugate Journal (2020)
-
Genetic interactions between Protein Kinase D and Lobe mutants during eye development of Drosophila melanogaster
Hereditas (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.