Abstract
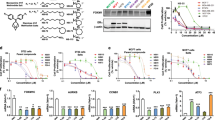
Fenretinide (4-HPR) is a synthetic retinoid with antitumor activity, which induces apoptosis in cancer cell lines of different histotypes. To identify genes contributing to its apoptotic activity in ovarian cancer cells, we monitored, by cDNA arrays, gene expression changes after 4-HPR exposure in A2780, a human ovarian carcinoma cell line sensitive to the retinoid. Among the differentially expressed transcripts, PLAcental Bone morphogenetic protein (PLAB), a proapoptotic gene, was the most highly induced. In a panel of ovarian carcinoma cell lines with different 4-HPR sensitivities, PLAB upregulation was associated with cellular response to 4-HPR, its overexpression increased basal apoptosis and its silencing by small interfering RNA decreased the ability of 4-HPR to induce apoptosis. PLAB induction by 4-HPR was p53- and EGR-1 independent and was regulated, at least in part, by increased stability of PLAB mRNA. PLAB up-modulation by 4-HPR also occurred in vivo: in ascitic cells collected from patients with ovarian cancer before and after 4-HPR treatment, PLAB was upmodulated in 2/4 patients. Our results in certain ovarian cancer cell lines indicate a role for PLAB as a mediator of 4-HPR-induced apoptosis. The correlation of increased PLAB in vivo with antitumor activity remains to be established.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
Accessions
GenBank/EMBL/DDBJ
References
Albertoni M, Shaw PH, Nozaki M, Godard S, Tenan M, Hamou MF et al. (2002). Anoxia induces macrophage inhibitory cytokine-1 (MIC-1) in glioblastoma cells independently of p53 and HIF-1. Oncogene 21: 4212–4219.
Appierto V, Cavadini E, Pergolizzi R, Cleris L, Lotan R, Canevari S et al. (2001). Decrease in drug accumulation and in tumour aggressiveness marker expression in a fenretinide-induced resistant ovarian tumour cell line. Br J Cancer 84: 1528–1534.
Appierto V, Villani MG, Cavadini E, Lotan R, Vinson C, Formelli F . (2004). Involvement of c-Fos in fenretinide-induced apoptosis in human ovarian carcinoma cells. Cell Death Differ 11: 270–279.
Baek SJ, Eling TE . (2006). Changes in gene expression contribute to cancer prevention by COX inhibitors. Prog Lipid Res 45: 1–16.
Baek SJ, Wilson LC, Eling TE . (2002). Resveratrol enhances the expression of non-steroidal anti-inflammatory drug-activated gene (NAG-1) by increasing the expression of p53. Carcinogenesis 23: 425–434.
Balmer JE, Blomhoff R . (2002). Gene expression regulation by retinoic acid. J Lipid Res 43: 1773–1808.
Chiesa F, Tradati N, Grigolato R, Boracchi P, Biganzoli E, Crose N et al. (2005). Randomized trial of fenretinide (4-HPR) to prevent recurrences, new localizations and carcinomas in patients operated on for oral leukoplakia: long-term results. Int J Cancer 115: 625–629.
Clifford JL, Menter DG, Wang M, Lotan R, Lippman SM . (1999). Retinoid receptor-dependent and -independent effects of N-(4-hydroxyphenyl)retinamide in F9 embryonal carcinoma cells. Cancer Res 59: 14–18.
Colombo N, Formelli F, Cantu MG, Parma G, Gasco M, Argusti A et al. (2006). A phase I–II preoperative biomarker trial of fenretinide in ascitic ovarian cancer. Cancer Epidemiol Biomarkers Prev 15: 1914–1919.
Cui W, Yu L, He H, Chu Y, Gao J, Tang L et al. (2001). Cloning of human myeloid-associated differentiation marker (MYADM) gene whose expression was up-regulated in NB4 cells induced by all-trans retinoic acid. Mol Biol Rep 28: 123–138.
De Cecco L, Marchionni L, Gariboldi M, Reid JF, Lagonigro MS, Caramuta S et al. (2004). Gene expression profiling of advanced ovarian cancer: characterization of a molecular signature involving fibroblast growth factor 2. Oncogene 23: 8171–8183.
De Palo G, Mariani L, Camerini T, Marubini E, Formelli F, Pasini B et al. (2002). Effect of fenretinide on ovarian carcinoma occurrence. Gynecol Oncol 86: 24–27.
Dziadek M, Timpl R . (1985). Expression of nidogen and laminin in basement membranes during mouse embryogenesis and in teratocarcinoma cells. Dev Biol 111: 372–382.
Formelli F, Cleris L . (1993). Synthetic retinoid fenretinide is effective against a human ovarian carcinoma xenograft and potentiates cisplatin activity. Cancer Res 53: 5374–5376.
Garaventa A, Luksch R, Lo Piccolo MS, Cavadini E, Montaldo PG, Pizzitola MR et al. (2003). Phase I trial and pharmacokinetics of fenretinide in children with neuroblastoma. Clin Cancer Res 9: 2032–2039.
Hail Jr N, Lotan R . (2001). Mitochondrial respiration is uniquely associated with the prooxidant and apoptotic effects of N-(4-hydroxyphenyl)retinamide. J Biol Chem 276: 45614–45621.
Hosack DA, Dennis Jr G, Sherman BT, Lane HC, Lempicki RA . (2003). Identifying biological themes within lists of genes with EASE. Genome Biol 4: R70.
Kadara H, Schroeder CP, Lotan D, Pisano C, Lotan R . (2006). Induction of GDF-15/NAG-1/MIC-1 in human lung carcinoma cells by retinoid-related molecules and assessment of its role in apoptosis. Cancer Biol Ther 5: 518–522.
Lovat PE, Corazzari M, Goranov B, Piacentini M, Redfern CP . (2004). Molecular mechanisms of fenretinide-induced apoptosis of neuroblastoma cells. Ann NY Acad Sci 1028: 81–89.
Lu X, Errington J, Curtin NJ, Lunec J, Newell D . (2001). The impact of p53 status on cellular sensitivity to antifolate drugs. Clin Cancer Res 7: 2114–2123.
Ma Y, Koza-Taylor PH, DiMattia DA, Hames L, Fu H, Dragnev KH et al. (2003). Microarray analysis uncovers retinoid targets in human bronchial epithelial cells. Oncogene 22: 4924–4932.
Modlich O, Prisack HB, Munnes M, Audretsch W, Bojar H . (2004). Immediate gene expression changes after the first course of neoadjuvant chemotherapy in patients with primary breast cancer disease. Clin Cancer Res 10: 6418–6431.
Myatt SS, Redfern CP, Burchill SA . (2005). p38MAPK-dependent sensitivity of Ewing's sarcoma family of tumors to fenretinide-induced cell death. Clin Cancer Res 11: 3136–3148.
Nagy L, Thomazy VA, Heyman RA, Davies PJ . (1998). Retinoid-induced apoptosis in normal and neoplastic tissues. Cell Death Differ 5: 11–19.
Newman D, Sakaue M, Koo JS, Kim KS, Baek SJ, Eling T et al. (2003). Differential regulation of nonsteroidal anti-inflammatory drug-activated gene in normal human tracheobronchial epithelial and lung carcinoma cells by retinoids. Mol Pharmacol 63: 557–564.
O’Connor PM, Jackman J, Bae I, Myers TG, Fan S, Mutoh M et al. (1997). Characterization of the p53 tumor suppressor pathway in cell lines of the National Cancer Institute anticancer drug screen and correlations with the growth-inhibitory potency of 123 anticancer agents. Cancer Res 57: 4285–4300.
Pellegrini R, Mariotti A, Tagliabue E, Bressan R, Bunone G, Coradini D et al. (1995). Modulation of markers associated with tumor aggressiveness in human breast cancer cell lines by N-(4-hydroxyphenyl) retinamide. Cell Growth Differ 6: 863–869.
Perego P, Giarola M, Righetti SC, Supino R, Caserini C, Delia D et al. (1996). Association between cisplatin resistance and mutation of p53 gene and reduced bax expression in ovarian carcinoma cell systems. Cancer Res 56: 556–562.
Prinetti A, Basso B, Appierto V, Villani MG, Valsecchi M, Loberto N et al. (2003). Altered sphingolipid metabolism in N-(4-hydroxyphenyl)-retinamide-resistant A2780 human ovarian carcinoma cells. J Biol Chem 278: 5574–5583.
Sharma A, Belna J, Logan J, Espat J, Hurteau JA . (2005). The effects of omega-3 fatty acids on growth regulation of epithelial ovarian cancer cell lines. Gynecol Oncol 99: 58–64.
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D et al. (1990). New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82: 1107–1112.
Shishodia S, Gutierrez AM, Lotan R, Aggarwal BB . (2005). N-(4-hydroxyphenyl)retinamide inhibits invasion, suppresses osteoclastogenesis, and potentiates apoptosis through downregulation of I(kappa)B(alpha) kinase and nuclear factor-kappaB-regulated gene products. Cancer Res 65: 9555–9565.
Supino R, Crosti M, Clerici M, Walters A, Cleris L, Zunino F et al. (1996). Induction of apoptosis by fenretinide (4HPR) in human ovarian carcinoma cells and its association with retinoic acid receptor expression. Int J Cancer 65: 491–497.
Tsai MS, Bogart DF, Li P, Mehmi I, Lupu R . (2002). Expression and regulation of Cyr61 in human breast cancer cell lines. Oncogene 21: 964–973.
Veronesi U, De Palo G, Marubini E, Costa A, Formelli F, Mariani et al. (1999). Randomized trial of fenretinide to prevent second breast malignancy in women with early breast cancer. J Natl Cancer Inst 91: 1847–1856.
Veronesi U, Mariani L, Decensi A, Formelli F, Camerini T, Miceli R et al. (2006). Fifteen-year results of a randomized phase III trial of fenretinide to prevent second breast cancer. Ann Oncol 17: 106–171.
Villani MG, Appierto V, Cavadini E, Bettiga A, Prinetti A, Clagett-Dame M et al. (2006). 4-oxo-fenretinide, a recently identified fenretinide metabolite, induces marked G2–M cell cycle arrest and apoptosis in fenretinide-sensitive and fenretinide-resistant cell lines. Cancer Res 66: 3238–3247.
Villani MG, Appierto V, Cavadini E, Valsecchi M, Sonnino S, Curley RW et al. (2004). Identification of the fenretinide metabolite 4-oxo-fenretinide present in human plasma and formed in human ovarian carcinoma cells through induction of cytochrome P450 26A1. Clin Cancer Res 10: 6265–6275.
Wang H, Maurer BJ, Reynolds CP, Cabot MC . (2001). N-(4-hydroxyphenyl)retinamide elevates ceramide in neuroblastoma cell lines by coordinate activation of serine palmitoyltransferase and ceramide synthase. Cancer Res 61: 5102–5105.
Wei JS, Whiteford CC, Cenacchi N, Son CG, Khan J . (2005). BBC3 mediates fenretinide-induced cell death in neuroblastoma. Oncogene 24: 7976–7983.
Zhao Q, Tao J, Zhu Q, Jia PM, Dou AX, Cheng F et al. (2004). Rapid induction of cAMP/PKA pathway during retinoic acid-induced acute promyelocytic leukemia cell differentiation. Leukemia 18: 285–292.
Acknowledgements
We are grateful to Dr Perego for kindly giving us IGROV-1/Pt cells. The study was supported by grants from the Associazione Italiana per la Ricerca sul Cancro, Milan, Italy and the American Cancer Society Research Scholar grant RSG-04-170-01-CNE to JRL.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Appierto, V., Villani, M., Cavadini, E. et al. Analysis of gene expression identifies PLAB as a mediator of the apoptotic activity of fenretinide in human ovarian cancer cells. Oncogene 26, 3952–3962 (2007). https://doi.org/10.1038/sj.onc.1210171
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210171