Abstract
Detrimental decision-making is a major problem among violent offenders. Non-invasive brain stimulation offers a promising method to directly influence decision-making and has already been shown to modulate risk-taking in non-violent controls. We hypothesize that anodal transcranial direct current stimulation (tDCS) over the right dorsolateral prefrontal cortex beneficially modulates the neural and behavioral correlates of risk-taking in a sample of violent offenders. We expect offenders to show more risky decision-making than non-violent controls and that prefrontal tDCS will induce stronger changes in the offender group. In the current study, 22 male violent offenders and 24 male non-violent controls took part in a randomized double-blind sham-controlled cross-over study applying tDCS over the right dorsolateral prefrontal cortex. Subsequently, participants performed the Balloon Analogue Risk Task (BART) during functional magnetic resonance imaging (fMRI). Violent offenders showed significantly less optimal decision-making compared to non-violent controls. Active tDCS increased prefrontal activity and improved decision-making only in violent offenders but not in the control group. Also, in offenders only, prefrontal tDCS influenced functional connectivity between the stimulated area and other brain regions such as the thalamus. These results suggest baseline dependent effects of tDCS and pave the way for treatment options of disadvantageous decision-making behavior in this population.
Similar content being viewed by others
Introduction
Violent crimes pose a significant burden on society as they involve enormous economic and societal costs. Existing evidence indicates that what leads someone to commit violent crimes comprises interactions of biological, psychosocial, and environmental factors1. Despite advances in our understanding of the etiology of violent offending, their link to effective treatment options is often missing. Popular therapy programs such as cognitive behavioral therapy may fail in certain groups more often, such as in violent offenders who tend to make risky decisions2. The exploration of biological pathways may advance our knowledge of the mechanisms of the different cognitive and emotional dysfunctions in violent populations, in order to shed light on ways to reduce these deficits.
One prominent explanatory biological pathway is described in the prefrontal dysfunction hypothesis3. Based on this hypothesis, studies have shown a hypoactivity of the prefrontal cortex in violent offenders which can be linked to emotion regulation as well as cognitive deficits, such as in the context of risky decision-making4. A promising non-invasive method that can alter neural functioning and thereby influence cognition and behavior is transcranial direct current stimulation (tDCS). Prefrontal tDCS has already been proven to be effective for neural modulation in various populations including criminal offenders5,6. In the current study, we aim to directly compare the effects of prefrontal tDCS on risky decision-making in violent offenders and non-violent controls at both the behavioral and neural level for the first time.
When decisions are made under risk, economic models state that individuals need to integrate information about possible wins and losses and their respective probabilities7. Individuals choose the option to which they assign the highest subjective value. Such situations are experimentally modeled by gambling tasks such as the Balloon Analogue Risk Task (BART)8. In the BART, participants inflate a virtual balloon to maximize their reward while facing the risk of explosion and losing the reward. High numbers of inflations have been linked to substance use or sexual risk behavior 8 and were associated with impulsive and psychopathic personality traits in a non-forensic sample9.
In offender samples, aberrant decision-making behavior may originate from general deficits in executive functioning10,11,12, cognitive control13,14, and emotion regulation15,16. A meta-analysis showed an increased propensity of aggressive individuals to engage in risky choices2. This propensity has been linked to different measures of aggression as well as poorer treatment outcomes17.
Only a few studies have examined the neural underpinnings of behavioral differences between offenders and non-offenders. Among other brain regions, the dorso-lateral prefrontal cortex (dlPFC), a central region for cognitive control and decision-making, showed aberrant activation patterns. Prehn et al. showed a diminished response in the anterior cingulate as well as the prefrontal cortex especially in emotionally hypo-reactive offenders during decision-making under risk4. Adolescents with conduct and substance use problems also showed hypoactivation in the prefrontal cortex, anterior insula, anterior cingulate and subcortical regions during risky decision-making18. This largely overlaps with the findings from a meta-analysis of individuals on the anti-social spectrum19.
As a non-invasive method, tDCS can alter cortical excitability through the subthreshold modulation of neuronal membrane potentials20,21. Anodal stimulation depolarizes the neuronal membrane and thereby increases spontaneous neuronal activity and the probability to induce an action potential22,23. Beneficial effects of unilateral or bilateral stimulation of the dlPFC have been found in healthy participants24 as well as patient groups with ADHD or addiction disorders25,26,27,28. However, across studies, unilateral stimulation was identified to reduce risk-taking behavior more effectively than bilateral protocols29, with right anodal/left cathodal stimulation as the most effective protocol30. Using either right anodal/left cathodal or left anodal/right cathodal dlPFC stimulation, a reduction of risk-taking in the BART was reported in a healthy sample31. Anodal stimulation of the right dlPFC also lowered risk-aversion in a gain frame condition, leading to optimized decision-making behavior32. Interestingly, in older adults as well as marijuana users, anodal tDCS over the dlPFC increased risk-taking33,34.
In the BART, earnings are largest if the participant finds the optimal number of button presses. Therefore, general task performance such as the total earnings can indicate successful decision-making. While tDCS seems to influence earnings in the BART as well35, it is unknown if tDCS can optimize decision-making in criminal offenders. Some support for this notion comes from the finding that in paedophilic offenders, cognitive control was improved after the stimulation of the left dlPFC compared to sham stimulation36. Offenders’ moral decision-making as well as self-reported aggression were also influenced advantageously by prefrontal tDCS6,37,38. In line with these findings, recent literature reviews on tDCS effects on aggression in healthy as well as criminal subjects found reductions in aggressive behavior using both self-report and behavioral measures39,40. In participants with psychopathic traits, increased inhibitory control due to tDCS was positively correlated with greater psychopathy scores41. In violent offenders, dlPFC stimulation reduced brain activity during emotion regulation5. Although investigation of brain activity after tDCS over the dlPFC in non-violent populations indicates increased activation during risk-taking in the BART at the stimulation site as well as the anterior cingulate42, tDCS may affect the whole brain via connectivity changes42,43 in both non-violent and offender groups.
In the current study, we compare the effects of prefrontal tDCS on risky decision-making in violent offenders and non-violent controls at both the behavioral and neural level for the first time. The goal of this study is to demonstrate that tDCS can be used to beneficially modulate the neural and behavioral correlates of risk-taking in a sample of violent offenders. Specifically, this study tests whether anodal stimulation of the right dlPFC facilitates its cortical excitability, reflected by increased right dlPFC activation during risk-taking after the active compared to the sham tDCS condition. We anticipate a tDCS-induced change in the connectivity of the dlPFC and other regions related to risk-taking. Thus, we expect to improve cognitive control and reduce risk-taking after active tDCS. We hypothesize that offenders show more risky decision-making than non-violent controls and hence, that prefrontal tDCS will induce stronger changes in the neural and behavioral correlates of risk-taking in the offender group.
Methods
Participants
We recruited 22 male violent offenders in the age range from 23 to 52 years (M = 37.2, SD = 8.6) at the parole office in Aachen, Germany. Inclusion criteria for offenders included commission of at least two violent crimes. The distribution of index crimes was as follows: 5 offenders committed or attempted murder or manslaughter, 11 committed assault or physical injury, 5 committed (armed) robbery, and one committed sexual assault. Due to the high rate of psychiatric diagnoses in this group, only participants reporting acute alcohol and drug abuse were excluded. For further information on the lifetime psychiatric diagnoses and medication of the violent offender group, please see supplementary Table 1. The non-violent control group included 24 men aged between 19 and 58 years (M = 31.4, SD = 10.5) without any former or current neurological or psychiatric disease. Non-violent controls were recruited by advertisements in Aachen and did not have any criminal record. For both groups, exclusion criteria for MRI were applied, such as metal implants and large-scale tattoos. Participants with neurological diseases were excluded. All participants were right-handed.
Ethics
Participation was voluntary and participants gave written and informed consent. The experimental procedures were in accordance with the declaration of Helsinki (World Medical Association, 2013) and the study was approved by the Ethics Committee of the Medical Faculty of the RWTH Aachen University. The trial protocol was registered at ClinicalTrials.gov (NCT03036683) on 01/02/2017.
Study design
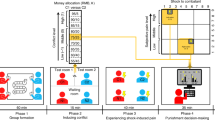
We implemented a double-blind sham-controlled cross-over design. Measurements took place in the Department of Psychiatry, Psychotherapy and Psychosomatics of the University Hospital RWTH Aachen, Germany. Participants visited the laboratory twice and underwent both active and sham tDCS in a randomized order with at least 48 h in between. This time period has been shown to be sufficient to wash out tDCS effects and prevent carry over effects44,45,46. The cross-over design increases statistical power and is advantageous for heterogeneous groups since every subject serves as a control for himself. The average time interval between measurements was five days with a maximum of two weeks.
Procedure
Non-violent controls were interviewed about demographic information and psychiatric symptoms using the SCID light47. They filled in questionnaires, performed a verbal intelligence test, and tests of executive functioning (Trail Making Test-A/-B, digit span forward and backwards)48. We assessed demographics, criminal records and psychopathology in the offender group at a separate appointment, which included a structured interview on their committed crimes, the German versions of DIPS (Diagnostic Interview for Mental Disorder)49, the PCL-R (Hare Psychopathy Checklist—Revised; German version)50, an assessment of verbal intelligence (WST)51, and various self-report questionnaires. The self-report questionnaires included German versions of the Becks Depression Inventory (BDI-II)52, the Aggression Questionnaire53, the Proactive Reactive Aggression Questionnaire54, the Barratt Impulsiveness Scale (BIS-11)55, and the Psychopathic Personality Inventory-Revised (PPI-R)56.
Prior to the stimulation, participants were told that they could increase their financial compensation with their performance in the fMRI tasks by earning the money saved in the total account during the BART. In fact, all individuals received the same fixed amount of additional money after debriefing.
Participants in both groups practiced the BART and then performed an alternating 1-back, 2-back or 3-back working memory task57 during the 20 min of stimulation. The working memory task was only applied to facilitate stimulation effects in the dlPFC and the results were not of interest for the current study. It has been shown that tasks that activate the stimulated brain region can enhance the stimulation effect compared to being at rest during the stimulation58. After that, participants went to the 3 Tesla scanner to perform a computer car race game (Carmageddon, Torus Games, Bayswater, Australia, 2000, reported elsewhere) followed by the BART and an anatomical scan (Fig. 1). In total, approximately 40 min passed in between the tDCS termination and the BART. After the session, participants completed a tDCS questionnaire assessing the blinding of participants and possible adverse effects as well as affective responses using the Positive and Negative Affect Schedule (PANAS)59. The results of the PANAS are reported in the supplementary material (supp. Tables 2 and 3).
Stimulation protocol
A DC-stimulator (neuroCare Group GmbH, Munich, Germany) was used to stimulate the right dlPFC. A code system assigned the participant to the sham and active condition in a randomized order, keeping both the participant and experimenter blinded. The electrodes were positioned according to the international 10–20 EEG system. The anode (5 × 5 cm) was placed at the F4 position to target the right dlPFC. The cathode (10 × 10 cm) served as a reference electrode. It was placed over the left eyebrow with at least 7 cm distance to the anode to avoid a current flow over the scalp. The electrodes were enveloped in sponges soaked in 0.9% NaCl to increase conductivity and were attached using rubber bands. After a ramp-up phase of 40 s, a direct current flow of 2 milliampere lasted for 20 min. Participants reported only mild sensations of tickling or itching during the stimulation and could not differentiate between active and sham stimulation (χ2(1) = 1.51, p = 0.219).
BART
The applied BART is a well-established computerized risk-taking task adapted for the use in the MRI60,61. Participants had to inflate virtual balloons to earn an increasing amount of money. In case of an explosion, the money from that trial was lost and a new balloon appeared. Control balloons would not explode or fetch money. Before each inflation, participants had the option to save their temporal reward to their permanent account. For further details and visualization, see supplementary Fig. 1. The BART lasted approximately 30 min.
Behavioral analysis
Behavioral analyses of different BART parameters were conducted using R Studio and the lme4 package62. The primary outcome was measured by the mean number of pumps in successfully cashed out trials (adjusted pumps). Explosion trials were excluded for the calculation since they represent the random structure of the program, but the number of explosion trials was used as covariate to control for their indirect influence. The secondary outcome was the total earnings. Mixed models for both dependent variables were computed including group (controls vs. offenders), condition (sham vs. active stimulation) and session (first vs. second measurement) as fixed factors, as well as a random intercept and the covariate of explosions for the adjusted pump model. All main effects and interactions were modeled based on theoretical assumptions (model space) and selected based on goodness of fit tests comparing the models with the anova function of the lme4 package. For an overview on the model space, see Supplementary Tables 4, 5. For both dependent variables, a model with the two-way interactions of group and stimulation as well as stimulation and session were selected. Type III analysis with Satterthwaite methods were performed for the selected models. Tukey correction at an original α = 0.05 was applied for post-hoc pairwise comparisons and degrees of freedom were adjusted via the Kenward-Roger method.
Bonferroni corrected Pearson correlations of the adjusted pumps and different personality scores were computed for each subgroup. Additionally, sample characteristics were compared between groups using independent sample t-tests.
Image acquisition and analysis
Data acquisition, preprocessing and first level modelling are described in the supplements.
For the whole brain analysis, a GLM was fitted using a flexible factorial design on the parametric modulation of the BOLD response modeled via the balloon size (representing increasing risk). Group (offenders vs. controls), tDCS condition (active vs. sham), as well as session (first vs. second) were included as fixed factors. Additionally, a random intercept was included in the model. A region of interest (ROI) analysis of the right dlPFC as the target area of stimulation was conducted using the same design. An inclusive mask of Brodmann areas 9 and 46 was created using the WFU PickAtlas63. Additionally, the activation in this mask was extracted and included in the correlational analysis.
To examine changes in functional task-dependent connectivity related to risky decision-making after tDCS stimulation, we conducted a psychophysiological interaction (PPI) analysis using the generalized PPI toolbox for SPM64. Therefore, we defined the rdlPFC as the seed region, again using the aforementioned mask. For each individual, the deconvolved time series of this seed region was extracted. The event types (inflation vs. baseline) were convolved with the canonical hemodynamic response function (HRF) to create the psychological regressor. An interaction term (reflecting the PPI) was calculated by multiplying the time series of the psychological variable with the time series of the seed. For the group-level analysis, we applied the same flexible factorial design reported for brain activation including the factors group, session, and condition. Whole brain results were family-wise error (FWE) corrected at cluster level using a height threshold of p < 0.05 and a cluster-defining threshold of p < 0.001.
Results
Questionnaires
Offenders reported elevated levels of depression, aggression, impulsivity as well as psychopathy compared with controls (Table 1).
Because verbal IQ and BDI-II scores were potentially confounding variables and differed between groups, correlations with the adjusted pumps and total earnings in the BART were computed separately for each group. No significant correlation emerged in either group (Supplementary Table 6).
BART
Goodness of fit tests (Table 2) showed that for both adjusted pumps and earnings, the models which included the main effects of group, stimulation, and session as well as the interaction of group and stimulation, and stimulation and session were significantly better than the other models of the given model space (see Supplementary Tables 4 and 5 for the full model space).
Concerning adjusted pumps, the main effect of group was significant (F(1,42.446) = 15.541, p < 0.001) with controls having a higher number of pumps than offenders. In addition, the interaction of session and stimulation was significant (F(2,42.463) = 15.273, p = < 0.001) and the interaction of group and stimulation (F(1,41.861) = 3.881, p = 0.055) was marginally significant (Fig. 2).
Estimated marginal means of adjusted pumps (mean number of balloons in non-explosion trails) and 95% confidence intervals for session one and two in the active and sham stimulation condition separated for the control and offender group. Asterisks indicate significant difference at p < 0.05 corrected for multiple comparisons applying the Tukey method.
Comparisons of the estimated marginal means (Table 3) showed that there was a significant difference between the active and sham stimulation conditions in session two (t(60.6) = − 2.803 p = 0.0334), but not in session one (t(59.8) = 0.708, p = 0.894). Session one and two differed in the active condition (t(61.0) = − 3.881, p = 0.0014), but not during the sham condition (t(59.5) = − 0.0434, p = 0.996). The marginally significant interaction of group and stimulation showed a significant difference between controls and offenders in the sham condition (t(58.8) 4.46, p = 0.002) and to a smaller degree in the active condition (t(58.8) = 4.46, p = 0.045). The adjusted pumps in the control group did not differ between sham and active conditions (t(40.6) = − 0.396, p = 0.979), but in the offender group, the adjusted pumps differed after participants underwent active and sham stimulation (t(42.3) = -3.042, p = 0.020).
The adjusted pumps correlated negatively with the RPQ (r = − 0.416, p < 0.001) and the AQ (r = − 0.474, p < 0.001) sum scores. No significant correlations emerged with psychopathy scores (PPI-R, r = − 0.231, p = 0.046) or impulsivity (BIS-11, r = − 0.244, p = 0.038). In the subgroups, no significant correlations remained (supplementary Tables 7–9). Notably, in the offender group a large effect of (r = − 0.531, p = 0.013) with the AQ was observed (Cohen, 1988) at uncorrected level.
Total earnings were significantly influenced by group (F(1,42.59) = 12.85, p < 0.001), stimulation (F(1,40.93) = 4.64, p = 0.037), and session (F(1,40.81) = 15.92, p < 0.001). The interaction of group and stimulation (Fig. 3) was significant as well (F(1,41,029) = 6.28, p = 0.016), but the interaction of session and stimulation was not (F(1,42.49) = 0.58, p = 0.451).
Estimated marginal means of total earnings (calculated as sum across all cash trials) and 95% confidence intervals for the active and sham stimulation condition separated for the control and offender group. Asterisks indicate significant difference at p < 0.05 corrected for multiple comparisons applying the Tukey method.
Post hoc comparisons showed that the controls and offenders significantly differed in their total earnings in the sham condition (t(65.2) = 4.36, p < 0.001), but not in the active condition (t(66.1) = 1.94, p = 0.222). The sham and active condition did not differ in this outcome variable in controls (t(40.1) = 0.272, p = 0.993), but there were differences across conditions in the offender group (t(41.4) = -3.133, p = 0.016) (see Table 4 for estimated marginal means and confidence intervals). Total earnings correlated significantly with the adjusted pumps (r = 0.872, p < 0.001) and the RPQ (r = -0.359, p = 0.001) in the whole sample. In the subgroups, no significant correlations of the total earnings and personality measures were observed.
Functional imaging
Inflating balloons in the BART compared to saving led to increased activation in the midcingulate cortex (MCC), insula, striatal, parietal as well as prefrontal regions. In addition, cerebellar and occipital regions were involved (supplementary Table 10).
The group comparison yielded a more pronounced risk-related neural activation (risky > control balloons) in the offender compared to the control group (Fig. 4, Table 5). Differences were detected in the motor cortex, dorsolateral and medial frontal cortex, the cuneus, as well as in occipital areas. The opposite contrast did not indicate any significant cluster that was more activated in controls compared to offenders.
tDCS effects
We performed a ROI analysis in the right dlPFC to test if active stimulation increased activity in the target area. There was no main effect of condition (active > sham), but a significant interaction of tDCS condition × group at a threshold of p < 0.001 uncorrected. The reported interaction holds significance only without adjustment for multiple comparisons, which has to be regarded with caution. Nevertheless, directed t-tests showed increased activation after active stimulation compared to sham in the offender group (x = 36 y = 42 z = 40, t = 4.46, k = 26) at a threshold of p < 0.001 uncorrected and applying FWE-correction at p < 0.05. In the control group, the stimulation condition had no significant effect on right dlPFC activity (t = 0.48, puncorr = 0.272, pFWE = 1). Activity in this region correlated positively with the adjusted pumps in the whole sample (r = 0.289, p = 0.008) only at uncorrected level across both groups. On the whole-brain level, the contrast active > sham yielded no significant results across groups. Directed t-tests revealed significant activation patterns of the contrast sham > active in the offender but not in the control group. After sham compared to active stimulation, offenders showed greater activation in the MCC as well as motor regions (Fig. 5, Table 6).
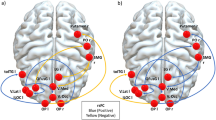
(A) ROI analysis of the right dlPFC, active > sham in the offender group, FWE-corrected, p < 0.05; (B) Risk-related neural activity increases after sham compared to active stimulation in the offender group. Cluster-level FWE-corrected, p < 0.05 (at cluster defining threshold at p < 0.001), minimal cluster threshold k = 225.
Psychophysiological Interactions
In the offender group, the PPI analysis revealed significant risk-related changes after active compared to sham stimulation in neural connectivity of the right dlPFC and other prefrontal and parietal regions, as well as the bilateral thalamus as shown in Fig. 6 and Table 7. No connectivity changes were observed for the control group.
Discussion
We tested the hypothesis that non-invasive brain stimulation may have the potential to reduce aggression in violent offenders. Indeed, tDCS of the right dlPFC affected decision-making and its neural underpinnings in violent offenders. Unexpectedly, offenders compared to non-violent controls overall behaved risk-averse in the BART, which led to poorer task outcomes. After active tDCS, this difference between offenders and non-violent controls was reduced as offenders showed riskier decision-making and thereby improved their performance after receiving active compared to sham stimulation. Active tDCS also only affected the offender and not the control group at the neural level underlining the behavioral modulation. In the control group, tDCS neither affected neural activity nor decision-making behavior.
Anodal prefrontal tDCS compared to sham stimulation decreased activation in different brain regions only in the offender and not in the control group. Reduced activity is in line with previous results on emotion regulation in offenders5. Since MCC activity changes have been related to risk-taking as well as threat processing65,66, we speculate that its deactivation contributes to the observed decrease of risk-avoidance in offenders. Our results complement findings that suggest complex tDCS effects including activation and deactivation depended on prior brain states67,68.
As in other studies42,69, seed-based connectivity of the right dlPFC showed extensive effects of tDCS only in the offender group specifically in subcortical regions such as the thalamus as well as occipital regions. This network re-organization may have supported observed performance improvements. The thalamus has also been linked to aggression in offender and patient samples70,71. It is an important hub for the organization of multiple cortical brain networks thereby being critical for integrating diverse information72.
Our behavioral results, showing a reduced rate of successful trials and probably more cautious approach behavior in violent offenders than in the control group, may query the image of offenders as impulsive and reckless individuals that are often linked to high aggression or clinical concepts of psychopathy73. This behavioral difference might not be influenced primarily by aggressiveness and impulsivity but cognitive processes that are important for optimal decision-making. In the violent offender group, IQ levels were lower than in the control group. Although we do not have an estimation for transfer or learning abilities, but only assessed verbal intelligence, this might point to generally lower cognitive abilities in the criminal offender group. Possibly, there were more problems in learning and estimating an optimal decision behavior in this group not due to reckless behavior, but due to other mental abilities that were missing for a good task performance. Although Snowden et al. found a correlation of specific psychopathic traits with risk-taking behavior in the BART, they did not find increased risk-taking in offenders compared to a community sample74. Others only reported minor differences comparing emotional hyperreactive offenders and controls4. In our study, psychopathy was weakly related to risk-taking only across the whole sample, which reflects the mixed75 and null findings76 of others. Still, direct comparisons of risk-taking in offenders and non-violent control groups remain scarce, and the present results contribute to filling this gap.
The reduced riskiness may question the ecological validity of the pumps in the BART in this population. While many studies found robust correlations between the BART performance and other risk indices in healthy controls8,77, no correlations with self-reported real life risk-taking were found in imprisoned inmates76. This is similarly supported in our findings because we did not find a correlation with self-reported impulsivity. We suggest that the number of pumps in the BART may reflect the ability to adapt a behavior to maximize rewards. Offenders had more difficulties adapting, as indicated by smaller earnings. Adapting choices in the BART requires impulse control, working memory, and learning capacities which are impaired in offenders11,78. Offenders may not adjust their behavior based on the available information79,80 and avoid losing even small wins because they depend more on the monetary reward. Such negative correlations of socio-economic status and risk-avoidance have been observed in older adults81. Moreover, it should be noted that this sample was on probation, potentially influencing their behavior as they may show more socially desirable behavior. However, social desirability was not evident in the self-report questionnaires on aggression (AQ and RPQ) since offenders described themselves as more aggressive as compared to the control group.
The described cognitive deficits may make violent offenders, like other clinical populations82, more susceptible to the effects of prefrontal tDCS than non-violent individuals. In the present study, only the offender group exhibited changes in behavior and neural activity after prefrontal tDCS. This heightened susceptibility supports our initial hypothesis and is in line with the growing body of literature on prefrontal deficits in violent offenders3,83,84. Prefrontal malfunction in offenders has been related to cognitive deficits78,85 and poor decision-making11. Facilitating prefrontal activity by tDCS might reduce this deficit. The results of a previous study comparing total earnings in the BART between a cathodal, anodal, and sham group35 may support this interpretation. Although the authors find differences in the form of lowered performance in the cathodal group, this is similar to our findings, considering the opposing effects of anodal and cathodal stimulation. Interestingly, in the non-violent control group, decision-making was closer to the reward optimization strategy and hence may reflect a ceiling effect unaffected by tDCS. Thus, tDCS effects may depend on the baseline levels of behavior and neural activation86. Moreover, individual differences such as learning capacity or smoking status may influence the direction of modulation effects87,88.
Limitations
The large time gap between the stimulation and the BART entailed the risk of reduced tDCS effects. Studies on tDCS over the motor cortex suggest long-lasting effects 60–120 min after stimulation89. It is unclear if the applied stimulation in the current study outlasted the time interval of approximately 40 min until the task started. The current sample appears rather small but it is comparable with other studies in the field4,79 and the cross-over design created the optimal sham control group. The offender group also appeared heterogeneous in their offenses, personality traits, psychiatric symptoms, and demographic variables. While representing the actual heterogeneity in this population, it remains unclear which characteristics account for the observed deficits. Larger samples would enable the investigation of offender subgroups. The reported interaction of stimulation and group for adjusted pumps was only marginally significant and the interaction of group and stimulation on dlPFC activity did not hold significance when correcting for multiple comparisons. This may decrease reliability of the findings. Nevertheless, in both cases the post hoc comparisons were significant as predicted by our hypotheses.
Conclusion
Our results suggest that prefrontal anodal tDCS operates in a baseline-dependent manner with violent offenders being more susceptible. Against the hypothesis, prefrontal tDCS increased risk-taking behavior, but a closer inspection of the data revealed an optimized decision-making performance in offenders. In offenders, activity and connectivity changes in a wide-ranging network of cortical and sub-cortical regions in response to active tDCS support the conclusion that behavioral changes are related to reorganized brain function. Although our results do not indicate that tDCS is suitable for reducing risk-taking in offenders, this proof-of-concept study corroborates the possibility to treat decision-making deficits in this population.
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available since making the data publicly available would contradict the agreement with the local ethics committee. Datasets are available from the corresponding author on reasonable request.
References
Barnes, J. C., Raine, A. & Farrington, D. P. The interaction of biopsychological and socio-environmental influences on criminological outcomes. Justice Q. 39, 26–50. https://doi.org/10.1080/07418825.2020.1730425 (2022).
Kuin, N., Masthoff, E., Kramer, M. & Scherder, E. The role of risky decision-making in aggression: A systematic review. Aggress. Violent Behav. 25, 159–172. https://doi.org/10.1016/J.AVB.2015.07.018 (2015).
Yang, Y. & Raine, A. Prefrontal structural and functional brain imaging findings in antisocial, violent, and psychopathic individuals: A meta-analysis. Psychiatry Res. Neuroimaging 174, 81–88. https://doi.org/10.1016/j.pscychresns.2009.03.012 (2009).
Prehn, K. et al. Neural correlates of risk taking in violent criminal offenders characterized by emotional hypo- and hyper-reactivity. Soc. Neurosci. 8, 136–147. https://doi.org/10.1080/17470919.2012.686923 (2013).
Hofhansel, L. et al. Stimulating the criminal brain: Different effects of prefrontal tDCS in criminal offenders and controls. Brain Stimul. 13, 1117–1120. https://doi.org/10.1016/j.brs.2020.03.022 (2020).
Molero-Chamizo, A., Martín Riquel, R., Moriana, J. A., Nitsche, M. A. & Rivera-Urbina, G. N. Bilateral prefrontal cortex anodal tDCS effects on self-reported aggressiveness in imprisoned violent offenders. Neuroscience 397, 31–40. https://doi.org/10.1016/j.neuroscience.2018.11.018 (2019).
Kahneman, D. & Tversky, A. Prospect theory: An analysis of decision under risk. Econ. J. Econ. Soc. 47, 263–292 (1979).
Lejuez, C. W. et al. Evaluation of a behavioral measure of risk taking: The Balloon Analogue Risk Task (BART). J. Exp. Psychol. Appl. 8, 75–84. https://doi.org/10.1037/1076-898X.8.2.75 (2002).
Hunt, M. K., Hopko, D. R., Bare, R., Lejuez, C. W. & Robinson, E. V. Construct validity of the Balloon Analog Risk Task (BART): Associations with psychopathy and impulsivity. Assessment 12, 416–428. https://doi.org/10.1177/1073191105278740 (2005).
Ogilvie, J. M., Stewart, A. L., Chan, R. C. K. & Shum, D. H. K. Neuropsychological measures of executive function and antisocial behavior: A meta-analysis. Criminology 49, 1063–1107. https://doi.org/10.1111/j.1745-9125.2011.00252.x (2011).
Syngelaki, E. M., Moore, S. C., Savage, J. C., Fairchild, G. & van Goozen, S. H. M. Executive functioning and risky decision making in young male offenders. Crim. Justice Behav. 36, 1213–1227. https://doi.org/10.1177/0093854809343095 (2009).
Humenik, A. M., Grounds, Z. K., Mayer, H. M. & Dolan, S. L. A systematic review of executive cognitive function in intimate partner violent offenders. Aggress. Violent Behav. 54, 101407. https://doi.org/10.1016/J.AVB.2020.101407 (2020).
Cooper, A. N., Seibert, G. S., May, R. W., Fitzgerald, M. C. & Fincham, F. D. School burnout and intimate partner violence: The role of self-control. Pers. Individ. Dif. 112, 18–25. https://doi.org/10.1016/J.PAID.2017.02.047 (2017).
Szczypiński, J. et al. Abnormal behavioral and neural responses in the right dorsolateral prefrontal cortex during emotional interference for cognitive control in pedophilic sex offenders. J. Psychiatr. Res. 151, 131–135. https://doi.org/10.1016/J.JPSYCHIRES.2022.04.012 (2022).
Roberton, T., Daffern, M. & Bucks, R. S. Maladaptive emotion regulation and aggression in adult offenders. Psychol. Crime Law 20, 933–954. https://doi.org/10.1080/1068316X.2014.893333 (2014).
Garofalo, C., Neumann, C. S. & Velotti, P. Psychopathy and aggression: The role of emotion dysregulation. J. Interpers. Violence 36, NP12640-64. https://doi.org/10.1177/0886260519900946 (2021).
Fishbein, D. et al. Deficits in behavioral inhibition predict treatment engagement in prison inmates. Law Hum. Behav. 33, 419–435. https://doi.org/10.1007/S10979-008-9163-7/METRICS (2009).
Crowley, T. J. et al. Risky decisions and their consequences: Neural processing by boys with Antisocial Substance Disorder. PLoS One 5, 1–20. https://doi.org/10.1371/JOURNAL.PONE.0012835 (2010).
Dugré, J. R. et al. Neurofunctional abnormalities in antisocial spectrum: A meta-analysis of fMRI studies on Five distinct neurocognitive research domains. Neurosci. Biobehav. Rev. 119, 168–183. https://doi.org/10.1016/J.NEUBIOREV.2020.09.013 (2020).
Nitsche, M. A. & Paulus, W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J. Physiol. 527(Pt 3), 633–639. https://doi.org/10.1111/J.1469-7793.2000.T01-1-00633.X (2000).
Nitsche, M. A. et al. Pharmacological modulation of cortical excitability shifts induced by transcranial direct current stimulation in humans. J. Physiol. 553, 293–301. https://doi.org/10.1113/JPHYSIOL.2003.049916 (2003).
Stagg, C. J., Antal, A. & Nitsche, M. A. Physiology of transcranial direct current stimulation. J. ECT 34, 144–152. https://doi.org/10.1097/YCT.0000000000000510 (2018).
Nitsche, M. A. et al. Shaping the effects of transcranial direct current stimulation of the human motor cortex. J. Neurophysiol. 97, 3109–3117. https://doi.org/10.1152/JN.01312.2006 (2007).
Wiegand, A., Sommer, A., Nieratschker, V. & Plewnia, C. Improvement of cognitive control and stabilization of affect by prefrontal transcranial direct current stimulation (tDCS). Sci. Rep. https://doi.org/10.1038/S41598-019-43234-2 (2019).
Allenby, C. et al. Transcranial direct current brain stimulation decreases impulsivity in ADHD. Brain Stimul. 11, 974–981. https://doi.org/10.1016/J.BRS.2018.04.016 (2018).
Weidler, C. et al. Consequences of prefrontal tDCS on inhibitory control and reactive aggression. Soc. Cogn. Affect. Neurosci. 17, 120–130. https://doi.org/10.1093/SCAN/NSAA158 (2022).
Boudewyn, M. A., Scangos, K., Ranganath, C. & Carter, C. S. Using prefrontal transcranial direct current stimulation (tDCS) to enhance proactive cognitive control in schizophrenia. Neuropsychopharmacology 45, 1877–1883. https://doi.org/10.1038/S41386-020-0750-8 (2020).
Alizadehgoradel, J. et al. Repeated stimulation of the dorsolateral-prefrontal cortex improves executive dysfunctions and craving in drug addiction: A randomized, double-blind, parallel-group study. Brain Stimul. 13, 582–593. https://doi.org/10.1016/j.brs.2019.12.028 (2020).
Khaleghi, A., Pirzad Jahromi, G., Zarafshan, H., Mostafavi, S. A. & Mohammadi, M. R. Effects of transcranial direct current stimulation of prefrontal cortex on risk-taking behavior. Psychiatry Clin. Neurosci. 74, 455–465. https://doi.org/10.1111/pcn.13025 (2020).
Brevet-Aeby, C., Brunelin, J., Iceta, S., Padovan, C. & Poulet, E. Prefrontal cortex and impulsivity: Interest of noninvasive brain stimulation. Neurosci. Biobehav. Rev. 71, 112–134. https://doi.org/10.1016/j.neubiorev.2016.08.028 (2016).
Fecteau, S. et al. Activation of prefrontal cortex by transcranial direct current stimulation reduces appetite for risk during ambiguous decision making. J. Neurosci. 27, 6212–6218. https://doi.org/10.1523/JNEUROSCI.0314-07.2007 (2007).
Ye, H. et al. Activation of the prefrontal cortex by unilateral transcranial direct current stimulation leads to an asymmetrical effect on risk preference in frames of gain and loss. Brain Res. 1648, 325–332. https://doi.org/10.1016/j.brainres.2016.08.007 (2016).
Boggio, P. S. et al. Modulation of decision-making in a gambling task in older adults with transcranial direct current stimulation. Eur. J. Neurosci. 31, 593–597. https://doi.org/10.1111/j.1460-9568.2010.07080.x (2010).
Boggio, P. S. et al. Modulation of risk-taking in marijuana users by transcranial direct current stimulation (tDCS) of the dorsolateral prefrontal cortex (DLPFC). Drug Alcohol Depend. 112, 220–225. https://doi.org/10.1016/J.DRUGALCDEP.2010.06.019 (2010).
Guo, H., Zhang, Z., Da, S., Sheng, X. & Zhang, X. High-definition transcranial direct current stimulation (HD-tDCS) of left dorsolateral prefrontal cortex affects performance in Balloon Analogue Risk Task (BART). Brain Behav. https://doi.org/10.1002/brb3.884 (2018).
Pezzoli, P. et al. The effects of acute transcranial direct current stimulation on attentional bias in pedophilic disorder: A preregistered pilot study. Neuromodul. Technol. Neural Interface 24, 879–889. https://doi.org/10.1111/NER.13285 (2021).
Choy, O., Raine, A. & Hamilton, R. H. Stimulation of the prefrontal cortex reduces intentions to commit aggression: A randomized, double-blind, placebo-controlled, stratified, parallel-group trial. J. Neurosci. 38, 6505–6512. https://doi.org/10.1523/JNEUROSCI.3317-17.2018 (2018).
Sergiou, C.-S. et al. Reduced reactive aggression after HD-tDCS and the modulation of electrophysiological responses in a forensic patient sample. Brain Stimul. 14, 1720. https://doi.org/10.1016/j.brs.2021.10.433 (2021).
Casula, A. et al. Non-invasive brain stimulation for the modulation of aggressive behavior—A systematic review of randomized sham-controlled studies. Life 13, 1220. https://doi.org/10.3390/LIFE13051220/S1 (2023).
Knehans, R. et al. Modulating behavioural and self-reported aggression with non-invasive brain stimulation: A literature review. Brain Sci. 12, 200. https://doi.org/10.3390/BRAINSCI12020200 (2022).
Weidacker, K., Weidemann, C. T., Boy, F. & Johnston, S. J. Cathodal tDCS improves task performance in participants high in Coldheartedness. Clin. Neurophysiol. 127, 3102–3109. https://doi.org/10.1016/J.CLINPH.2016.05.274 (2016).
Weber, M. J., Messing, S. B., Rao, H., Detre, J. A. & Thompson-Schill, S. L. Prefrontal transcranial direct current stimulation alters activation and connectivity in cortical and subcortical reward systems: A tDCS-fMRI study. Hum. Brain Mapp. 35, 3673–3686. https://doi.org/10.1002/hbm.22429 (2014).
Holla, B. et al. Effect of prefrontal tDCS on resting brain fMRI graph measures in alcohol use disorders: A randomized, double-blind, sham-controlled study. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 102, 109950. https://doi.org/10.1016/J.PNPBP.2020.109950 (2020).
Boggio, P. S. et al. Effects of transcranial direct current stimulation on working memory in patients with Parkinson’s disease. J. Neurol. Sci. 249, 31–38. https://doi.org/10.1016/j.jns.2006.05.062 (2006).
Fregni, F. et al. Anodal transcranial direct current stimulation of prefrontal cortex enhances working memory. Exp. Brain Res. 166, 23–30. https://doi.org/10.1007/s00221-005-2334-6 (2005).
Jo, J. M. et al. Enhancing the working memory of stroke patients using tDCS. Am. J. Phys. Med. Rehabil. 88, 404–409. https://doi.org/10.1097/PHM.0b013e3181a0e4cb (2009).
Wittchen, H.-U., Wunderlich, U., Gruschwitz, S., Zaudig, M. SKID I. Strukturiertes Klinisches Interview für DSM-IV. Achse I: Psychische Störungen. Interviewheft und Beurteilungsheft. Eine deutschsprachige, erweiterte Bearb. d. amerikanischen Originalversion des SKID I (1997).
Reitan, R. M. Validity of the trail making test as an indicator of organic brain damage. Percept. Mot. Skills 8, 271–276. https://doi.org/10.2466/pms.1958.8.3.271 (1958).
Margraf, J., Cwik, J. C., Pflug, V. & Schneider, S. Strukturierte klinische Interviews zur Erfassung psychischer Störungen über die Lebensspanne: Gütekriterien und Weiterentwicklungen der DIPS-Verfahren. Z Klin Psychol Psychother 46, 176–186. https://doi.org/10.1026/1616-3443/a000430 (2017).
Mokros A, Hollerbach P, Nitschke J HE. Deutsche Version der Hare Psychopathy Checklist-Revised (PCL-R). (2017).
Schmidt, K.-H., Metzler, P. WST-Wortschatztest. Diagnostica 1992;40.
Hautzinger, M., Keller, F., Kühner, C. Beck Depressions-Inventar (BDI-II). Beck Depress (2006).
Buss, A. H. & Perry, M. The aggression questionnaire. J. Pers. Soc. Psychol. 63, 452–459. https://doi.org/10.1037/0022-3514.63.3.452 (1992).
Raine, A. et al. The reactive–proactive aggression questionnaire: Differential correlates of reactive and proactive aggression in adolescent boys. Aggress. Behav. 32, 159–171. https://doi.org/10.1002/AB.20115 (2006).
Patton, J. H., Stanford, M. S. & Barratt, E. S. Factor structure of the barratt impulsiveness scale. J. Clin. Psychol. 51, 768–774. https://doi.org/10.1002/1097-4679(199511)51:6%3c768::AID-JCLP2270510607%3e3.0.CO;2-1 (1995).
Alpers, G. W. & Eisenbarth, H. Psychopathic Personality Inventory-Revised—Deutsche Fassung (Hogrefe, 2008).
Kirchner, W. K. Age differences in short-term retention of rapidly changing information. J. Exp. Psychol. 55, 352–358. https://doi.org/10.1037/h0043688 (1958).
Andrews, S. C., Hoy, K. E., Enticott, P. G., Daskalakis, Z. J. & Fitzgerald, P. B. Improving working memory: The effect of combining cognitive activity and anodal transcranial direct current stimulation to the left dorsolateral prefrontal cortex. Brain Stimul. 4, 84–89. https://doi.org/10.1016/j.brs.2010.06.004 (2011).
Watson, D., Clark, L. A. & Tellegen, A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Pers. Soc. Psychol. 54, 1063–1070. https://doi.org/10.1037/0022-3514.54.6.1063 (1988).
Rao, H., Korczykowski, M., Pluta, J., Hoang, A. & Detre, J. A. Neural correlates of voluntary and involuntary risk taking in the human brain: An fMRI Study of the Balloon Analog Risk Task (BART). Neuroimage 42, 902–910. https://doi.org/10.1016/j.neuroimage.2008.05.046 (2008).
Wagels, L. et al. Blunted insula activation reflects increased risk and reward seeking as an interaction of testosterone administration and the MAOA polymorphism. Hum. Brain Mapp. 38, 4574–4593. https://doi.org/10.1002/HBM.23685 (2017).
Bates, D., Mächler, M., Bolker, B. M. & Walker, S. C. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48. https://doi.org/10.18637/JSS.V067.I01 (2015).
Maldjian, J. A., Laurienti, P. J., Kraft, R. A. & Burdette, J. H. An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. Neuroimage https://doi.org/10.1016/S1053-8119(03)00169-1 (2003).
McLaren, D. G., Ries, M. L., Xu, G. & Johnson, S. C. A generalized form of context-dependent psychophysiological interactions (gPPI): A comparison to standard approaches. Neuroimage 61, 1277–1286. https://doi.org/10.1016/J.NEUROIMAGE.2012.03.068 (2012).
McCormick, E. M. & Telzer, E. H. Adaptive adolescent flexibility: Neurodevelopment of decision-making and learning in a risky context. J. Cogn. Neurosci. 29, 413–423. https://doi.org/10.1162/jocn_a_01061 (2017).
Rahman, S. S. et al. Differential contribution of anterior and posterior midcingulate subregions to distal and proximal threat reactivity in marmosets. Cereb. Cortex 31, 4765–4780. https://doi.org/10.1093/CERCOR/BHAB121 (2021).
Holland, R., Leff, A. P., Penny, W. D., Rothwell, J. C. & Crinion, J. Modulation of frontal effective connectivity during speech. Neuroimage 140, 126–133. https://doi.org/10.1016/J.NEUROIMAGE.2016.01.037 (2016).
Li, L. M. et al. Brain state and polarity dependent modulation of brain networks by transcranial direct current stimulation. Hum. Brain Mapp. 40, 904–915. https://doi.org/10.1002/HBM.24420 (2019).
Indahlastari, A. et al. Individualized tDCS modeling predicts functional connectivity changes within the working memory network in older adults. Brain Stimul. 14, 1205–1215. https://doi.org/10.1016/j.brs.2021.08.003 (2021).
Herpertz, S. C. et al. Brain mechanisms underlying reactive aggression in borderline personality disorder—Sex matters. Biol. Psychiatry 82, 257–266. https://doi.org/10.1016/j.biopsych.2017.02.1175 (2017).
Raine, A. et al. Reduced prefrontal and increased subcortical brain functioning assessed using positron emission tomography in predatory and affective murderers. Behav. Sci. Law 16, 319–332. https://doi.org/10.1002/(SICI)1099-0798(199822)16:3%3c319::AID-BSL311%3e3.0.CO;2-G (1998).
Hwang, K., Bertolero, M. A., Liu, W. B. & D’Esposito, M. The human thalamus is an integrative hub for functional brain networks. J. Neurosci. 37, 5594–5607. https://doi.org/10.1523/JNEUROSCI.0067-17.2017 (2017).
Hare, R. D. & Neumann, C. S. Psychopathy as a clinical and empirical construct. Annu. Rev. Clin. Psychol. 4, 217–246. https://doi.org/10.1146/annurev.clinpsy.3.022806.091452 (2008).
Snowden, R. J., Smith, C. & Gray, N. S. Risk taking and the triarchic model of psychopathy. J. Clin. Exp. Neuropsychol. 39, 988–1001. https://doi.org/10.1080/13803395.2017.1300236 (2017).
Yao, X. et al. Psychopathy and decision-making: Antisocial factor associated with risky decision-making in offenders. Front. Psychol. https://doi.org/10.3389/fpsyg.2019.00166 (2019).
Swogger, M. T., Walsh, Z., Lejuez, C. W. & Kosson, D. S. Psychopathy and risk taking among jailed inmates. Crim. Justice Behav. 37, 439–452. https://doi.org/10.1177/0093854810361617 (2010).
Lauriola, M., Panno, A., Levin, I. P. & Lejuez, C. W. Individual differences in risky decision making: A meta-analysis of sensation seeking and impulsivity with the balloon analogue risk task. J. Behav. Decis. Mak. 27, 20–36. https://doi.org/10.1002/bdm.1784 (2014).
Meijers, J., Harte, J. M., Meynen, G. & Cuijpers, P. Differences in executive functioning between violent and non-violent offenders. Psychol. Med. 47, 1784–1793. https://doi.org/10.1017/S0033291717000241 (2017).
Loureiro, R. J. et al. Decision-making under risk and theory of mind in adolescent offenders in provisional deprivation of liberty. Trends Psychiatry Psychother. 44, e20200155. https://doi.org/10.47626/2237-6089-2020-0155 (2022).
Beszterczey, S., Nestor, P. G., Shirai, A. & Harding, S. Neuropsychology of decision making and psychopathy in high-risk ex-offenders. Neuropsychology 27, 491–497. https://doi.org/10.1037/a0033162 (2013).
Weissberger, G. H. et al. Subjective socioeconomic status is associated with risk aversion in a community-based cohort of older adults without dementia. Front. Psychol. https://doi.org/10.3389/FPSYG.2022.963418 (2022).
Anselmo, A. et al. Can we rewire criminal mind via non-invasive brain stimulation of prefrontal cortex? Insights from clinical, forensic and social cognition studies. Curr. Psychol. https://doi.org/10.1007/S12144-022-03210-Y (2022).
Hofhansel, L. et al. Morphology of the criminal brain: Gray matter reductions are linked to antisocial behavior in offenders. Brain Struct. Funct. 225, 2017–2028. https://doi.org/10.1007/s00429-020-02106-6 (2020).
Raine, A., Lencz, T., Bihrle, S., LaCasse, L. & Colletti, P. Reduced prefrontal gray matter volume and reduced autonomic activity in antisocial personality disorder. Arch. Gen. Psychiatry 57, 119–127. https://doi.org/10.1001/archpsyc.57.2.119 (2000).
Hoppenbrouwers, S. S. et al. Inhibitory deficits in the dorsolateral prefrontal cortex in psychopathic offenders. Cortex 49, 1377–1385. https://doi.org/10.1016/j.cortex.2012.06.003 (2013).
Shen, B. et al. High-definition tDCS alters impulsivity in a baseline-dependent manner. Neuroimage 143, 343–352. https://doi.org/10.1016/J.NEUROIMAGE.2016.09.006 (2016).
Falcone, B., Wada, A., Parasuraman, R. & Callan, D. E. Individual differences in learning correlate with modulation of brain activity induced by transcranial direct current stimulation. PLoS One 13, e0197192. https://doi.org/10.1371/JOURNAL.PONE.0197192 (2018).
Pripfl, J., Neumann, R., Köhler, U. & Lamm, C. Effects of transcranial direct current stimulation on risky decision making are mediated by “hot” and “cold” decisions, personality, and hemisphere. Eur. J. Neurosci. 38, 3778–3785. https://doi.org/10.1111/ejn.12375 (2013).
Farnad, L., Ghasemian-Shirvan, E., Mosayebi-Samani, M., Kuo, M. F. & Nitsche, M. A. Exploring and optimizing the neuroplastic effects of anodal transcranial direct current stimulation over the primary motor cortex of older humans. Brain Stimul. 14, 622–634. https://doi.org/10.1016/j.brs.2021.03.013 (2021).
Acknowledgements
This work was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)—269953372/GRK2150 and supported by the Brain Imaging Facility of the Interdisciplinary Centre for Clinical Research (IZKF) Aachen within the Faculty of Medicine at the RWTH Aachen University.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Leandra Kuhn: Investigation, Formal analysis, Writing—Original draft, Visualization; Olivia Choy: Conceptualization, Investigation, Writing—Review & Editing; Lara Keller: Investigation, Writing—Review & Editing; Ute Habel: Funding acquisition, Conceptualization, Resources, Writing—Review & Editing; Lisa Wagels: Conceptualization, Methodology, Formal analysis, Supervision, Writing—Review & Editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kuhn, L., Choy, O., Keller, L. et al. Prefrontal tDCS modulates risk-taking in male violent offenders. Sci Rep 14, 10087 (2024). https://doi.org/10.1038/s41598-024-60795-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-60795-z
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.