Abstract
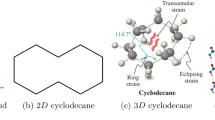
Cyclooctane is a cycloalkane consisting of carbon and hydrogen atoms arranged in a closed ring structure. Cyclooctane chains can be found in various organic compounds and are significant in the field of organic chemistry due to their diverse reactivity and properties. The atom-bond connectivity index (\(\mathcal{A}\mathcal{B}\mathcal{C}\)), the geometric-arithmetic index (\(\mathcal{G}\mathcal{A}\)), the arithmetic–geometric index (\(\mathcal{A}\mathcal{G}\)) and the forgotten index (\(\mathcal{F}\)) are four well-studied molecular descriptors that have found applications in QSPR and QSAR studies. These topological descriptors have shown significant correlations with different physiochemical properties of octane isomers. In this work, the expected values of four degree based topological descriptors for random cyclooctane chains are calculated. An analytical comparison is given between the expected values of \(\mathcal{A}\mathcal{B}\mathcal{C}\), \(\mathcal{G}\mathcal{A}\), \(\mathcal{A}\mathcal{G}\), and \(\mathcal{F}\) indices of random cyclooctane chains.
Similar content being viewed by others
Introduction
A significant branch of mathematics that deals with mathematical models of graphs is called chemical graph theory. It is a branch of mathematics that combines chemistry and graph theory. The physical and chemical properties of molecules correspond with their molecular geometry, which is derived from the vast amount of data used for the analysis. Generally, the characteristics of a molecule obtained through chemical examinations can be efficiently determined by calculating the topological indices. In many cases, theoretical chemistry plays a vital role in chemical graph theory.
Molecular graph theory is a branch of theoretical chemistry that represents chemical compounds, specifically molecules, as graphs. In this context, a graph is a mathematical structure composed of vertices (atoms) and edges (bonds) that connect these vertices. The representation allows for the abstraction and analysis of molecular structures, facilitating the study of various properties and behaviours of chemical compounds. Let \(\Pi =\Pi (V, E)\) be a simple, finite connected graph of order n with vertex set \(V\left(\Pi \right)\) and an edge set \(E\left(\Pi \right)\). The degree of a vertex \({u}_{i}\) is denoted by \(d_{i}\) and is defined as the number of edges incident to it. For undefined notions related to graph theory see1.
Molecular descriptors are the numerical or categorical representations of the structural and chemical features of a molecule. These molecular descriptors are important in the field of computational chemistry and biology. They provide information about molecular size, chemical composition, shape and other relevant properties of the molecular structure which may be used for designing drug, predicting toxicity and quantitative structure property relationship studies. Numerous molecular descriptors have been introduced by different researchers and are important in studying the characteristics of chemical structures. The first distance based topological index was introduces by H. Wiener2, while he was working on the boiling point of paraffin. The Zagreb indices and their variants3 are among the most studied degree based topological indices. They have been used to study branching problem in the early seventeen century. Consider the following general graph invariant
A well-known topological index called the \(\mathcal{F}\) index was introduced by Furtula and Gutman4 in 2015. It is defined as the sum of squares of degrees of the vertices of chemical graphs.
It was observed that the predictive ability of \(\mathcal{F}\) index and the first Zagreb index is same. The correlation coefficient of \(\mathcal{F}\) index for the properties acentric factor and entropy is greater than 0.95.
The Atom-Bond Connectivity index5 was proposed in 1998 by a Cuban mathematician named Ernesto Estrada. The \(\mathcal{A}\mathcal{B}\mathcal{C}\) index is a helpful predictive index that is used to study the formation of heat in alkanes. It is defined as.
Vukičević and Furtula6 introduced a topological index called the geometric-arithmetic index \(\mathcal{G}\mathcal{A}\). The geometric-arithmetic index is a very useful tool in the investigations of QSAR and QSPR studies. It is defined as
The arithmetic–geometric index \(\mathcal{A}\mathcal{G}\)7 was recently introduced as a modification of the well-known geometric–arithmetic index \(\mathcal{G}\mathcal{A}\). This is defined by
For more details on the computations of topological indices for different chemical structures, see8,9,10,11.
In this work, we compute the expected values of four degree based topological indices for the class of random cyclooctane chain: the atom-bond connectivity index, the arithmetic–geometric index, the geometric- arithmetic index, and the forgotten index. An analytical comparison between the expected value of these topological indices with same probability has been given. More precisely, we have proved that the expected value of \(\mathcal{A}\mathcal{B}\mathcal{C}\) index is always less than the expected value of \(\mathcal{G}\mathcal{A}\) index and that the expected value of \(\mathcal{A}\mathcal{G}\) index is less than the expected value of forgotten index.
Random cyclooctane chain
A cyclooctane is a cycloalkane which is a type of saturated hydrocarbon with eight carbon atoms arranged in a cycle. It has a chemical formula of C8H16. Cyclooctane is a stable and nonpolar compound with a simple structure. In organic chemistry, it is used as a reference compound and is part of different organic molecules and reactions. The simple and symmetrical ring structure of cyclooctanes make them ideal model to understand the properties of cyclic hydrocarbons. The study of cyclooctane and its derivatives is important in stereochemistry, particularly when it comes to puckering conformational changes in cycloalkanes. The simple structure and reactivity of cycloocatne make it an important reference point for researchers.
For a long time, chemists gave more attention to the derivatives of saturated hydrocarbons, which are used in drug synthesis, kinetic combustion, and organic synthesis etc. For example, they are used as reagents, synthetic organics. They are also used in the production of adhesives, coatings, and many other purposes. Some scientists got interested in octagonal graphs12. Brunvoll et al.1 studied the number of isomers in octagonal graphs. Many scientists showed their interest in the topological indices of cyclooctanes. Shouliu Wei et. al.13 calculated the Wiener indices of cyclooctanes. Three types of Kirchoff indices of cyclooctanes have been determined by Yoy Linhua et. al. in14. Jia-Bao Liu et. al.15 calculated the Gutman index and Schultz index of the cyclooctane chains. Recently, Zahid Raza et. al.16 calculated some topological index such as harmonic index and sum-connectivity index of cyclooctane chains. Liu H. et. al. have computed some expected values of sombor indices of hexagonal chains, phenylene graphs17. For more details, see8,9,10,11,18,19,20,21,22,23,24,25,26,27,28,29,30.
Materials and methods
The topological indices of the molecular structures that have been derived from their corresponding chemical graphs are called the molecular descriptors. There are many topological indices that have some applications in structural chemistry, especially in QSPR/QSAR research. The cyclooctanes have distinctive physicochemical properties due to saturated and unsaturated hydrocarbons. The cyclooctane chains are made up of a specific arrangement of eight-membered rings. A random cyclooctane chain is an arrangement of octagons such that any two consecutive octagons are attached by an edge in a random way. We use the notation \(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\) m to denote a cyclooctane chain with m number of octagons. For \({\mathcalligra{z}}=1, 2,\) Fig. 1 represents the unique arrangement in \(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\) m. For \({\mathcalligra{z}}=3\), we get four \(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\) m chains as shown in Fig. 2. The four types of cyclooctane chains are denoted by \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z+1 }^{1}\), \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z+1 }^{2}\), \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z+1 }^{3}\) and \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z+1 }^{4}\)(see Fig. 3). Therefore, \(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\) (m; ρ1, ρ2, ρ3) can be attained by stepwise addition of a terminal octagon. The possible four structures that can be made at each step from a random selection (z = 3, 4,…,k) are
-
(i)
\({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\)→ \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\) with probability \({m}_{1}\),
-
(ii)
\({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\)→\({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\) with probability \({m}_{2}\),
-
(iii)
\({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\)→\({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\) with probability \({m}_{3}\),
-
(iv)
\({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\)→ \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\) with probability \(r=1-{m}_{1}{-m}_{2}-{m}_{3}\)
If we assume that the probabilities are constant and independent of the step parameter, then this process is a zeroth-order Markov process. In order to compute the atom-bond connectivity index, the arithmetic–geometric, geometric-arithmetic, and forgotten index of \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}\), we need to find the edge partition of \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}\) depending on the degree of end vertices of each edge. It is easy to see that it contains only (2, 2), (2, 3), and (3, 3)-types of edges. Therefore, the mathematical expression of \(\mathcal{A}\mathcal{B}\mathcal{C}\), \(\mathcal{G}\mathcal{A}\), \(\mathcal{A}\mathcal{G}\), and \(\mathcal{F}\) indices can be written as:
Results
A random cyclooctane chain \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}\) is a local arrangement. Hence, \(\mathcal{F}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)\), \(\mathcal{A}\mathcal{B}\mathcal{C}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)\), \(\mathcal{G}\mathcal{A}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)\), and \(\mathcal{A}\mathcal{G}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)\) are the random variables. We use the notation \({E}_{z}^{\mathcal{F}}=E[\mathcal{F}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)]\), \({E}_{z}^{ABC}=E[\mathcal{A}\mathcal{B}\mathcal{C}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)]\) ,\({E}_{z}^{\mathcal{G}\mathcal{A}}=E[\mathcal{G}\mathcal{A}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)]\), and \({E}_{z}^{\mathcal{A}\mathcal{G}}=E[\mathcal{A}\mathcal{G}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)]\) to denote the expected values of the forgotten, the atom bond connectivity, the geometric arithmetic and the arithmetic geometric indices of \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}\) respectively.
Theorem 1
Let \(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3}\right)\) be the random cyclooctane and \(z\ge 2\). Then
Proof
It is easy to see that \({E}_{2}^{\mathcal{F}}=166\), which is indeed true. For z \(\ge 3,\) there are four possibilities:
-
(i)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\) with probability \({m}_{1}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+5\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+2\), \({x}_{33}\left(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+2\),
From Eq. (5), we have \(\mathcal{F}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)=\mathcal{F}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+102\).
-
(ii)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\) with probability \({m}_{2}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (5), we have \(\mathcal{F}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)=\mathcal{F}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+102\).
-
(iii)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\) with probability \({m}_{3}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (5), we have \(\mathcal{F}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)=\mathcal{F}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+102\).
-
(iv)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\) with probability \(1-{m}_{1}-{m}_{2}-{m}_{3}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (5), we have \(\mathcal{F}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)=\mathcal{F}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+102\)
Thus, we get
Since \({E[E}_{z}^{\mathcal{F}}]={E}_{z}^{\mathcal{F}}\), by applying operator on Eq. (9), we obtain
Using the initial conditions and solving recurrence relation, we get
Theorem 2
Let \(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3}\right)\) be the random cyclooctane and \(z\ge 2\). Then
Proof
It is easy to calculate that \({E}_{2}^{\mathcal{A}\mathcal{B}\mathcal{C}}=\frac{2+24\sqrt{2}}{3}\), which is indeed true. For z \(\ge 3,\) we have four possibilities:
-
(a)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\) with probability \({m}_{1}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+5\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+2\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+2\),
From Eq. (6), we have \(\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)=\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{8+21\sqrt{2}}{6}\).
-
(b)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\) with probability \({m}_{2}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{23}\left({R\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (6), we have \(\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)=\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{2+12\sqrt{2}}{3}.\)
-
(c)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\) with probability \({m}_{3}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (6), we have \(\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)=\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{2+12\sqrt{2}}{3}\).
-
(d)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\) with probability \(1-{m}_{1}-{m}_{2}-{m}_{3}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (6), we have \(\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)=\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{2+12\sqrt{2}}{3}\).
Thus, we get
Since \({E[E}_{z}^{\mathcal{A}\mathcal{B}\mathcal{C}}]={E}_{z}^{\mathcal{A}\mathcal{B}\mathcal{C}}\), by applying operator on Eq. (11), we obtain
Using the initial conditions and solving the recurrence relation, we get
Theorem 3
Let \(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3}\right)\) be the random cyclooctane and \(z\ge 2\). Then
Proof
Observe that \({E}_{2}^{\mathcal{G}\mathcal{A}}=\frac{65+8\sqrt{6}}{5}\) , which is indeed true.. For z \(\ge 3,\) we have four possibilities:
-
(a)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\) with probability \({m}_{1}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+5\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+2\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+2\),
From Eq. (7), we have \(\mathcal{G}\mathcal{A}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)=\mathcal{G}\mathcal{A}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{35+4\sqrt{6}}{5}\).
-
(b)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\) with probability \({m}_{2}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{23}\left({R\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (7), we have \(\mathcal{G}\mathcal{A}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)=\mathcal{G}\mathcal{A}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{25+8\sqrt{6}}{5}.\)
-
(iii)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\) with probability \({m}_{3}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (7), we have \(\mathcal{G}\mathcal{A}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)=\mathcal{G}\mathcal{A}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{25+8\sqrt{6}}{5}\).
-
(iv)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\) with probability \(1-{m}_{1}-{m}_{2}-{m}_{3}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (7), we have \(\mathcal{G}\mathcal{A}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)=\mathcal{G}\mathcal{A}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{25+8\sqrt{6}}{5}\).
Thus, we get
Since \({E[E}_{z}^{\mathcal{G}\mathcal{A}}]={E}_{z}^{\mathcal{G}\mathcal{A}}\), by applying operator on Eq. (13), we obtain
Using the initial conditions and solving recurrence relation, we get
Theorem 4
Let \(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3}\right)\) be the random cyclooctane and \(z\ge 2\). Then
Proof
It is easy to see that \({E}_{2}^{\mathcal{A}\mathcal{G}}=\frac{39+5\sqrt{6}}{3}\) which is indeed true. For z \(\ge 3\), we have four possibilities:
-
(a)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\) with probability \({m}_{1}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+5\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+2\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+2\),
From Eq. (8), we have \(\mathcal{A}\mathcal{G}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{1}\right)=\mathcal{A}\mathcal{G}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{42+5\sqrt{6}}{6}\).
-
(b)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\) with probability \({m}_{2}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{23}\left({R\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (8), we have \(\mathcal{A}\mathcal{G}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{2}\right)=\mathcal{A}\mathcal{G}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{15+5\sqrt{6}}{3}\).
-
(c)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\) with probability \({m}_{3}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (8), we have \(\mathcal{A}\mathcal{G}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{3}\right)=\mathcal{A}\mathcal{G}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{15+5\sqrt{6}}{3}\)
-
(d)
If \({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\to {\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\) with probability \(1-{m}_{1}-{m}_{2}-{m}_{3}\), then
\({x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{22}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+4\), \({x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{23}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\), \({x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)={x}_{33}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+1\),
From Eq. (8), we have \(\mathcal{A}\mathcal{G}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z}^{4}\right)=\mathcal{A}\mathcal{G}\left({\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}}_{z-1}\right)+\frac{15+5\sqrt{6}}{3}\).
Thus, we get
Since, \({E[E}_{z}^{\mathcal{A}\mathcal{G}}]={E}_{z}^{\mathcal{A}\mathcal{G}}\), by applying operator on Eq. (15), we obtain
Using the initial conditions and solving the recurrence relation, we get
Now our focus is the special cyclooctanes such as \({\mathcal{C}\mathcal{Z}}_{\mathcalligra{m}}, {\mathcal{C}\mathcal{L}}_{\mathcalligra{m}}, {\mathcal{C}\mathcal{O}}_{\mathcalligra{m}}\), and \({\mathcal{C}\mathcal{M}}_{\mathcalligra{m}}\), which are shown in Fig. 4. Such cyclooctane chains can be obtained as: \({\mathcal{C}\mathcal{Z}}_{\mathcalligra{m}}= \mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}(m; 1, 0, 0), {\mathcal{C}\mathcal{L}}_{\mathcalligra{m}}=\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}(m; 0, 1, 0), {\mathcal{C}\mathcal{O}}_{\mathcalligra{m}}= \mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}(m; 0, 0, 1),\) and \({\mathcal{C}\mathcal{M}}_{\mathcalligra{m}}= \mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}(m; 0, 0, 0).\) With the help of Theorems 1–4, we can calculate the expected value of these special type of cyclooctane chains.
Corollary 1
For \(m\ge 2\), we have the following results:
-
1.
\(\mathcal{F}\left({\mathcal{C}\mathcal{O}}_{\mathcalligra{m}}\right)=102 {z} -38\)
$$\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{C}\mathcal{O}}_{\mathcalligra{m}}\right)=\left(\frac{8+21\sqrt{2}}{6}\right){z}-(2-\sqrt{2})$$$$\mathcal{G}\mathcal{A}\left({\mathcal{C}\mathcal{O}}_{\mathcalligra{m}}\right)=\left(\frac{35+4\sqrt{6}}{5}\right){z}-1$$$$\mathcal{A}\mathcal{G}\left({\mathcal{C}\mathcal{O}}_{\mathcalligra{m}}\right)=\left(\frac{42+5\sqrt{6}}{6}\right) {z}-1$$ -
2.
\(\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{C}\mathcal{M}}_{\mathcalligra{m}}\right)=\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{C}\mathcal{Z}}_{\mathcalligra{m}}\right)=\mathcal{A}\mathcal{B}\mathcal{C}\left({\mathcal{C}\mathcal{L}}_{\mathcalligra{m}}\right)=\left(\frac{2+12\sqrt{2}}{3}\right) {z}-\frac{2}{3}\)
$$\mathcal{G}\mathcal{A}\left({\mathcal{C}\mathcal{M}}_{\mathcalligra{m}}\right)=\mathcal{G}\mathcal{A}\left({\mathcal{C}\mathcal{Z}}_{\mathcalligra{m}}\right)=\mathcal{G}\mathcal{A}\left({\mathcal{C}\mathcal{L}}_{\mathcalligra{m}}\right)=\left(\frac{25+8\sqrt{6}}{5}\right) {z}+\frac{15-8\sqrt{6}}{5}$$$$\mathcal{A}\mathcal{G}\left({\mathcal{C}\mathcal{M}}_{\mathcalligra{m}}\right)=\mathcal{A}\mathcal{G}\left({\mathcal{C}\mathcal{Z}}_{\mathcalligra{m}}\right)=\mathcal{A}\mathcal{G}\left({\mathcal{C}\mathcal{L}}_{\mathcalligra{m}}\right)=\left(\frac{12+5\sqrt{6}}{3}\right) {z}+\frac{9-5\sqrt{6}}{3}$$
Comparison between the expected values of topological indices
In this section, the expected values of the forgotten index, the arithmetic–geometric index, the geometric-arithmetic index, and the atom-bond connectivity index are analyzed and compared. In Tables 1, 2, 3 and 4, we have computed the expected values of these indices for different values of \({m}_{1}\). Observe that the value of forgotten index is always greater than the expected values of other topological indices. The graphical comparison between the forgotten index, arithmetic–geometric index, geometric-arithmetic index and atom-bond connectivity index for the cyclooctane chains with the same probabilities is shown in Figs. 5, 6, 7 and 8. One can see that the expected value of the forgotten index is always greater than the expected value of arithmetic–geometric index and the expected value of arithmetic–geometric index is always greater than the expected value of geometric-arithmetic index and the expected value of geometric-arithmetic index is greater than the expected value of atom-bond connectivity index. We give an analytical proof of this fact in the next theorems.
Theorem 5
Let \({z}\ge 2,\) then
Proof
It is true for \({z}=2\). Now, let \({z}>2\), by using Theorem 1 and 2, we have
Theorem 6
If \({z}\ge 2,\) then
Proof
It is true for \(\mathcalligra{z}=2\). Let \(\mathcalligra{z}>2\), by using Theorem 2 and 3, we have.
Theorem 7.
\(E[F\left(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)\right]>E[\mathcal{A}\mathcal{G}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)]>E[\mathcal{G}\mathcal{A}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)]>E[\mathcal{A}\mathcal{B}\mathcal{C}(\mathcal{R}\mathcal{C}\mathcal{O}\mathcal{C}\left(z;{m}_{1},{m}_{2},{m}_{3})\right)]\).
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Brunvoll, J., Cyvin, S. J. & Cyvin, B. N. Enumeration of tree-like octagonal systems. J. Math. Chem. 21, 193–196 (1997).
Wiener, H. Structural determination of paraffin boiling points. J. Am. Chem. Soc. 69(1), 17–20 (1947).
Gutman, I., &Trinajstić, N. Graph theory and molecular orbitals. Total φ-electron energy of alternant hydrocarbons. Chem. Phys. Lett. 17(4), 535–538 (1972).
Furtula, B. & Gutman, I. A forgotten topological index. J. Math. Chem. 53(4), 1184–1190 (2015).
Estrada, E., Torres, L., Rodriguez, L. & Gutman, I. An atom-bond connectivity index modelling the enthalpy of formation of alkanes. Indian J. Chem. 37A, 849–855 (1998).
Vukicevic, B. & Furtula, B. Topological index based on the ratios of geometrical and arithmetical means of end-vertex degrees of edges. J. Math. Chem. 46, 1369–1376 (2009).
Shegehall, V. S. & Kanabur, R. Arithmetic-geometric indices of path graph. J. Math. Comput. Sci. 16, 19–24 (2015).
Hui, Z.-H., Yousaf, S., Aslam, A., Binyamin, M. A. & Kanwal, S. On expected values of some degree based topological descriptors of random phenylene chains. Mol. Phys. 121, 16. https://doi.org/10.1080/00268976.2023.2225648 (2023).
Huang, R.-R., Aftab, S., Noureen, S. & Aslam, A. Analysis of porphyrin, PETIM and zinc porphyrin dendrimers by atom-bond sum-connectivity index for drug delivery. Mol. Phys. 121, 15. https://doi.org/10.1080/00268976.2023.2214073 (2023).
Yun, Z., Aslam, A., Kanwal, S., Saeed, S. & Razzaque, A. Physio-chemical properties of HCP(U) and HEX(U) crystal structure lattice via eccentricity dependent chemical invariants. Mol. Phys. 121, 13. https://doi.org/10.1080/00268976.2023.2202267 (2023).
Hui, Z.-H., Naeem, M., Rauf, A. & Aslam, A. Estimating the physicochemical properties of antiemetics using degree-based topological descriptors. Mol. Phys. 121, 5. https://doi.org/10.1080/00268976.2023.2189491 (2023).
Wei, S., Ke, X. & Wang, Y. Wiener indices in random cyclooctane chains. Wuhan Univ. J. Nat. Sci. 23(6), 498–502 (2018).
Hechao, L. I. U., Rangwei, W. U., &Lihua, Y. O. U. Three types of Kirchhoff indices in the random cyclooctane chains. 华南师范大学学报 (自然科学版) 53(2), 96–103 (2021).
Liu, J. B., Gu, J. J. & Wang, K. The expected values for the Gutman index, Schultz index, and some Sombor indices of a random cyclooctane chain. Int. J. Quantum Chem. 123(3), e27022 (2023).
Raza, Z. Zagreb connection indices for some benzenoid systems. Polycycl. Aromat. Compds. 42(4), 1814–1827 (2022).
Raza, Z. & Imran, M. Expected values of some molecular descriptors in random cyclooctane chains. Symmetry 13(11), 2197 (2021).
Huang, G., Kuang, M. & Deng, H. The expected values of Kirchhoff indices in the random polyphenyl and spiro chains. ARS Math. Contemp. 9(2), 197–207 (2015).
Fang, X., You, L. & Liu, H. The expected values of Sombor indices in random hexagonal chains, phenylene chains and Sombor indices of some chemical graphs. Int. J. Quantum Chem. 121(17), e26740 (2021).
Huang, G., Kuang, M. & Deng, H. The expected values of Hosoya index and Merrifield–Simmons index in a random polyphenylene chain. J. Comb. Optim. 32(2), 550–562 (2016).
Liu, H., Zeng, M., Deng, H. & Tang, Z. Some indices in the random spiro chains. Iran. J. Math. Chem. 11(4), 255–270 (2020).
Liu, J. B., Gu, J. J., & Wang, K. The expected values and limiting behaviours for the Gutman index, Schultz index, multiplicative degree-Kirchhoff index and additive degree-Kirchhoff index of a random cyclooctane chain. arXiv preprint arXiv:2203.12923 (2022).
Qi, J., Fang, M. & Geng, X. The expected value for the wiener index in the random spiro chains. Polycycl. Aromat. Compd. 43(2), 1788–1798 (2023).
Qi, J., Ni, J. & Geng, X. The expected values for the Kirchhoff indices in the random cyclooctatetraene and spiro chains. Discrete Appl. Math. 321, 240–249 (2022).
Raza, Z., Arockiaraj, M., Bataineh, M. S., &Maaran, A. Cyclooctane chains: mathematical expected values based on atom degree and sum-degree of Zagreb, harmonic, sum-connectivity, and Sombor descriptors. Eur. Phys. J. Spec. Top. 4, 1–10 (2023).
Sigarreta, S., Sigarreta, S., & Cruz-Suárez, H. On bond incident degree indices of random spiro chains. Polycycl. Aromat. Compds. 8, 1–13 (2022).
Sigarreta, S. C., & Cruz-Suarez, H. Degree based topological indices of a general random chain. arXiv preprint arXiv:2205.06385 (2022).
Wei, S., Ke, X. & Hao, G. Comparing the excepted values of atom-bond connectivity and geometric–arithmetic indices in random spiro chains. J. Inequal. Appl. 2018(1), 45 (2018).
Mansoor, S. et al. Recent advancements in Se- and Te-enriched cocatalysts for boosting photocatalytic splitting of water to produce hydrogen. Res. Chem. Intermed. 49, 3723–3745. https://doi.org/10.1007/s11164-023-05077-5 (2023).
Tayyab, M., Liu, Y., Liu, Z., Xu, Z., Yue, W., Zhou, L., Lei, J. & Zhang, J. A new breakthrough in photocatalytic hydrogen evolution by amorphous and chalcogenide enriched cocatalysts. Chem. Eng. J. 5, 140601 (2022).
Liu, Y. et al. Single-atom Pt loaded zinc vacancies ZnO–ZnS induced type-V electron transport for efficiency photocatalytic H2 evolution. Sol. RRL 5, 2100536. https://doi.org/10.1002/solr.202100536 (2021).
Acknowledgements
This work was supported by the China Henan International Joint Laboratory for Multidimensional Topology and Carcinogenic Characteristics Analysis of Atmospheric Particulate Matter PM 2.5; the Key Research Project in Universities of Henan Province (grant no. 21B110004) and Researchers Supporting Project Number (RSP2024R401), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
All the author contributed equally.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yousaf, S., Iqbal, Z., Tariq, S. et al. Computation of expected values of some connectivity based topological descriptors of random cyclooctane chains. Sci Rep 14, 7713 (2024). https://doi.org/10.1038/s41598-024-57175-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-57175-y
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.