Abstract
The lumbopelvic muscle mechanical properties (MMPs) are clinically relevant, but their dependence on sex remains unknown. Therefore, this study aimed to identify if lumbopelvic MMPs depend on the sex in a young adult population. Thirty-five healthy nulliparous women and 35 healthy men were analyzed (age range: 18–50). Lumbopelvic MMPs, that is, tone, stiffness, elasticity, relaxation and creep, assessed with MyotonPRO®, and pelvic floor (PF) health questionnaires were compared between-sexes. Intra-group correlations between sociodemographic and clinical data, and MMPs were also determined. The MMPs of PF were different between healthy non-climacteric adults of both sexes, with women showing higher values of tone and stiffness and lower values of elasticity and viscoelastic properties than men (in all cases, p < 0.03). At lumbar level, tone and stiffness were higher for men at both sides (in all cases, p < 0.04), and relaxation was lower at left side (p = 0.02). The MMPs showed few correlations with sociodemographic data within women. However, within males, there were positive correlations for PF stiffness and viscoelastic parameters with age, BMI and function (0.334 < r < 0.591) and, at lumbar level, negative correlations for tone and stiffness ( − 0.385 < r < −0.590) and positive correlations for viscoelastic properties (0.564 < r < 0.719), with BMI. This indicated that between-sexes differences of lumbopelvic MMPs depend on the specific location of assessment in healthy non-obese young individuals. Women show higher tone and stiffness and lower elasticity and viscoelasticity than men, at PF level.
Similar content being viewed by others
Introduction
According to the regional interdependence concept1,2, the lumbopelvic region is a functional unit3 that could have specific characteristics depending on sex4,5. The pelvic floor (PF) includes a complex anatomic structure, with fascial components (endopelvic fascia, ligaments, or perineal membrane), muscular components (pelvic floor muscles (PFM) including levator ani muscles, muscles of the urogenital diaphragm, and superficial perineal muscles)6, and neural components (both afferent and efferent nerves, visceral and somatic)7. Despite the skeletal and visceral differences, the histomorphology8 and architecture9 of PFM are similar between female and male individuals. The most relevant anatomic difference is the genital hiatus, which is broader in women10,11. Furthermore, the PFM are considered an integral part of the trunk and lumbopelvic stability12,13. According to this, in women populations, it has been shown that pelvic floor disorders (PFD) can cause pain, disability, and instability of the spine14, and lumbopelvic interventions can improve PFD, such as stress urinary incontinence15. In men, chronic pelvic pain syndrome is associated with spasms in the quadratus lumborum and the iliopsoas and hypomobility of the thoracolumbar spine16. However, other data, such as the lumbopelvic muscle mechanical properties (MMPs), remain unknown, even when their status is relevant in both sexes17,18. The study of MMPs is of interest even in asymptomatic populations, a less studied population19,20, since the consequences of risk factors for PFD on the PFM, such as an alteration of MMPs, may not be evident until later in life21.
Usually, the PFM analysis focuses on strength, assessed with dynamometry22, and muscle activity, assessed with electromyography23, both in women and men populations24. However, these methodologies do not adequately measure muscle tone or other MMPs, as they do not directly measure resistance to change in muscle length18. Moreover, there is neither a single accepted standardized way of measuring muscle tone nor normative values25, and the palpation method is not entirely validated26. Therefore, standardizing a reliable and reproducible examination is needed27. In this line, new technologies allow the evaluate MMPs, as is the case of MyotonPRO (Myoton AS, Tallinn, Estonia), a manual myotonometric device that does not require high levels of expertise28, contrary to other assessment tools, such as ultrasound imaging, and can be used by novices with acceptable results29, following standardized protocols30. Although manual myotonometry has been successfully applied to different clinical states and locations, including lumbar31,32 and PF18,28 regions, no study has determined whether the lumbopelvic MMPs are different between sexes.
Thus, this study aimed to identify if lumbopelvic MMPs depend on sex in a young adult population. Secondarily, intra-sex associations between lumbopelvic MMPs and sociodemographic characteristics were analyzed.
Methods
Design
A cross-sectional case–control study with consecutive case recruitment was designed. The strengthening the reporting of observational studies in epidemiology (STROBE) method was used. The Research Ethics Committee of Córdoba (registration number 5174, October 2021) approved the project. The study was conducted in accordance with the Declaration of Helsinki. All participants signed the informed consent form.
Participants
The inclusion criteria for women were: healthy adult women, between 18 and 50 years old; nulliparous; without any type of delivery or pregnancy. The men group inclusion criterion was: healthy adult men, between 18 and 50 years old. Exclusion criteria for both sexes were: history of surgery at lumbopelvic level; engagement in regular physical training (only recreationally active, without high impact exercises, less than 6 h per week, was allowed); moderate or heavy occupational physical activity33; experience of any spinal pain in the six months before enrolment in the study34; climacteric period; any type of incontinence; body mass index (BMI) > 30 kg/m2. Participants of both sexes were paired by age (± 3 years).
Sample size
To identify a moderate effect size (Cohen d index = 0.7) based on a minimum detectable change (MDC) of 0.86 and a pooled standard deviation of 1.23 for frequency (muscle tone), as previously reported28, an error type I of 0.05 and 0.80 of power, at least 34 women/men per group were necessary (G*power 3.1.9.2, t-test for difference between two independent measures).
Procedures
As recommended, the patient emptied the bladder before the evaluation35. A physical therapist with more than five years of experience assessed the inclusion and exclusion criteria. Two validated questionnaires for women's PF assessment were applied. The Pelvic floor distress inventory (PFDI-20) includes 20 questions divided into three scales according to the symptoms: symptoms of genital prolapse, questions 1 to 6 (POPDI-6); colorectal-anal symptoms, questions 7 to 14 (CRADI-8); and urinary symptoms, questions 15 to 20 (UDI-6). The total score is the sum of the three blocks with a maximum score of 300, where higher scores mean high distress36,37. The Pelvic floor impact questionnaire (PFIQ-7) includes seven questions about the impact of symptoms on activities, relationships, or feelings concerning urinary prolapse (UIQ-7), colorectal-anal conditions (CRAIQ-7), and genital conditions (POPIQ-7). Again, the total score is the sum of the three blocks with a maximum score of 300, where a higher score means a high impact36,37. The International Prostate Symptoms Score (IPSS), which assesses the intensity of lower urinary tract symptoms during the past 30 days, was applied to the men38,39. It consists of 7 questions on a Likert scale with five possible answers. The final score was interpreted as follows: mild (0–7 points), moderate (8–19 points), and severe (20–35 points)40. Sociodemographic and clinical data, such as age, BMI, and level of physical activity according to the Global Physical Activity Questionnaire (GPAQ)41, were also collected.
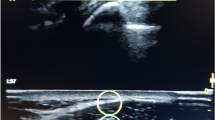
The MMPs measurement protocol of lumbopelvic muscles is described elsewhere28,42,43. A physical therapist, previously trained in myotonometric measures (2 h of training in several body locations), carried out the protocol with a manual tonometer (MyotonPRO® Myoton AS, Tallinn, Estonia). The measurements were taken with the participants in supine position, with the knees flexed and the soles of the feet on the table, to ensure that both lower limbs were symmetrical and relaxed during the measurement. The measurement site was located by visualization and palpation in the largest area of muscle bulk during contraction (verbal order: “stop the flow of urine44) on both sides of the central perineal body. This area was selected because it contains the most contractile portion of the perineal muscles. Once the muscle was relaxed, a mark was placed with a dermographic marker to ensure the measurement site18. At this moment, the men were asked to keep the sexual organs away from the perineal body, without tension or traction of the skin42. The 100 mm long probe of the MyotonPRO® was placed perpendicular to the skin's surface, on the mark location, to perform the measurements (Fig. 1).
Lumbar MMPs were assessed with the patient in prone position for 2 min. The therapist identified the measurement points according to the visual and palpatory approach, at 2.5 cm to the right and left of L5 erector spinae to evaluate the erector spinae. In this case, the 35 mm probe was placed vertically on the skin surface of the muscle belly to perform the test32.
All recordings were performed during five seconds of apnea after exhalation32. The average of three mechanical impulses were applied for each assessment, according to the multiple mode of the MyotonPRO®. The coefficient of variation among the mechanical impulses was lower than 3% for the MMPs28. A randomization process was performed to establish the order of the evaluations (right/left), both in PF and lumbar muscles.
The description of the MMPs can be summarized as45: Frequency, characterizing muscle tension or tone in resting state (Hz); biomechanical properties, such as Stiffness (N/m) and logarithmic Decrement in the amplitude of oscillation (Ø), which defines the inverse of the elasticity; and viscoelastic properties, such as Relaxation time of stress (ms), and Creep (Deborah Number -De-), that characterizes fluidity. The MMPs evaluation has been demonstrated to be valid and reliable in different body locations30, including lumbar (intraclass correlation coefficient: intra-rater, from 0.80 to 0.9943,46,47) and PF (intraclass correlation coefficient: intra-rater, from 0.63 to 0.86; inter-rater, from 0.70 to 0.92, except for creep, that was from 0.40 to 0.4618,28) regions, in healthy individuals.
At the end of the procedures, a Visual analogue scale (VAS) was applied to identify any presence of pain and its intensity during the evaluation28. All procedures lasted less than 30 min for each subject.
Statistical analysis
Frequencies and percentages were used to describe qualitative data. Age, BMI and all outcomes showed a normal distribution of data (Kolmogorov–Smirnov test p > 0.05) and were described by the mean, standard deviation or 95% confidence interval (95%CI), and range. Questionnaire variables were not normally distributed and were described by median and interquartile range. Since some between-sides statistical differences were identified (p < 0.05) for the MMPs when submitted to paired Student t-tests, each side was analyzed separately.
Sociodemographic and clinical data were compared between sexes with unrepeated Student t-tests, except for the level of physical activity, which was compared with X2 test. For the primary aim of the study, unpaired Student t-tests were applied to identify differences in MMPs of PF and lumbar muscles between both sexes. To identify intra-group associations between MMPs and sociodemographic and clinical features in each group, Pearson r or Spearman ρ (rs) coefficients were calculated. Correlations were considered negligible (0.0 to 0.19), fair (0.20 to 0.39), moderate (0.40 to 0.69), strong (0.70 to 0.89), or almost perfect (0.0 to 1.00)[48.
All contrasts were bilateral, and p < 0.05 was considered significant. The IBM-SPSS version 28® software (Chicago, IL, USA) was used for the analyses.
Results
Sociodemographic and clinical data
Thirty-five nulliparous women and 35 men were analyzed. There were no differences between groups in age, while BMI was higher in men (mean difference: −2.07 kg/m2, 95%CI = −3.78, −0.36). Other sociodemographic and clinical data of both groups are included in Table 1. No individual reported pain (VAS = 0) due to the evaluation.
Between sexes differences in lumbopelvic MMPs
All MMPs of PF were different between sexes at both sides, with women showing higher values of tone and stiffness, and lower elasticity and viscoelasticity (in all cases, p < 0.03). At lumbar level, tone and stiffness were higher for men on both sides, and relaxation was lower on the left side. No differences were identified for decrement and creep (Table 2).
Correlations between sociodemographic and clinical status, and lumbopelvic MMPs depending on the sex
For the women group, age showed no correlation with any lumbopelvic MMP. However, at PF level, the BMI was directly related to the viscoelastic properties at both sides in fair to moderate fashion (0.364 < r < 0.452), that is, the higher BMI, the higher relaxation and creep. Tone, stiffness and decrement of PF showed no correlations with BMI.
At lumbar level, the women showed a consistent trend of correlations between BMI and MMPs. Thus, tone and stiffness were moderate and inversely related to BMI (-0.412 < r < -0.480), while viscoelastic properties were moderate and directly related to BMI (0.465 < r < 0.522). The lumbar elasticity showed no relation with BMI. Finally, considering the clinical status of PF, no correlation was identified between PFIQ-7 and PFDI-20 and any lumbopelvic MMP (Table 3).
For the men group, the age was correlated inversely with tone on the right side, and directly with decrement, relaxation and creep in fair to moderate intensity, at both sides of PF (|0.334|< r <|0.446|). The age was also directly related to the decrement at lumbar level (0.395 < r < 0.470). Regarding BMI, all lumbopelvic MMPs, except the PF tone, were related with the BMI in fair to strong intensity at both sides (|0.385|< r <|0.719|). In all cases, the higher BMI, the lower tone and stiffness, while the higher BMI, the higher decrement, relaxation and creep.
The IPSS questionnaire showed some correlations with MMPs only at PF level. Specifically, fair to moderate relations were identified between IPSS, and frequency on the right side and stiffness on the left side in an inverse fashion, and between IPSS, and decrement of the left side, relaxation of the right side and creep of both sides, in a direct fashion (|0.350 < rs < 0.438|) (Table 4).
Discussion
The results showed that all MMPs of PF are different between healthy non-climacteric adults of both sexes, with women showing higher values of tone and stiffness and lower values of elasticity and viscoelastic properties than men. On the contrary, tone, stiffness and relaxation on the left side of lumbar muscles were different between sexes, being men that had higher tone and stiffness and lower relaxation than women. Further, the existence of significant and non-significant correlations between lumbopelvic MMPs and sociodemographic and clinical data depended on each sex. Thus, women lumbopelvic MMPs were not correlated with age, while men MMPs on both locations showed consistent trends of correlations with age, with higher intensity and PF level. Similarly, men showed more and higher intensity correlations between BMI and lumbopelvic MMPs, than women, mainly when PF was considered. Regarding pelvic clinical values, only the MMPs of men at PF level showed some correlations, in this case, with the IPSS. In summary, lumbopelvic MMPs are different between sexes, and these differences depend on the location, with PF being more rigid and less viscoelastic in women and showing relations to age and clinical status in men.
No individual reported pain or any discomfort during the evaluation, which reinforces the idea that the determination of MMPs can be applied in clinical setting, due to its innocuousness.
Location dependence of the between sexes differences of the lumbopelvic MMPs
The magnitude of the differences in MMPs of PF between sexes exceeds the MDC established for these measures28. Thus, although the genital hiatus is broader in women10,11 and is part of the main source of PF weakness, the more robust development of connective tissue in the female PF9 can explain the higher tone and stiffness and the lower elasticity and viscoelasticity of PF in women. In fact, the perineal body and deep perineal muscles are larger, and their bifurcation near the urethra is broader and longer in women than in men. Moreover, the complex of the deep perineal muscle and perineal body forms a perineal membrane in women, while, in men, deep perineal muscle is a relatively small, median structure9. Other between-sexes differences in PFM could also justify the need for higher rigidity of women's PF. For example, the male PF has a much steeper and narrower funnel shape than the female, which levator ani muscles develop a higher working load9. Furthermore, most men are not familiar with PFM contractions and training, probably due to the lower incidence of PFDs and, consequently, the lower necessity to perform specific exercises focused on the PFM49, which could lead to less tone at this level.
Several between-sex differences in MMPs were also identified at lumbar level, with men demonstrating higher tone and stiffness, but below the MDC for these muscles47. It is well known that men’s muscles can be stronger than women's ones50, which can also determine a higher stiffness in different contraction states in men51. All this can be explained by the higher prevalence of slower type-I and type-IIA fibers, which could show less tension and stiffness in females than males52, although, in the current study, the elasticity and viscoelasticity were similar during rest for both sexes. In summary, women's PF is more rigid and less viscoelastic than men's, while lumbar muscles show fewer differences between sexes.
The current results did not show a constant pattern of between-sides asymmetries. Thus, only lumbar relaxation was different depending on the side, but with fewer between-sides differences (< 1.0 ms) than between-sexes differences (> 1.5 ms). Nevertheless, it was reported that the symmetry of MMPs could depend on specific states and disorders. Thus, when asymmetrical processes are considered, such as scoliosis53,54, surgery of Achilles tendon rupture55 or obstetric scars and lesions56,57 associated to episiotomies58 and deliveries42, bilateral differences in MMPs are common. However, when no pathologic or traumatic processes occur, fewer asymmetries are found in PF42 and other regions59, as shown by our sample of both sexes.
Regarding the correlation analysis, on the one hand, MMPs of PF showed no relationship with age and clinical status of PF and some low but significant associations with BMI in women. On the other hand, in men, MMPs were more correlated with age and BMI, and even the clinical status of PF showed correlations with MMPs of PF. This could mean that other features than age or BMI, such as hormonal status60 or exercise34, could be related to the MMPs of PF in nulliparous adult women. Moreover, the absence of relation between the MMPs and age found in women could also be due to the age range of our sample, exclusively composed of young adults. In fact, in healthy subjects, sarcopenia begins around the age of 50 years, which was the high range limit of the current sample61, and the lower muscular development of the female sex and the presence of adrenal androgens after menopause could make sarcopenia less apparent before the climacteric period62.
Clinical relevance of the assessment of lumbopelvic MMPs in both sexes
The assessment of MMPs in the lumbopelvic ring is of clinical relevance, since PF hypertonicity is often associated with urological, gynecological, gastrointestinal and sexual problems as well as chronic pelvic pain in both sexes63, and tone alterations of PFM are related to postoperative male urinary incontinence64. Similarly, stiffness, as the resistance to deformation, is also relevant for PFM assessment65 and should be measured quantitatively25. Thus, a fast, innocuous and reliable MMPs assessment of PFM can help in PF examination27. In this sense, the internal vaginal probes used for dynamometry may induce changes in PFM recruitment by the mere presence of the probes22, and the size of levator hiatus can condition the evaluations66 in women. In men, a digital rectal examination is often used to evaluate PFM, but this invasive technique might cause discomfort, resulting in a lack of cooperation and poor outcomes67. On the contrary, the external application of myotonometry and its innocuousness is described as an advantage68.
Other previous methods used to objectively fvassess perineal body tone included, in men, the Beco perineometer, which measures the introflection values of the perineal body relative to the ischiatic spines. Nevertheless, this mechanical device determines the descent of the perineum in centimeters64, which could be inadequate for muscle assessment. Furthermore, the PFM assessment does not commonly consider bilateral evaluations69,70, as occurs with dynamometry and electromyography, while the determination of MMPs allows individualizing each side, which is considered relevant under certain conditions, such as asymmetric diseases or deliveries6,25,42 as previously commented. Moreover, the assessment of MMPs with manual tonometry has demonstrated relevance in different fields71,72, including lumbar 32,43,46 and PFM18,28,73.
In summary, the differences in MMPs depending on sex suggest that the physiology of these structures could also be different in healthy states, and, consequently, specific analysis approaches could be recommended for each sex. Moreover, the external application of myotonometry, its innocuousness, speed of application, bilateral evaluation, and low need for training29, increase the interest of this method in clinical setting.
Limitations of the study
Some limitations should be recognized. Only healthy non-obese young adults who did not practice regular exercise were included, and all women were nulliparous and non-climacteric. Therefore, the external validity of the results is limited to populations with similar characteristics. All evaluations were performed in the same rest positions, which could increase the consistency of the results, but other positions and states could lead to determining different results74. The menstrual cycle phase was not controlled in the women group, which could be relevant in clinical evaluation60. Future research, considering other ages and clinical states, is recommended.
Conclusions
Between-sexes differences in lumbopelvic MMPs depend on the specific location of the assessment in healthy non-obese young individuals, only recreationally active. Thus, the MMPs of PF differ between healthy non-climacteric adults of both sexes, with women showing higher values of tone and stiffness and lower values of elasticity and viscoelastic properties than men. In contrast, lumbar MMPs show less and lower differences. Only MMPs of men are related to age, BMI and clinical status of PF. Lumbopelvic MMPs evaluation with external tonometry could be relevant in clinical setting, due to its innocuousness and bilateral applicability.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request. The data are not publicly available due to privacy of research participants.
References
Wainner, R. S., Whitman, J. M., Cleland, J. A. & Flynn, T. W. Regional interdependence: a musculoskeletal examination model whose time has come. J. Orthop. Sports Phys. Ther. 37, 658–660 (2007).
Sueki, D. G., Cleland, J. A. & Wainner, R. S. A regional interdependence model of musculoskeletal dysfunction: research, mechanisms, and clinical implications. J. Man. Manip Ther 21, 90–102 (2013).
Ballard, M. T., Drury, C. & Bazrgari, B. Changes in lumbo-pelvic coordination of individuals with and without low back pain when wearing a hip orthosis. Front Sports Act. Living 2, 1–9 (2020).
Janssen, M. M. A., Drevelle, X., Humbert, L., Skalli, W. & Castelein, R. M. Differences in male and female Spino-pelvic alignment in asymptomatic young adults. Spine 34, E826–E832 (2009).
Gombatto, S. P., Collins, D. R., Sahrmann, S. A., Engsberg, J. R. & Van Dillen, L. R. Gender differences in pattern of hip and lumbopelvic rotation in people with low back pain. Clin. Biomech. 21, 263–271 (2006).
Quaghebeur, J., Petros, P., Wyndaele, J. J. & De Wachter, S. Pelvic-floor function, dysfunction, and treatment. Eur. J. Obstetrics and Gynecol. Reprod. Biol. 265, 143–149 (2021).
Avila Gonzalez, C. A. et al. Frontiers in fascia research. J. Bodyw. Mov. Ther. 22, 873–880 (2018).
Jundt, K. et al. Is the histomorphological concept of the female pelvic floor and its changes due to age and vaginal delivery correct?. Neurourol. Urodyn. 24, 44–50 (2005).
Wu, Y. et al. Architecture of structures in the urogenital triangle of young adult males; comparison with females. J. Anat. 233, 447–459 (2018).
Ashton-Miller, J. A. & DeLancey, J. O. L. Functional anatomy of the female pelvic floor. Ann N Y Acad Sci 1101, 266–296 (2007).
Myers, R. P. et al. Puboperineales: Muscular boundaries of the male urogenital hiatus in 3D from magnetic resonance imaging. J. Urol. 164, 1412–1415 (2000).
Eliasson, K., Elfving, B., Nordgren, B. & Mattsson, E. Urinary incontinence in women with low back pain. Man Ther 13, 206–212 (2008).
Ehsani, F., Arab, A. M., Assadi, H., Karimi, N. & Shanbehzadeh, S. Evaluation of pelvic floor muscles activity with and without abdominal maneuvers in subjects with and without low back pain. J Back Musculoskelet Rehabil 29, 241–247 (2016).
Bush, H. M. et al. The association of chronic back pain and stress urinary incontinence. J. Womens Health Phys. Therap. 37, 11–18 (2013).
Wang, Y. et al. Modified lumbo-pelvic exercise to alleviate mild stress urinary incontinence in middle-aged females. Sci Rep 13, (2023).
Archambault-Ezenwa, L., Markowski, A. & Barral, J. P. A comprehensive physical therapy evaluation for male chronic pelvic pain syndrome: a case series exploring common findings. J. Bodyw. Mov. Ther. 23, 825–834 (2019).
Alcaraz-Clariana, S. et al. Paravertebral muscle mechanical properties and spinal range of motion in patients with acute neck or low back pain: a case-control study. Diagnostics 11, 352 (2021).
Davidson, M. J., Bryant, A. L., Bower, W. F. & Frawley, H. C. Myotonometry reliably measures muscle stiffness in the thenar and perineal muscles. Physiother. Can. 69, 104–112 (2017).
Ng, S. F., Lok, M. K., Pang, S. M. & Wun, Y. T. Stress urinary incontinence in younger women in primary care: prevalence and opportunistic intervention. J Womens Health 23, 65–68 (2014).
Shishido, K., Peng, Q., Jones, R., Omata, S. & Constantinou, C. E. Influence of pelvic floor muscle contraction on the profile of vaginal closure pressure in continent and stress urinary incontinent women. J. Urol. 179, 1917–1922 (2008).
McKenzie, S., Watson, T., Thompson, J. & Briffa, K. Stress urinary incontinence is highly prevalent in recreationally active women attending gyms or exercise classes. Int. Urogynecol. J. 27, 1175–1184 (2016).
Auchincloss, C. & McLean, L. Does the presence of a vaginal probe alter pelvic floor muscle activation in young, continent women?. J. Electromyograp. Kinesiol. 22, 1003–1009 (2012).
Smith, M. D., Coppieters, M. W. & Hodges, P. W. Postural response of the pelvic floor and abdominal muscles in women with and without incontinence. Neurourol. Urodyn. 26, 377–385 (2007).
Kim, J. K. et al. A prospectively collected observational study of pelvic floor muscle strength and erectile function using a novel personalized extracorporeal perineometer. Sci. Rep. 11, 1–6 (2021).
Bo, K. et al. An international urogynecological association (IUGA)/International Continence Society (ICS) joint report on the terminology for the conservative and nonpharmacological management of female pelvic floor dysfunction. Neurourol. Urodyn. 36, 221–244 (2017).
Davidson, M. J., Nielsen, P. M. F., Taberner, A. J. & Kruger, J. A. Is it time to rethink using digital palpation for assessment of muscle stiffness?. Neurourol Urodyn 39, 279–285 (2020).
Meister, M. R., Shivakumar, N., Sutcliffe, S., Spitznagle, T. & Lowder, J. L. Physical examination techniques for the assessment of pelvic floor myofascial pain: a systematic review. Am. J. Obstet. Gynecol. 219(497), e1-497.e13 (2018).
Rodrigues-de-Souza, D. P. et al. Absolute and relative reliability of the assessment of the muscle mechanical properties of pelvic floor muscles in women with and without urinary incontinence. Diagnostics 11, 2315 (2021).
Aird, L., Samuel, D. & Stokes, M. Quadriceps muscle tone, elasticity and stiffness in older males: reliability and symmetry using the MyotonPRO. Arch. Gerontol. Geriatr. 55, e31–e39 (2012).
Muckelt, P. E. et al. Protocol and reference values for minimal detectable change of MyotonPRO and ultrasound imaging measurements of muscle and subcutaneous tissue. Sci. Rep. 12, 13654 (2022).
Hu, X. et al. Quantifying paraspinal muscle tone and stiffness in young adults with chronic low back pain: a reliability study. Sci. Rep. 8, 14343 (2018).
Garrido-Castro, J. L. et al. Mechanical properties of lumbar and cervical paravertebral muscles in patients with axial spondyloarthritis: a case - control study. Diagnostics 11, 1662 (2021).
Fimland, M. S., Vie, G., Holtermann, A., Krokstad, S. & Nilsen, T. I. L. Occupational and leisure-time physical activity and risk of disability pension: prospective data from the HUNT study. Norway. Occup. Environ. Med. 75, 23–28 (2018).
Oleksy, L. et al. The influence of pelvis reposition exercises on pelvic floor muscles asymmetry: a randomized prospective study. Medicine 28, e13988 (2019).
Bernard, S. et al. Pelvic-floor properties in women reporting urinary incontinence after surgery and radiotherapy for endometrial cancer. Phys Ther 97, 438–448 (2017).
Barber, M. D., Walters, M. D. & Bump, R. C. Short forms of two condition-specific quality-of-life questionnaires for women with pelvic floor disorders (PFDI-20 and PFIQ-7). Am. J. Obstet Gynecol. 193, 103–113 (2005).
Sánchez-Sánchez, B. et al. Cultural adaptation and validation of the Pelvic floor distress inventory short form (PFDI-20) and pelvic floor impact questionnaire short form (PFIQ-7) Spanish versions. Eur. J. Obstet Gynecol. Reprod. Biol. 170, 281–285 (2013).
Badía, X., García-Losa, M., Dal-Ré, R. & Serra, M. Validation of a harmonized spanish version of the ipss: evidence of equivalence with the original american scale. Adult Urol. 52, 614–620 (1998).
Badia, X., Garcia-Losa, M. & Dal-Re, R. Ten-language translation and harmonization of the international prostate symptom score: developing a methodology for multinational clinical trials. Eur. Urol. 31, 129–140 (1997).
Coll, M. A. V. & Guerra, M. J. G. Incontinencia urinaria, una visión desde la Atención Primaria. Semergen 31, 270–283 (2005).
World Health Organisation. Global physical activity questionnaire (GPAQ). Publications https://www.who.int/publications/m/item/global-physical-activity-questionnaire (2021).
Rodrigues-de-Souza, D. P. et al. Asymmetries of the muscle mechanical properties of the pelvic floor in nulliparous and multiparous women, and men: a cross-sectional study. Symmetry (Basel) 14, 2124 (2022).
Alcaraz-Clariana, S. et al. Paravertebral muscle mechanical properties in patients with axial spondyloarthritis or low back pain: a case-control study. Diagnostics 11, 1898 (2021).
Ben Ami, N., Feldman, R. & Dar, G. Verbal instruction for pelvic floor muscle contraction among healthy young males. Int J Environ Res Public Health 19, 12031 (2022).
Frawley, H. et al. An international continence society (ICS) report on the terminology for pelvic floor muscle assessment. Neurourol. Urodyn. 40, 1217–1260 (2021).
Kelly, J. P. et al. Characterization of tissue stiffness of the infraspinatus, erector spinae, and gastrocnemius muscle using ultrasound shear wave elastography and superficial mechanical deformation. J. Electromyograp. Kinesiol. 38, 73–80 (2018).
Lohr, C., Braumann, K. M., Reer, R., Schroeder, J. & Schmidt, T. Reliability of tensiomyography and myotonometry in detecting mechanical and contractile characteristics of the lumbar erector spinae in healthy volunteers. Eur. J. Appl. Physiol. 118, 1349–1359 (2018).
Akoglu, H. User’s guide to correlation coefficients. Turk J. Emerg. Med. 18, 91–93 (2018).
Wu, M.-L.-Y., Wang, C.-S., Xiao, Q., Peng, C.-H. & Zeng, Tie-Ying. The therapeutic effect of pelvic floor muscle exercise on urinary incontinence after radical prostatectomy: a meta-analysis. Asian J. Androl. 21, 170–176 (2019).
Benfica, P. et al. Reference values for muscle strength: a systematic review with a descriptive meta-analysis. Braz J. Phys. Ther. 22, 355–369 (2018).
Cruz-Montecinos, C. et al. Gender differences on effects of forearm rotation on compressive stiffness of flexor carpi ulnaris during submaximal handgrip contractions. J. Anat. https://doi.org/10.1111/joa.13914 (2023).
Haizlip, K. M., Harrison, B. C. & Leinwand, L. A. Sex-based differences in skeletal muscle kinetics and fiber-type composition. Physiology 30, 30–39 (2015).
Burwell, R. G. & Dangerfield, P. H. Pathogenesis of progressive adolescent idiopathic scoliosis platelet activation and vascular biology in immature vertebrae: an alternative molecular hypothesis. Acta Orthop. Belg. 72, 247–260 (2006).
Liu, Y. et al. Asymmetric biomechanical characteristics of the paravertebral muscle in adolescent idiopathic scoliosis. Clin. Biomech. 65, 81–86 (2019).
Bravo-Sánchez, A., Abián, P., Jimenez, F. & Abián-Vicén, J. Structural and mechanical properties of the Achilles tendon in senior badminton players: operated vs non-injured tendons. Clin. Biomech. 85, 105366 (2021).
Gilbert, I., Gaudreault, N. & Gaboury, I. Exploring the effects of standardized soft tissue mobilization on the viscoelastic properties, pressure pain thresholds, and tactile pressure thresholds of the cesarean section scar. J. Integrat. Complement. Med. 28, 355–362 (2022).
Driusso, P. et al. Are there differences in short-term pelvic floor muscle function after cesarean section or vaginal delivery in primiparous women? A systematic review with meta-analysis. Int. Urogynecol. J. 31, 1497–1506 (2020).
Začesta, V., Rezeberga, D., Plaudis, H., Drusany-Staric, K. & Cescon, C. Could the correct side of mediolateral episiotomy be determined according to anal sphincter EMG?. Int. Urogynecol. J. 29, 1501–1507 (2018).
Bravo-Sánchez, A., Abián, P., Jiménez, F. & Abián-Vicén, J. Myotendinous asymmetries derived from the prolonged practice of badminton in professional players. PLoS One 14, 1–13 (2019).
Khowailed, I. A., Lee, Y. & Lee, H. Assessing the differences in muscle stiffness measured with shear wave elastography and myotonometer during the menstrual cycle in young women. Clin. Phys. Funct. Imag. 42, 320–326 (2022).
Deschenes, M. R. Effects of aging on muscle fibre type and size. Sports Med. 34, 809–824 (2004).
Doherty, T. J. The influence of aging and sex on skeletal muscle mass and strength. Curr Opin Clin Nutr Metab Care 4, (2001).
van Reijn-Baggen, D. A. et al. Pelvic floor physical therapy for pelvic floor hypertonicity: a systematic review of treatment efficacy. Sex Med. Rev. 10, 209–230 (2022).
Rigatti, L. et al. The relationship between continence and perineal body tone before and after radical prostatectomy: a pilot study. Neurourol. Urodyn. 32, 215–223 (2013).
Pool-Goudzwaard, A. et al. Contribution of pelvic floor muscles to stiffness of the pelvic ring. Clin. Biomechan. 19, 564–571 (2004).
Tailor, V. K., Bhide, A. A., Fernando, R., Digesu, G. A. & Khullar, V. Does size matter? Perineometer and digital examination of a model levator hiatus. Neurourol. Urodyn. 39, 1338–1344 (2020).
Bø, K. & Hilde, G. Retest reliability of surface electromyography on the pelvic floor musclesGrape, HH Dedering, A Jonasson. AF. Neurourol. Urodyn. 32, 215–223 (2013).
Proulx, L. et al. Women with chronic pelvic pain demonstrate increased lumbopelvic muscle stiffness compared to asymptomatic controls. J. Womens Health 32, 239–247 (2023).
Halski, T., Ptaszkowski, K., Slupska, L., Dymarek, R. & Paprocka-Borowicz, M. Relationship between lower limbs position and pelvic floor muscle surface electromyography activity in menopausal women: a prospective observational study. Clin. Interv. Aging 12, 75–83 (2017).
Ptaszkowski, K., Małkiewicz, B., Zdrojowy, R., Paprocka-Borowicz, M. & Ptaszkowska, L. Assessment of the elastographic and electromyographic of pelvic floor muscles in postmenopausal women with stress urinary incontinence symptoms. Diagnostics 11, 2051 (2021).
Schneebeli, A., Falla, D., Clijsen, R. & Barbero, M. Myotonometry for the evaluation of Achilles tendon mechanical properties: a reliability and construct validity study. BMJ Open Sport Exerc. Med. 6, e000726 (2020).
Drenth, H. et al. Psychometric properties of the Myotonpro in dementia patients with Paratonia. Gerontology 64, 401–412 (2018).
Ramírez-Jiménez, M., Alburquerque-Sendín, F., Garrido-Castro, J. L. & Rodrigues-de-Sousa, D. P. Effects of hypopressive exercises on post-partum abdominal diastasis, trunk circumference, and mechanical properties of abdominopelvic tissues: a case series. Physiother. Theory Pract. 39, 49–60 (2021).
Sapsford, R. R., Richardson, C. A., Maher, C. F. & Hodges, P. W. Pelvic floor muscle activity in different sitting postures in continent and incontinent women. Arch. Phys. Med. Rehabil. 89, 1741–1747 (2008).
Author information
Authors and Affiliations
Contributions
D.P.R.-S., F.A.-S. and J.L.G.-C. conceived and designed the research. D.P.R.-S., A.C.-C., S.A.-C., L.G.-L. and M.C.C.-P. conducted the material preparation and data collection. A.C.-C., S.A.-C., L.G.-L., M.C.C.-P., F.A.-S. and J.L.G.-C. constructed and treated the data base. D.P.R.-S., F.A.-S. and J.L.G.-C. analyzed the data. D.P.R.-S., A.C.-C. and F.A.-S. wrote the first draft of the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rodrigues-de-Souza, D.P., Casas-Castro, A., Carmona-Pérez, M. et al. Between-sexes differences in lumbopelvic muscle mechanical properties of non-climacteric adults: a cross-sectional design. Sci Rep 13, 21612 (2023). https://doi.org/10.1038/s41598-023-48984-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-48984-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.